Abstract
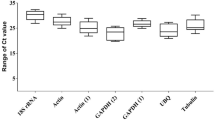
Quantitative real-time PCR (qRT-PCR) is a standard method to measure gene expression in function exploring. Accurate and reproducible data of qRT-PCR requires appropriate reference genes, which are stably expressed under different experimental conditions. However, no housekeeping genes were validated as internal controls for qRT-PCR in Sinonovacula constricta. In this study, we classified the transcriptome data of two tissues for Vibrio infection and Cd2+ stress into ten clusters based on the gene expression patterns. Among them, cluster 5 had the most stable gene expression patterns regardless of tissues and treatments as the database for candidate reference genes. A total of 55 orthologs of classical housekeeping genes in the clam transcriptome were annotated. Combined the expression profiles and housekeeping genes in S. constricta, we chose eight candidate reference genes and validated their expression in Vibrio-infected samples and different tissues by qRT-PCR. Their expression stability was analyzed by three different algorithms geNorm, NormFinder and BestKeeper. Although the rank of the eight candidate reference genes is different in different treatments using different software, RS9 could be the best reference genes for normalization of qRT-PCR expression data in S. constricta under various treatments considering the above analysis. Meanwhile, the ranking of genes based on the CV values of transcriptomic data was similar to the validation results. This study provides for the first time a list of suitable reference genes for S. constricta and a valuable resource for further studies of clam immune defense systems.






Similar content being viewed by others
References
Andersen CL, Jensen JL, Ørntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250
Aychek T, Miller K, Sagi-Assif O, Levy-Nissenbaum O, Israeli-Amit M, Pasmanik-Chor M, Jacob-Hirsch J, Amariglio N, Rechavi G, Witz IP (2008) E-selectin regulates gene expression in metastatic colorectal carcinoma cells and enhances HMGB1 release. Int J Cancer 123:1741–1750
Banni M, Hajer A, Sforzini S, Oliveri C, Boussetta H, Viarengo A (2014) Transcriptional expression levels and biochemical markers of oxidative stress in Mytilus galloprovincialis exposed to nickel and heat stress. Comp Biochem Phy C 160:23–29
Barsalobres-Cavallari CF, Severino FE, Maluf MP, Maia IG (2009) Identification of suitable internal control genes for expression studies in Coffea arabica under different experimental conditions. BMC Mol Biol 10:1
Bathige SDNK., Umasuthan N, Kasthuri SR, Whang I, Lim BS, Nam BH, Lee J (2013) A bifunctional invertebrate-type lysozyme from the disk abalone, Haliotis discus discus: genome organization, transcriptional profiling and biological activities of recombinant protein. Dev Comp Immunol 41:282–294
Boujedidi H, Bouchet-Delbos L, Cassard-Doulcier AM, Njiké-Nakseu M, Maitre S, Prévot S, Dagher I, Agostini H, Voican CS, Emilie D (2012) Housekeeping gene variability in the liver of alcoholic patients. Alcohol Clin Exp Res 36:258–266
Browne GJ, Proud CG (2002) Regulation of peptide-chain elongation in mammalian cells. FEBS J 269:5360–5368
Bustin S, Benes V, Nolan T, Pfaffl M (2005) Quantitative real-time RT-PCR—a perspective. J Mol Endocrinol 34:597–601
Callewaert L, Michiels CW (2010) Lysozymes in the animal kingdom. J Biosci 35:127–160
Costa J, Passos M, Leitão C, Vasconcelos G, Saraiva M, Figueiredo J, Van den Hurk R, Silva J (2012) Levels of mRNA for bone morphogenetic proteins, their receptors and SMADs in goat ovarian follicles grown in vivo and in vitro. Reprod Fert Develop 24:723–732
Cubero-Leon E, Ciocan CM, Minier C, Rotchell JM (2012) Reference gene selection for qPCR in mussel, Mytilus edulis, during gametogenesis and exogenous estrogen exposure. Environ Sci Pollut R 19:2728–2733
Cui Y, Wei Z, Shen Y, Li C, Shao Y, Zhang W, Zhao X (2018) A novel C1q-domain-containing protein from razor clam Sinonovacula constricta mediates G-bacterial agglutination as a pattern recognition receptor. Dev Comp Immunol 79:166–174
Dhar AK, Bowers RM, Licon KS, Veazey G, Read B (2009) Validation of reference genes for quantitative measurement of immune gene expression in shrimp. Mol Immunol 46:1688–1695
Dheda K, Huggett JF, Bustin SA, Johnson MA, Rook G, Zumla A (2004) Validation of housekeeping genes for normalizing RNA expression in real-time PCR. Biotechniques 37:112–119
Du Y, Zhang L, Xu F, Huang B, Zhang G, Li L (2013) Validation of housekeeping genes as internal controls for studying gene expression during Pacific oyster (Crassostrea gigas) development by quantitative real-time PCR. Fish Shellfish Immunol 34:939–945
Fernandes JM, Mommens M, Hagen Ø, Babiak I, Solberg C (2008) Selection of suitable reference genes for real-time PCR studies of Atlantic halibut development. Comp Biochem Phy B 150:23–32
Filby AL, Tyler CR (2007) Appropriate’housekeeping’genes for use in expression profiling the effects of environmental estrogens in fish. BMC Mol Biol 8:10
Ginzinger DG (2002) Gene quantification using real-time quantitative PCR: an emerging technology hits the mainstream. Exp Hematol 30:503–512
Guénin S, Mauriat M, Pelloux J, Van Wuytswinkel O, Bellini C, Gutierrez L (2009) Normalization of qRT-PCR data: the necessity of adopting a systematic, experimental conditions-specific, validation of references. J Exp Bot 60:487–493
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8:R19
Hsiao L-L, Dangond F, Yoshida T, Hong R, Jensen RV, Misra J, Dillon W, Lee KF, Clark KE, Haverty P (2001) A compendium of gene expression in normal human tissues. Physiol Genom 7:97–104
Ingerslev H-C, Pettersen EF, Jakobsen RA, Petersen CB, Wergeland HI (2006) Expression profiling and validation of reference gene candidates in immune relevant tissues and cells from Atlantic salmon (Salmo salar L.). Mol Immunol 43:1194–1201
Koramutla MK, Aminedi R, Bhattacharya R (2016) Comprehensive evaluation of candidate reference genes for qRT-PCR studies of gene expression in mustard aphid, Lipaphis erysimi (Kalt). Sci Rep 6:25883
Kouadjo KE, Nishida Y, Cadrin-Girard JF, Yoshioka M, St-Amand J (2007) Housekeeping and tissue-specific genes in mouse tissues. BMC Genom 8:127
Kubista M, Andrade JM, Bengtsson M, Forootan A, Jonák J, Lind K, Sindelka R, Sjöback R, Sjögreen B, Strömbom L (2006) The real-time polymerase chain reaction. Mol Aspects Med 27:95–125
Lee AS, Kranzusch PJ, Cate JH (2015) eIF3 targets cell proliferation mRNAs for translational activation or repression. Nature 522:111
Li C, Li H, Su X, Li T (2011) Identification and characterization of a clam ferritin from Sinonovacula constricta. Fish Shellfish Immunol 30:1147–1151
Liu N, Pan L, Gong X, Tao Y, Hu Y, Miao J (2014a) Effects of benzo (a) pyrene on differentially expressed genes and haemocyte parameters of the clam Venerupis philippinarum. Ecotoxicology 23:122–132
Liu HH, Xiang LX, Shao JZ (2014b) A novel C1q-domain-containing (C1qDC) protein from Mytilus coruscus with the transcriptional analysis against marine pathogens and heavy metals. Dev Comp Immunol 44:70–75
Masutani M, Sonenberg N, Yokoyama S, Imataka H (2007) Reconstitution reveals the functional core of mammalian eIF3. EMBO J 26:3373–3383
Mateo DR, Greenwood SJ, Araya MT, Berthe FC, Johnson GR, Siah A (2010) Differential gene expression of γ-actin, Toll-like receptor 2 (TLR-2) and interleukin-1 receptor-associated kinase 4 (IRAK-4) in Mya arenaria haemocytes induced by in vivo infections with two Vibrio splendidus strains. Dev Comp Immunol 34:710–714
Meistertzheim AL, Tanguy A, Moraga D, Thébault MT (2007) Identification of differentially expressed genes of the Pacific oyster Crassostrea gigas exposed to prolonged thermal stress. FEBS J 274:6392–6402
Morga B, Arzul I, Faury N, Renault T (2010) Identification of genes from flat oyster Ostrea edulis as suitable housekeeping genes for quantitative real time PCR. Fish Shellfish Immunol 29:937–945
Niu D, Wang L, Bai Z, Xie S, Zhao H, Li J (2015) Identification and expression characterization of the myostatin (MSTN) gene and association analysis with growth traits in the razor clam Sinonovacula constricta. Gene 555:297–304
Niu D, Du Y, Wang Z, Xie S, Nguyen H, Dong Z, Shen H, Li J (2017) Construction of the First high-density genetic linkage map and analysis of quantitative trait loci for growth-related traits in Sinonovacula constricta. Mar Biotechnol 19:1–9
Peng C, Zhao X, Liu S, Shi W, Han Y, Guo C, Peng X, Chai X, Liu G (2017) Ocean acidification alters the burrowing behaviour, Ca2+/Mg2+-ATPase activity, metabolism, and gene expression of a bivalve species, Sinonovacula constricta. Mar Ecol Prog Ser 575:107–117
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Ran Z, Chen H, Ran Y, Yu S, Li S, Xu J, Liao K, Yu X, Zhong Y, Ye M (2017) Fatty acid and sterol changes in razor clam Sinonovacula constricta (Lamarck 1818) reared at different salinities. Aquaculture 473:493–500
Ren Q, Qi YL, Hui KM, Zhang Z, Zhang CY, Wang W (2012) Four invertebrate-type lysozyme genes from triangle-shell pearl mussel (Hyriopsis cumingii). Fish Shellfish Immunol 33:909–915
Robertson G, Schein J, Chiu R, Corbett R, Field M, Jackman SD, Mungall K, Lee S, Okada HM, Qian JQ (2010) De novo assembly and analysis of RNA-seq data. Nat Methods 7:909–912
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140
Selvey S, Thompson EW, Matthaei K, Lea RA, Irving M, Griffiths LR (2001) β-Actin—an unsuitable internal control for RT-PCR. Mol Cell Probes 15:307–311
Shi J, Liu M, Shi J, Zheng G, Wang Y, Wang J, Chen Y, Lu C, Yin W (2012) Reference gene selection for qPCR in Ammopiptanthus mongolicus under abiotic stresses and expression analysis of seven ROS-scavenging enzyme genes. Plant Cell Rep 31:1245–1254
Song H, Dang X, He Y-q, Zhang T, Wang H-y (2017) Selection of housekeeping genes as internal controls for quantitative RT-PCR analysis of the veined rapa whelk (Rapana venosa). PeerJ 5:e3398
Stürzenbaum SR, Kille P (2001) Control genes in quantitative molecular biological techniques: the variability of invariance. Comp Biochem Phy B 130:281–289
Thellin O, Zorzi W, Lakaye B, De Borman B, Coumans B, Hennen G, Grisar T, Igout A, Heinen E (1999) Housekeeping genes as internal standards: use and limits. J Biotechnol 75:291–295
Thompson FL, Iida T, Swings J (2004) Biodiversity of vibrios. Microbiol Mol Biol R 68:403–431
Thorrez L, Van Deun K, Tranchevent L-C, Van Lommel L, Engelen K, Marchal K, Moreau Y, Van Mechelen I, Schuit F (2008) Using ribosomal protein genes as reference: a tale of caution. PLoS One 3:e1854
Valenzuela-Castillo A, Mendoza-Cano F, Enríquez-Espinosa T, Grijalva-Chon JM, Sánchez-Paz A (2017) Selection and validation of candidate reference genes for quantitative real-time PCR studies in the shrimp Penaeus vannamei under viral infection. Mol Cell Probes 33:42–50
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:research0034.1
Volland M, Blasco J, Hampel M (2017) Validation of reference genes for RT-qPCR in marine bivalve ecotoxicology: systematic review and case study using copper treated primary Ruditapes philippinarum hemocytes. Aquatic Toxicol 185:86–94
Wang Z, Shao Y, Li C, Zhang W, Duan X, Zhao X, Qiu Q, Jin C (2016) RNA-seq analysis revealed ROS-mediated related genes involved in cadmium detoxification in the razor clam Sinonovacula constricta. Fish Shellfish Immunol 57:350–361
Zhang Y, He X, Yu Z (2011) Two homologues of inhibitor of NF-kappa B (IκB) are involved in the immune defense of the Pacific oyster, Crassostrea gigas. Fish Shellfish Immunol 30:1354–1361
Zhang Y, Chen D, Smith MA, Zhang B, Pan X (2012) Selection of reliable reference genes in Caenorhabditis elegans for analysis of nanotoxicity. PLoS One 7:e31849
Zhao X, Duan X, Wang Z, Zhang W, Li Y, Jin C, Xiong J, Li C (2017) Comparative transcriptome analysis of Sinonovacula constricta in gills and hepatopancreas in response to Vibrio parahaemolyticus infection. Fish Shellfish Immunol 67:523–535
Zheng W-j, Sun L (2011) Evaluation of housekeeping genes as references for quantitative real time RT-PCR analysis of gene expression in Japanese flounder (Paralichthys olivaceus). Fish Shellfish Immunol 30:638–645
Acknowledgements
This work was financially supported by Zhejiang Major Program of Science and Technology (2016C02055-9, 2015C32004), Natural Science Foundation of Ningbo (2015C10009, 2015C10062), Start Research Fund projects for Xuelin Zhao, Ningbo University Fund (XYL17010) and the K.C. Wong Magna Fund in Ningbo University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Xuelin Zhao declares that she does not have conflict of interest. Jianping Fu declares that he does not have conflict of interest. Liting Jiang declares that she does not have conflict of interest. Weiwei Zhang declares that she does not have conflict of interest. Yina Shao declares that hshe does not have conflict of interest. Chunhua Jin declares that he does not have conflict of interest. Jinbo Xiong declares that he does not have conflict of interest. Chenghua Li declares that he does not have conflict of interest.
Ethical approval
S. constricta is a commercially cultured animals, and all the experiments were conducted in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The study protocol was approved by the Experimental Animal Ethics Committee of Ningbo University, China.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure S1
Melting curves for the eight candidate reference genes. (TIF 2487 KB)
Supplementary Table S1
Details of orthologues of clam Sinonovacula constricta blasting with housekeeping genes in Crassostrea gigas. (XLSX 11 KB)
Supplementary Table S2
Expression values (CT) of candidate reference genes in vibrio-infected samples. (XLSX 10 KB)
Supplementary Table S3
Expression values (CT) of candidate reference genes in five tissues. (XLSX 10 KB)
Rights and permissions
About this article
Cite this article
Zhao, X., Fu, J., Jiang, L. et al. Transcriptome-based identification of the optimal reference genes as internal controls for quantitative RT-PCR in razor clam (Sinonovacula constricta). Genes Genom 40, 603–613 (2018). https://doi.org/10.1007/s13258-018-0661-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-018-0661-9