Abstract
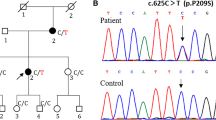
The kinesin heavy chain isoform 5A (KIF5A) gene, which encodes a microtubule-based motor protein, plays an important role in the transport of organelles in the nerve cells. Mutations in the KIF5A showed a wide phenotypic spectrum from hereditary spastic paraplegia (HSP) to axonal Charcot–Marie–Tooth peripheral neuropathy type 2 (CMT2). This study identified three pathogenic KIF5A mutations in Korean CMT2 patients by whole exome sequencing. Two mutations (p.Arg204Trp and p.Arg280His) were previously reported, but p.Leu558Pro was determined to be a novel de novo mutation. All the mutations were not observed in the healthy controls and were located in highly conserved domains among vertebrate species. The p.Arg204Trp mutation was identified from a CMT2 patient with additional complex phenotypes of HSP, ataxia, fatigability and pyramidal sign, but the p.Arg280His and p.Leu588Pro mutations were identified in each axonal CMT2 patient. The p.Arg204Trp mutation was previously reported in a HSP patient with no CMT symptom. The p.Arg280His mutation was reported in a CMT2 patient, which was similarly with our case. However, it was also once reported in a HSP patient with pes cavus. As the first report in Korea, this study identified three KIF5A mutations as the underlying cause of axonal peripheral neuropathy with or without the HSP phenotype. We confirmed a wide inter- and intra-allelic phenotypic spectrum by the mutations in the KIF5A.

Similar content being viewed by others
References
Antoniadi T, Buxton C, Dennis G, Forrester N, Smith D, Lunt P, Burton-Jones S (2015) Application of targeted multi-gene panel testing for the diagnosis of inherited peripheral neuropathy provides a high diagnostic yield with unexpected phenotype-genotype variability. BMC Med Genet 16:84
Blair MA, Ma S, Hedera P (2006) Mutation in KIF5A can also cause adult-onset hereditary spastic paraplegia. Neurogenetics 7:47–50
Carosi L, Lo Giudice T, Di Lullo M, Lombardi F, Babalini C, Gaudiello F, Marfia GA, Massa R, Kawarai T, Orlacchio A (2015) Hereditary spastic paraplegia: a novel mutation and expansion of the phenotype variability in SPG10. J Neurol Neurosurg Psychiatry 86:702–704
Choi B-O, Kim J, Lee KL, Yu JS, Hwang JH, Chung KW (2007) Rapid diagnosis of CMT1A duplications and HNPP deletions by multiplex microsatellite PCR. Mol Cells 23:39–48
Choi B-O, Koo SK, Park MH, Rhee H, Yang SJ, Choi KG, Jung SC, Kim HS, Hyun YS, Nakhro K, Lee HJ, Woo HM et al (2012) Exome sequencing is an efficient tool for genetic screening of Charcot-Marie-Tooth disease. Hum Mutat 33:1610–1615
Choi B-O, Nakhro K, Park HJ, Hyun YS, Lee JH, Kanwal S, Jung S-C, Chung KW (2015) A cohort study of MFN2 mutations and phenotypic spectrums in Charcot–Marie–Tooth disease 2A patients. Clin Genet 87:594–598
Crimella C, Baschirotto C, Arnoldi A, Tonelli A, Tenderini E, Airoldi G, Martinuzzi A, Trabacca A, Losito L, Scarlato M et al (2012) Mutations in the motor and stalk domains of KIF5A in spastic paraplegia type 10 and in axonal Charcot-Marie-Tooth type 2. Clin Genet 82:157–164
Duis J, Dean S, Applegate C, Harper A, Xiao R, He W, Dollar JD, Sun LR, Waberski MB, Crawford TO et al (2016) KIF5A mutations cause an infantile onset phenotype including severe myoclonus with evidence of mitochondrial dysfunction. Ann Neurol 80:633–637
Fichera M, Lo Giudice M, Falco M, Sturnio M, Amata S, Calabrese O, Bigoni S, Calzolari E, Neri M (2004) Evidence of kinesin heavy chain (KIF5A) involvement in pure hereditary spastic paraplegia. Neurology 63:1108–1110
Fink JK (2006) Hereditary spastic paraplegia. Curr Neurol Neurosci Rep 6:65–76
Fridman V, Murphy SM (2014) The spectrum of axonopathies: from CMT2 to HSP. Neurology 83:580–581
Fridman V, Bundy B, Reilly MM, Pareyson D, Bacon C, Burns J, Day J, Feely S, Finkel RS, Grider T et al (2015) CMT subtypes and disease burden in patients enrolled in the Inherited Neuropathies Consortium natural history study: a cross-sectional analysis. J Neurol Neurosurg Psychiatry 86:873–878
Goizet C, Boukhris A, Mundwiller E, Tallaksen C, Forlani S, Toutain A, Carriere N, Paquis V, Depienne C, Durr A et al (2008) Complicated forms of autosomal dominant hereditary spastic paraplegia are frequent in SPG10. Hum Mutat 30:e376
Harding AE (1983) Classification of the hereditary ataxias and paraplegias. Lancet 1:1151–1155
Hyun YS, Hong YB, Choi BO, Chung KW (2016) Clinico-genetics in Korean Charcot-Marie-Tooth disease type 2Z with MORC2 mutations. Brain 139:e40
Iqbal Z, Rydning SL, Wedding IM, Koht J, Pihlstrøm L, Rengmark AH, Henriksen SP, Tallaksen CM, Toft M (2017) Targeted high throughput sequencing in hereditary ataxia and spastic paraplegia. PLoS ONE 12:e0174667
Kaji S, Kawarai T, Miyamoto R, Nodera H, Pedace L, Orlacchio A, Izumi Y, Takahashi R, Kaji R (2016) Late-onset spastic paraplegia type 10 (SPG10) family presenting with bulbar symptoms and fasciculations mimicking amyotrophic lateral sclerosis. J Neurol Sci 364:45–49
Liu Y-T, Laura M, Hersheson J, Horga A, Jaunmuktane Z, Brandner S, Pittman A, Hughes D, Polke JM, Sweeney MG et al (2014) Extended phenotypic spectrum of KIF5A mutations: from spastic paraplegia to axonal neuropathy. Neurology 83:612–619
López E, Casasnovas C, Giménez J, Santamaría R, Terrazas JM, Volpini V (2015) Identification of two novel KIF5A mutations in hereditary spastic paraplegia associated with mild peripheral neuropathy. J Neurol Sci 358:422–427
Nam SH, Hong YB, Hyun YS, Nam DE, Kwak G, Hwang SH, Choi BO, Chung KW (2016) Identification of genetic causes of inherited peripheral neuropathies by targeted gene panel sequencing. Mol Cells 39:382–388
Niclas J, Navone F, Hom-Booher N, Vale RD (1994) Cloning and localization of a conventional kinesin motor expressed exclusively in neurons. Neuron 12:1059–1072
Noreau A, Dion PA, Rouleau GA (2014) Molecular aspects of hereditary spastic paraplegia. Exp Cell Res 325:18–26
Reid E, Kloos M, Ashley-Koch A, Hughes L, Bevan S, Svenson IK, Graham FL, Gaskell PC, Dearlove A, Pericak-Vance MA et al (2002) A kinesin heavy chain (KIF5A) mutation in hereditary spastic paraplegia (SPG10). Am J Hum Genet 71:1189–1194
Rydzanicz M, Jagła M, Kosinska J, Tomasik T, Sobczak A, Pollak A, Herman-Sucharska I, Walczak A, Kwinta P, Płoski R (2017) KIF5A de novo mutation associated with myoclonic seizures and neonatal onset progressive leukoencephalopathy. Clin Genet 91:769–773
Xia C, Rahman A, Yang Z, Goldstein LS (1998) Chromosomal localization reveals three kinesin heavy chain genes in mouse. Genomics 52:209–213
Yoo DH, Choi YC, Nam DE, Choi SS, Kim JW, Choi BO, Chung KW (2017) Identification of FASTKD2 compound heterozygous mutations as the underlying cause of autosomal recessive MELAS-like syndrome. Mitochondrion 35:54–58
Acknowledgements
We would like to thank the patients and their families for their participation in this study. This study was supported by the Kongju National University (2014).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Nam DE, Yoo DH, Choi SS, Choi B-O, and Chung KW declare that they have no conflict of interest.
Informed consent
Written informed consent was obtained from all the participants or the parents of those younger than 18 years of age according to the protocol approved by the Institutional Review Board of Samsung Medical Center at Sungkyunkwan University (SMC, 2014-08-057-002).
Rights and permissions
About this article
Cite this article
Nam, D.E., Yoo, D.H., Choi, S.S. et al. Wide phenotypic spectrum in axonal Charcot–Marie–Tooth neuropathy type 2 patients with KIF5A mutations. Genes Genom 40, 77–84 (2018). https://doi.org/10.1007/s13258-017-0612-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-017-0612-x