Abstract
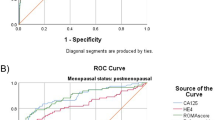
A case–control study included 83 ovarian cancer patients, 76 patients with benign ovarian tumors, and 79 healthy control subjects in the control group. Objective of the study is to analyze biomarker concentrations included in the two novel ovarian tumor differential diagnostic tests (risk of ovarian malignancy algorithm and OVA1) approved by food and drug administration in patients with ovarian tumors and to establish a new ovarian cancer risk assessment algorithm in conjunction with ultrasound score and menopausal status. Ovarian cancer diagnostic tests, developed in the training setting, were evaluated in the independent validation settings of Asian Pacific ovarian cancer biomarker research group study population and Denmark Pelvic Mass project population. Results show that mean serum concentrations of cancer antigen 125 (CA125), human epididymis secretory protein 4 (HE4), and beta-2-microglobulin were upregulated, but apolipoprotein A1, transferrin, and transthyretin were downregulated among ovarian cancer patients. When only one biomarker was introduced in the logistic regression analysis, together with ultrasonographic score and menopausal status, HE4 (area under the curve (AUC) = 0.930; 95 % confidence interval (CI) 0.891–0.969) was more accurate than CA125 (AUC = 0.902; 95 % CI 0.855–0.949) in ovarian cancer diagnostic, but when both biomarkers were included in the logistic regression analyses, ovarian cancer diagnostic accuracy was increased (AUC = 0.939; 95 % CI 0.902–0.977). In conclusions, human epididymis secretory protein 4 and CA125 in combination with ultrasonographic features and menopausal status has high accuracy in ovarian tumor differentiation.





Similar content being viewed by others
References
Abraham J. OVA1 test for preoperative assessment of ovarian cancer. Community Oncol. 2010;7:249–50.
Heintz AP, Odicino F, Maisonneuve P, Benedet JL, Creasman WT, Ngan HY, Pecorelli S, Beller U. Carcinoma of the ovary. Int J Gynaecol Obstetetrics. 2006;95:161–S192.
Hellstrom I, Raycraft J, Hayden-Ledbetter M, Ledbetter JA, Schummer M, McIntosh M, Drescher C, Urban N, Hellström KE. The HE4 (WFDC2) protein is a biomarker for ovarian cancer. Cancer Res. 2003;63:3695–700.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, Thun MJ. Cancer statistics, 2006. CA Cancer J Clin. 2006;56:106–30.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007;57(1):43–66.
Kadija S, Stefanovic A, Jeremic K, Radojevic MM, Nikolic L, Markovic I, Atanackovic J. The utility of human epididymal protein 4, cancer antigen 125, and risk for malignancy algorithm in ovarian cancer and endometriosis. Int J Gynecol Cancer. 2012;22(2):238–44.
Kozak KR, Su F, Whitelegge JP, Faull K, Reddy S, Farias-Eisner R. Characterization of serum biomarkers for detection of early stage ovarian cancer. Proteomics. 2005;17:4589–96.
Midulla C, Manganaro L, Longo F, Viggiani V, Frati L, Granato T, Anastasi E. HE4 combined with MDCT imaging is a good marker in the evaluation of disease extension in advanced epithelial ovarian carcinoma. Tumour Biol. 2012. [Epub ahead of print].
Molina R, Escudero JM, Augé JM, Filella X, Foj L, Torné A, Lejarcegui J, Pahisa J. HE4 a novel tumour marker for ovarian cancer: comparison with CA 125 and ROMA algorithm in patients with gynaecological diseases. Tumour Biol. 2011;32(6):1087–95.
Moore RG, Miller MC, Steinhoff MM, Skates SJ, Lu KH, Lambert-Messerlian G, Bast Jr RC. Serum HE4 levels are less frequently elevated than CA125 in women with benign gynecologic disorders. Am J Obstet Gynecol. 2012;206(4):351.e1–8. Epub 2011 Dec 30.
Moore RG, McMeekin DS, Brown AK, DiSilvestro P, Miller MC, Allard WJ, Gajewski W, Kurman R, Bast Jr RC, Skates SJ. A novel multiple marker bioassay utilizing HE4 and CA125 for the prediction of ovarian cancer in patients with a pelvic mass. Gynecol Oncol. 2009;112(1):40–6.
Nosov V, Su F, Amneus M, Birrer M, Robins T, Kotlerman J, Reddy S, Farias-Eisner R. Validation of serum biomarkers for detection of early-stage ovarian cancer. Am J Obstet Gynecology. 2009;200(6):639.e1–5.
Shapiro S, Wilk M. An analysis of variance test for normality (complete samples). Biometrika. 1965;52:591–611.
Tingulstad S, Hagen B, Skjeldestad FE, Halvorsen T, Nustad K, Onsrud M. The risk-of-malignancy index to evaluate potential ovarian cancers in local hospitals. Obstet Gynecol. 1999;93(3):448–52.
Acknowledgments
Study was performed during PhD studies owing to project for PhD studies promotion in Latvia (P. Stradins University project number 2009/0147/1DP/1.1.2.1.2/09/IPIA/VIAA/009) and Latvian University project for early cancer diagnostics (project number 2009/0220/1DP/1.1.1.2.0/09/APIA/VIAA/016). Authors thank Beth Schodin, Estrid Hogdall, and Claus Hogdall for their assistance in novel ovarian cancer discriminatory test evaluation in independent study populations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Macuks, R., Baidekalna, I. & Donina, S. An ovarian cancer malignancy risk index composed of HE4, CA125, ultrasonographic score, and menopausal status: use in differentiation of ovarian cancers and benign lesions. Tumor Biol. 33, 1811–1817 (2012). https://doi.org/10.1007/s13277-012-0440-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0440-1