Abstract
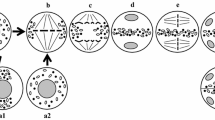
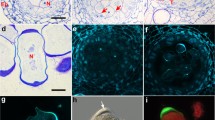
True endomitosis in the anther tapetum of the three Cymbidium species viz. C. aloifolium, C. devonianum and C. tigrinum is described. In these Cymbidiums, most tapetal cells go through endomitosis instead of either normal mitosis or so called “inhibited” mitosis. The nuclear membrane does not disappear, but during metaphase the chromosomes are considerably condensed far more than in normal mitosis, and nucleolus persists throughout the endomitotic cycle. Endomitosis may not be unusual to the tapetal cells of these peculiar Cymbidiums but may have a wider application and explain many of the cytological phenomena occurring in the tapetal cells of other plants. Further, physical localization of 45S rRNA genes during endomitotic divisions may help to confirm their atypical activities as well as provide insight into the course and degree of tapetum polyploidization. Analysis of DNA-methylation levels is also recommended for understanding the role of nucleolus for spindle formation during endomitosis.

Similar content being viewed by others
References
Aragon-Alcaide L, Reader S, Bevan A, Shaw P, Miller T, Moore G. Association of homologous chromosomes during floral development. Curr Biol. 1997;7:905–8.
Bannigan A, Lizotte-waniewski M, Riley M, Baskin TI. Emerging molecular mechanisms that power and regulate the anastral mitotic spindle of flowering plants. Cell Motil Cytoskeleton. 2008;65:1–11.
Berger CA, Witkus ER, Joseph TC. Tapetal cell and meiotic divisions in Antirrhinum majus L. and Linaria vulgaris Hill. Caryologia. 1951;4:110–4.
Brown SW. The structure and meiotic behavior of the differentiated chromosomes of tomato. Genetics. 1949;34:437–61.
D'amato F. Polyploidy in the differentiation and function of tissues and cells in plants. A critical examination of the literature. Caryologia. 1952;4:311–58.
D'amato F. A brief discussion of "endomitosis". Caryologia. 1954;6:341–4.
D'amato E. Role of polyploidy in reproductive organs and tissues. In: Johri BM, editor. Embryology of angiosperms. Berlin, Germany: Springer- Verlag; 1984. p. 519–66.
Geitler L. Die Entstehung der polyploiden Somakerne der Heteropteren durch Chromosomen-teilung ohne Kernteilung. Chromosoma. 1939;1:1–22.
Geitler L. Endomitose und endomitotische Poly-ploidisierung. Protoplasmatologia. 1953;6C:1–89.
Heitz E. Uber intraindividuale Polyploidie. Arch Julius Klaus-Stift. 1953;28:260–71.
Hussain S, Benavente SB, Nascimento E, Dragoni I, Kurowski A, Gillich A, Humphreys P, Frye M. The nucleolar RNA methyltransferase Misu (NSun2) is required for mitotic spindle stability. J Cell Biol. 2009;186:27–40.
Levan A, Hauschka TS. Endomitotic reduplication mechanisms in ascites tumors of the mouse. J Natl Canc Inst. 1953;14:1–43.
Mechelke F. Die Entstehung der polyploiden Zellkerne des Antherentapetums bei Antirrhinum majus L. Chromosoma. 1952;5:246–95.
Oksala T. Timing relationships in mitosis and meiosis. Caryologia Suppl. 1954;6:272–81.
Oksala T. The mitotic mechanism of two mouse ascites tumours. Hereditas. 1956;42:161–88.
Oksala T, Therman E. Endomitosis in Tapetal Cells of Eremurus (Liliaceae). Am J Bot. 1977; 64:866–72.
Sharma SK, Rajkumari K, Kumaria S, Tandon P, Rao SR. Karyo-morphological characterization of natural genetic variation in some threatened Cymbidium species of Northeast India. Caryologia. 2010;63:99–105.
Sharma SK, Kumaria S, Tandon P, Rao SR. Single primer amplification reaction (SPAR) reveals inter- and intra-specific natural genetic variation in five species of Cymbidium (Orchidaceae). Gene. 2011;483:54–62.
Sharma SK, Mehra P, Kumari J, Kumar S, Kumaria S, Tandon P, Rao SR. Physical localization and probable transcriptional activity of 18 S-5.8 S-26S rRNA gene loci in some Asiatic Cymbidiums (Orchidaceae) from north-east India. Gene. 2012; doi:10.1016/j.gene.2012.03.007 (in press)
Sharma SK, Dkhar J, Kumaria S, Tandon P, Rao SR. Assessment of phylogenetic inter-relationships in the genus Cymbidium (Orchidaceae) based on internal transcribed spacer region of rDNA. Gene. 2012;495:10–5.
Somashekarappa HM, Narayana Y, Radhakrishna AP, Karunakara N, Balakrishna KM, Siddappa K. Bioindicators in the tropical forest of Kaiga environment. J Environ Radioact. 1996;31:189–98.
Timonen S, Therman E. Endomitotic nuclear growth in a human cervical carcinoma. Ann Chir Gynaecol Fenn. 1956;45:237–44.
TJio JH, Levan A. Chromosome analysis of three hyperdiploid ascites tumours of the mouse. K Fysiogr Sallsk Handl (Lund) NF. 1954;65:1–38.
Tschermak-Woess E. Endomitose. Handb allgem Pathol. 1971;2:569–625.
Witkus ER. Endomitotic tapetal cell divisions in Spinacia. Am J Bot. 1945;32:326–30.
Weiss H, Maluszynska J. Molecular Cytogenetic Analysis of polyploidization in the anther tapetum of diploid and autotetraploid Arabidopsis thaliana plants. Ann Bot. 2001;87:729–35.
Acknowledgements
The present work is supported by a grant from University Grants Commission, Government of India, New Delhi through University with Potential for Excellence (UPE) – Bioscience program. We thank the Head of the Department of Biotechnology and Bioinformatics, NEHU, Shillong for providing facilities. Sincere thanks are due to all members of Plant Biotechnology Laboratories of Department of Botany and Department of Biotechnology and Bioinformatics, NEHU, Shillong for their constant encouragement and help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, S.K., Kumaria, S., Tandon, P. et al. Endomitosis in tapetal cells of some Cymbidiums (Orchidaceae). Nucleus 55, 21–25 (2012). https://doi.org/10.1007/s13237-012-0049-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13237-012-0049-1