Abstract
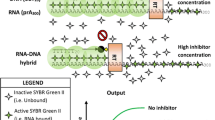
Current in vitro assays for the activity of HIV-RT (reverse transcriptase) require radio-labeled or chemically modified nucleotides to detect reaction products. However, these assays are inherently end-point measurements and labor intensive. Here we describe a novel non-radioactive assay based on the principle of pyrosequencing coupled-enzyme system to monitor the activity of HIV-RT by indirectly measuring the release of pyrophosphate (PPi), which is generated during nascent strand synthesis. The results show that our assay could monitor HIV-RT activity with high sensitivity and is suitable for rapid high-throughput drug screening targeting anti-HIV therapies due to its high speed and convenience. Moreover, this assay can be used to measure primase activity in an easy and sensitive manner, which suggests that this novel approach could be wildly used to analyze the activity of PPi-generated and ATP-free enzyme reactions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andre, M., Morgeaux, S., and Fuchs, F. (2000). Quantitative detection of RTactivity by PERTassay: feasibility and limits to a standardized screening assay for human vaccines. Biologicals 28, 67–80.
Antoun, M.D., Rios, Y.R., Mendoza, N.T., and Proctor, G. (1994). Reverse transcriptase inhibition as prescreen for potential antiviral bioactives. P R Health Sci J 13, 17–18.
Arnold, B.A., Hepler, R.W., and Keller, P.M. (1998). One-step fluorescent probe product-enhanced reverse transcriptase assay. Biotechniques 25, 98–106.
Chang, A., Ostrove, J.M., and Bird, R.E. (1997). Development of an improved product enhanced reverse transcriptase assay. J Virol Methods 65, 45–54.
Clark, A.D., Jr., Jacobo-Molina, A., Clark, P., Hughes, S.H., and Arnold, E. (1995). Crystallization of human immunodeficiency virus type 1 reverse transcriptase with and without nucleic acid substrates, inhibitors, and an antibody Fab fragment. Methods Enzymol 262, 171–185.
Hizi, A., Tal, R., Shaharabany, M., Currens, M.J., Boyd, M.R., Hughes, S.H., and McMahon, J.B. (1993). Specific inhibition of the reverse transcriptase of human immunodeficiency virus type 1 and the chimeric enzymes of human immunodeficiency virus type 1 and type 2 by nonnucleoside inhibitors. Antimicrob Agents Chemother 37, 1037–1042.
Jacobo-Molina, A., Ding, J., Nanni, R.G., Clark, A.D., Jr., Lu, X., Tantillo, C., Williams, R.L., Kamer, G., Ferris, A.L., Clark, P., et al. (1993). Crystal structure of human immunodeficiency virus type 1 reverse transcriptase complexed with double-stranded DNA at 3.0 A resolution shows bent DNA. Proc Natl Acad Sci U S A 90, 6320–6324.
Kanyara, J.N., and Njagi, E.N. (2005). Anti-HIV-1 activities in extracts from some medicinal plants as assessed in an in vitro biochemical HIV-1 reverse transcriptase assay. Phytother Res 19, 287–290.
Kohlstaedt, L.A., Wang, J., Friedman, J.M., Rice, P.A., and Steitz, T.A. (1992). Crystal structure at 3.5 A resolution of HIV-1 reverse transcriptase complexed with an inhibitor. Science 256, 1783–1790.
Lahser, F.C., and Malcolm, B.A. (2004). A continuous nonradioactive assay for RNA-dependent RNA polymerase activity. Anal Biochem 325, 247–254.
Lovatt, A., Black, J., Galbraith, D., Doherty, I., Moran, M.W., Shepherd, A.J., Griffen, A., Bailey, A., Wilson, N., and Smith, K.T. (1999). High throughput detection of retrovirus-associated reverse transcriptase using an improved fluorescent product enhanced reverse transcriptase assay and its comparison to conventional detection methods. J Virol Methods 82, 185–200.
Odawara, F., Abe, H., Kohno, T., Nagai-Fujii, Y., Arai, K., Imamura, S., Misaki, H., Azuma, H., Ikebuchi, K., Ikeda, H., et al. (2002). A highly sensitive chemiluminescent reverse transcriptase assay for human immunodeficiency virus. J Virol Methods 106, 115–124.
Porstmann, T., Meissner, K., Glaser, R., Dopel, S.H., and Sydow, G. (1991). A sensitive non-isotopic assay specific for HIV-1 associated reverse transcriptase. J Virol Methods 31, 181–188.
Pyra, H., Boni, J., and Schupbach, J. (1994). Ultrasensitive retrovirus detection by a reverse transcriptase assay based on product enhancement. Proc Natl Acad Sci U S A 91, 1544–1548.
Ronaghi, M., Uhlen, M., and Nyren, P. (1998). A sequencing method based on real-time pyrophosphate. Science 281, 363–365.
Sarafianos, S.G., Das, K., Hughes, S.H., and Arnold, E. (2004). Taking aim at a moving target: designing drugs to inhibit drug-resistant HIV-1 reverse transcriptases. Curr Opin Struct Biol 14, 716–730.
Silver, J., Maudru, T., Fujita, K., and Repaske, R. (1993). An RT-PCR assay for the enzyme activity of reverse transcriptase capable of detecting single virions. Nucleic Acids Res 21, 3593–3594.
Suzuki, K., Craddock, B.P., Kano, T., and Steigbigel, R.T. (1993a). Colorimetric reverse transcriptase assay for HIV-1. J Virol Methods 41, 21–28.
Suzuki, K., Craddock, B.P., Okamoto, N., Kano, T., and Steigbigel, R. T. (1993b). Detection of human immunodeficiency virus (HIV) by colorimetric assay for reverse transcriptase activity on magnetic beads. Biotechnol Appl Biochem 18 (Pt 1), 37–44.
Suzuki, K., Saito, T., Kondo, M., Osanai, M., Watanabe, S., Kano, T., Kano, K., and Imai, M. (1995). Poly A-linked non-isotopic microtiter plate reverse transcriptase assay for sensitive detection of clinical human immunodeficiency virus isolates. J Virol Methods 55, 347–356.
Urabe, T., Sano, K., Tanno, M., Mizoguchi, J., Otani, M., Lee, M.H., Takasaki, T., Kusakabe, H., Imagawa, D.T., and Nakai, M. (1992). A non-radioisotopic reverse transcriptase assay using biotin-11-deoxyuridinetriphosphate on primer-immobilized microtiter plates. J Virol Methods 40, 145–154.
Vassiliou, W., Epp, J.B., Wang, B.B., Del Vecchio, A.M., Widlanski, T., and Kao, C.C. (2000). Exploiting polymerase promiscuity: A simple colorimetric RNA polymerase assay. Virology 274, 429–437.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, C., Wu, Y., Sun, Y. et al. A novel non-radioactive assay for HIV-RT (RdDp) based on pyrosequencing for high-throughput drug screening. Protein Cell 1, 284–290 (2010). https://doi.org/10.1007/s13238-010-0031-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13238-010-0031-0