Abstract
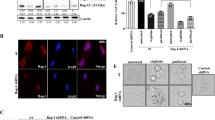
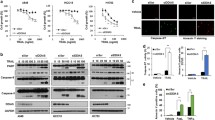
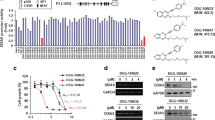
Bcl-2 interacting cell death suppressor (BIS), an anti-stress and ant-apoptotic protein, has been reported to be expressed at high levels in various cancers. In a previous study, we reported on a high level of expression of BIS in non small cell lung cancer tissues. To explore the significance this finding in lung cancer, in this study, we investigated the effect of BIS depletion on the survival of A549 cells upon treatment with anti-tumor agents. BIS knock out A549 cells, prepared by the CRISPR/Cas9 system, revealed a substantial decrease in survival to cisplatin treatment. Western blotting and quantitative real time PCR assays indicated that, among the anti-apoptotic Bcl-2 family proteins, the expression of Mcl-1 was decreased by BIS depletion at the protein level not at the mRNA level. Since BIS expression has been shown to be regulated by HSF1, we subsequently illustrated the sensitization effect of KRIBB11, a HSF1 inhibitor, on cisplatin-induced toxicity in A549 cells, accompanied by a decrease in both BIS and Mcl-1 expression. Our results suggest that BIS-mediated Mcl-1 stabilization represents a potential therapeutic target for cancer therapy.
Similar content being viewed by others
References
Jemal, A. et al. Global cancer statistics. CA Cancer J Clin 61:69–90 (2011).
Youlden, D. R., Cramb, S. M. & Baade, P. D. Th International Epidemiology of Lung Cancer: geographical distribution and secular trends. J Thorac Oncol 3:819–831 (2008).
Molina, J. R., Yang, P., Cassivi, S. D., Schild, S. E. & Adjei, A. A. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc 83:584–594 (2008).
Sandler, A. et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 355:2542–2550 (2006).
Scagliotti, G. V. et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol 26:3543–3551 (2008).
Schiller, J. H. et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346:92–98 (2002).
Seah, S. et al. Activation of surrogate death receptor signaling triggers peroxynitrite-dependent execution of cisplatin-resistant cancer cells. Cell Death Dis 6:e1926 (2015).
Lee, J. H. et al. Bis, a Bcl-2-binding protein that synergizes with Bcl-2 in preventing cell death. Oncogene 18:6183–6190 (1999).
Zhu, H., Liu, P. & Li, J. BAG3: a new therapeutic target of human cancers? Histol Histopathol 27:257–261 (2012).
Rosati, A., Graziano, V. De Laurenzi, V., Pascale, M. & Turco, M. C. BAG 3: a multifaceted protein that regulates major cell pathways. Cell Death Dis 2:e141 (2011).
Jung, S. E. et al. Down-modulation o Bis sensitizes cell death in C6 glioma cells induced by oxygen-glucose deprivation. Brain Res 1349:1–10 (2010).
Romano, M. F. et al. BAG3 protein controls B-chronic lymphocytic leukaemia cell apoptosis. Cell Death Differ 10:383–385 (2003).
Liu, P., Xu, B., Li, J. & Lu, H. BAG3 gene silencing sensitizes leukemic cells to Bortezomib-induced apoptosis. FEBS Lett 583:401–406 (2009).
Jacobs, A. T. & Marnett, L. J. HSF1-mediated BAG3 expression attenuates apoptosis in 4-hydroxynonenal-treated colon cancer cells via stabilization of anti-apoptotic Bcl-2 proteins. J Biol Chem 284:9176–9183 (2009).
Chiappetta, G. et al. The antiapoptotic protein BAG3 is expressed in thyroid carcinomas and modulates apoptosis mediated by tumor necrosis factor-related apoptosis-inducing ligand. J Clin Endocrinol Metab 92:1159–1163 (2007).
Li, N. et al. PKCdelta-mediated phosphorylation of BAG3 at Ser187 site induces epithelial-mesenchymal transition and enhances invasiveness in thyroid cancer FRO cells. Oncogene 32:4539–4548 (2013).
Festa, M. et al. BAG3 protein is overexpressed in human glioblastoma and is a potential target for therapy. Am J Pathol 178:2504–2512 (2011).
Rosati, A. et al. Expression of the antiapoptotic protein BAG 3 is a feature of pancreatic adenocarcinoma and its overexpression is associated with poorer survival. Am J Pathol 181:1524–1529 (2012).
Falco, A. et al. BAG3 is a novel serum biomarker for pancreatic adenocarcinomas. Am J Gastroenterol 108:1178–1180 (2013).
Rosati, A. et al. BAG3 promotes pancreatic ductal adenocarcinoma growth by activating stromal macrophages. Nat Commun 6:8695 (2015).
Zhang, Y., Wang, J. H., Lu, Q. & Wang, Y. J. Bag3 promotes resistance to apoptosis through Bcl-2 family members in non-small cell lung cancer. Oncol Rep 27:109–113 (2012).
Chiappetta, G. et al. The anti-apoptotic BAG3 protein is expressed in lung carcinomas and regulates small cell lung carcinoma (SCLC) tumor growth. Oncotarget 5:6846–6853 (2014).
Yeo, C. D. et al. Bis Expression in Patients with Surgically Resected Lung Cancer and its Clinical Significance. Ann Surg Oncol 22(suppl 3):1365–1370 (2015).
Isaka, T., Nestor, A. L., Takada, T. & Allison, D. C. Chromosomal variations within aneuploid cancer lines. J Histochem Cytochem 51:1343–1353 (2003).
Yoon, Y. J. et al. KRIBB11 inhibits HSP70 synthesis through inhibition of heat shock factor 1 function by impairing the recruitment of positive transcription elongation factor b to the hsp70 promoter. J Biol Chem 286: 1737–1747 (2011).
Mani, J. et al. Knockdown of BAG3 sensitizes bladder cancer cells to treatment with the BH3 mimetic ABT-737. World J Urol (2015).
Li, S. et al. BAG3 promoted starvation-induced apoptosis of thyroid cancer cells via attenuation of autophagy. J Clin Endocrinol Metab 99:E2298–2307 (2014).
Kong, D. H. et al. BAG3 sensitizes cancer cells exposed to DNA damaging agents via direct interaction with GRP78. Biochim Biophys Acta 1833: 3245–3253 (2013).
Boiani, M., Daniel, C., Liu, X., Hogarty, M. D. & Marnett, L. J. The stress protein BAG3 stabilizes Mcl-1 protein and promotes survival of cancer cells and resistance to antagonist ABT-737. J Biol Chem 288:6980–6990 (2013).
Moretti, L., Li, B., Kim, K. W., Chen, H. & Lu, B. AT-101, a pan-Bcl-2 inhibitor, leads to radiosensitization of non-small cell lung cancer. J Thorac Oncol 5:680–687 (2010).
Mei, H., Lin, Z., Wang, Y., Wu, G. & Song, Y. Autophagy inhibition enhances pan-Bcl-2 inhibitor AT-101-induced apoptosis in non-small cell lung cancer. Neoplasma 61:186–192 (2014).
Ren, T. et al. Small-molecule BH3 mimetic and pan-Bcl-2 inhibitor AT-101 enhances the antitumor efficacy of cisplatin through inhibition of APE1 repair and redox activity in non-small-cell lung cancer. Drug Des Devel Ther 9:2887–2910 (2015).
Akgul, C., Moulding, D. A., White, M. R. & Edwards, S. W. In vivo localisation and stability of human Mcl-1 using green fluorescent protein (GFP) fusion proteins. FEBS Lett 478:72–76 (2000).
An, S., He, D., Wagner, E. & Jiang, C. Peptide-lik Polymers Exerting Effective Glioma-Targeted siRNA Delivery and Release for Therapeutic Application. Small 11:5142–5150 (2015).
Wang, B. K. et al. Gold-nanorods-siRNA nanoplex for improved photothermal therapy by gene silencing. Biomaterials 78:27–39 (2016).
Franceschelli, S. et al. Bag3 gene expression is regulated by heat shock factor 1. J Cell Physiol 215:575–577 (2008).
Du, Z. X. et al. Proteasome inhibitor MG132 induces BAG3 expression through activation of heat shock factor 1. J Cell Physiol 218:631–637 (2009).
Yoo, H. J., Im, C. N., Youn, D. Y., Yun, H. H. & Lee, J. H. Bis i Induced by Oxidative Stress via Activation of HSF1. Korean J Physiol Pharmacol 18:403–409 (2014).
Zhang, X. & Qian, S. B. Chaperone-mediated hierarchical control in targeting misfolded proteins to aggresomes. Mol Biol Cell 22:3277–3288 (2011).
Behl, C. BAG3 and friends: co-chaperones in selective autophagy during aging and disease. Autophagy 7:795–798 (2011).
Yoon, T., Kang, G. Y., Han, A. R., Seo, E. K. & Lee, Y. S. 2,4-Bis(4-hydroxybenzyl)phenol inhibits heat shock transcription factor 1 and sensitizes lung cancer cells to conventional anticancer modalities. J Nat Prod 77:1123–1129 (2014).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8:2281–2308 (2013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cui, M.N., Yun, HH., Lee, N.E. et al. Depletion of BIS sensitizes A549 cells to treatment with cisplatin. Mol. Cell. Toxicol. 12, 63–71 (2016). https://doi.org/10.1007/s13273-016-0009-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-016-0009-y