Abstract
Background
Sevoflurane (Sev) is a widely used general anesthetic that can cause neurotoxicity. Histone acetylation is a vital epigenetic mechanism responding to environmental stresses.
Objective
This study was conducted to analyze the role of histone deacetylase 3 (HDAC3) in Sev-induced neurotoxicity and provide a theoretical reference for the treatment of anesthetic neurotoxicity.
Results
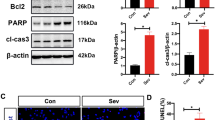
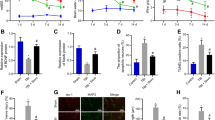
HDAC3 was upregulated in Sev-treated HT-22 cells. Inhibition of HDAC3 upregulated cell viability and downregulated apoptosis, oxidative stress and secretion of inflammatory cytokines. HDAC3 reduced H3K27ac and H3K9ac levels on the TrkC promoter to inhibit TrkC expression. TrkC downregulation reversed the alleviative role of si-HDAC3 in Sev-induced neurotoxicity.
Conclusion
HDAC3 enhanced Sev-induced neurotoxicity by reducing H3K27ac and H3K9ac to repress TrkC.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Apai C, Shah R, Tran K, Pandya Shah S (2021) Anesthesia and the developing brain: a review of sevoflurane-induced neurotoxicity in pediatric populations. Clin Ther 43:762–778
Chai G et al (2022) Sevoflurane inhibits histone acetylation and contributes to cognitive dysfunction by enhancing the expression of ANP32A in aging mice. Behav Brain Res 431:113949
Chelladurai P et al (2021) Targeting histone acetylation in pulmonary hypertension and right ventricular hypertrophy. Br J Pharmacol 178:54–71
Chen YT et al (2012) Expression patterns of histone deacetylases in experimental stroke and potential targets for neuroprotection. Clin Exp Pharmacol Physiol 39:751–758
D’Mello SR (2020) Histone deacetylase-3: Friend and foe of the brain. Exp Biol Med (maywood) 245:1130–1141
De Hert S, Moerman A (2015) Sevoflurane. F1000Res 4: 626
Dedoni S, Marras L, Olianas MC, Ingianni A, Onali P (2021) The neurotrophin receptor trkc as a novel molecular target of the antineuroblastoma action of valproic acid. Int J Mol Sci 22:7790
Gonzalez S et al (2022) Small molecule modulation of TrkB and TrkC neurotrophin receptors prevents cholinergic neuron atrophy in an Alzheimer’s disease mouse model at an advanced pathological stage. Neurobiol Dis 162:105563
Guo J et al (2017) Antidepressant imipramine protects bupivacaine-induced neurotoxicity in dorsal root ganglion neurons through coactivation of TrkA and TrkB. J Cell Biochem 118:3960–3967
Gupta R, Ambasta RK, Kumar P (2021) Histone deacetylase in neuropathology. Adv Clin Chem 104:151–231
Islam MT (2017) Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol Res 39:73–82
Jia J et al (2020) The Role of Histone acetylation in the sevoflurane-induced Inhibition of neurogenesis in the hippocampi of young mice. Neuroscience 432:73–83
Jin H et al (2020) Paeonol attenuates isoflurane anesthesia-induced hippocampal neurotoxicity via modulation of JNK/ERK/P38MAPK pathway and regulates histone acetylation in neonatal rat. J Matern Fetal Neonatal Med 33:81–91
Lee YM, Song BC, Yeum KJ (2015) Impact of volatile anesthetics on oxidative stress and inflammation. Biomed Res Int 2015:242709
Liao Y et al (2020) HDAC3 inhibition ameliorates ischemia/reperfusion-induced brain injury by regulating the microglial cGAS-STING pathway. Theranostics 10:9644–9662
Liu M, Li M, Zhou Y, Zhou Q, Jiang Y (2020) HSP90 inhibitor 17AAG attenuates sevoflurane-induced neurotoxicity in rats and human neuroglioma cells via induction of HSP70. J Transl Med 18:166
Liu Y et al (2018) Galantamine improves cognition, hippocampal inflammation, and synaptic plasticity impairments induced by lipopolysaccharide in mice. J Neuroinflammation 15:112
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Louis Sam Titus ASC, Sharma D, Kim MS, D’Mello SR (2019) The Bdnf and Npas4 genes are targets of HDAC3-mediated transcriptional repression. BMC Neurosci 20:65
Lu LX, Yon JH, Carter LB, Jevtovic-Todorovic V (2006) General anesthesia activates BDNF-dependent neuroapoptosis in the developing rat brain. Apoptosis 11:1603–1615
Norwood J, Franklin JM, Sharma D, D’Mello SR (2014) Histone deacetylase 3 is necessary for proper brain development. J Biol Chem 289:34569–34582
Oztas E, Abudayyak M, Celiksoz M, Ozhan G (2019) Inflammation and oxidative stress are key mediators in AKB48-induced neurotoxicity in vitro. Toxicol in Vitro 55:101–107
Parhamifar L, Andersen H, Moghimi SM (2019) Lactate dehydrogenase assay for assessment of polycation cytotoxicity. Methods Mol Biol 1943:291–299
Reichardt LF (2006) Neurotrophin-regulated signalling pathways. Philos Trans R Soc Lond B Biol Sci 361:1545–1564
Sartor GC et al (2019) Enhancement of BDNF expression and memory by HDAC inhibition requires BET bromodomain reader proteins. J Neurosci 39:612–626
Scott-Solomon E, Kuruvilla R (2018) Mechanisms of neurotrophin trafficking via Trk receptors. Mol Cell Neurosci 91:25–33
Sun M, Xie Z, Zhang J, Leng Y (2022) Mechanistic insight into sevoflurane-associated developmental neurotoxicity. Cell Biol Toxicol 38:927–943
Xiao H, Liu B, Chen Y, Zhang J (2016) Learning, memory and synaptic plasticity in hippocampus in rats exposed to sevoflurane. Int J Dev Neurosci 48:38–49
Yang L et al (2022) HDAC3 of dorsal hippocampus induces postoperative cognitive dysfunction in aged mice. Behav Brain Res 433:114002
Yu L et al (2018) Lentivirus-mediated HDAC3 inhibition attenuates oxidative stress in APPswe/PS1dE9 mice. J Alzheimers Dis 61:1411–1424
Yu X, Yu W, Wu L, Yang W, Lu Y (2021) Chitotriosidase attenuates brain inflammation via HDAC3/NF-kappaB pathway in D-galactose and aluminum-induced rat model with cognitive impairments. Neurosci Res 172:73–79
Yu Z et al (2020) Enriched environment improves sevoflurane-induced cognitive impairment during late-pregnancy via hippocampal histone acetylation. Braz J Med Biol Res 53:e9861
Zhang J, Chen Z, Luo X, Yang Z (2020a) TrkC overexpression protects sevoflurane-induced neurotoxicity in human induced pluripotent stem cell-derived neurons. Dev Neurosci 42:105–113
Zhang N, Wang D, Yang X, Hou D (2020b) Long noncoding RNA small nucleolar RNA host gene 1 contributes to sevoflurane-induced neurotoxicity through negatively modulating microRNA-181b. NeuroReport 31:416–424
Zhou X et al (2016) Dose-dependent effects of sevoflurane exposure during early lifetime on apoptosis in hippocampus and neurocognitive outcomes in Sprague-Dawley rats. Int J Physiol Pathophysiol Pharmacol 8:111–119
Zhu X et al (2017) HDAC3 negatively regulates spatial memory in a mouse model of Alzheimer’s disease. Aging Cell 16:1073–1082
Acknowledgement
Not applicable.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: JZ, XF, Dan Wang; Methodology: JZ, XF; Data curation: JZ, XF, DW; Validation: JZ; Supervision: XF; Writing – original draft: JZ; Writing – review and editing: JZ, XF, DW.
Corresponding author
Ethics declarations
Conflict of interest
Jiegang Zhou, Xinwei Feng and Dan Wang declare that there is no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, J., Feng, X. & Wang, D. HDAC3 deacetylates H3K27ac and H3K9ac on the TrkC promoter to exacerbate sevoflurane-induced neurotoxicity. Mol. Cell. Toxicol. (2023). https://doi.org/10.1007/s13273-023-00394-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s13273-023-00394-7