Abstract
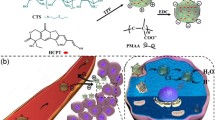
A new platform of paclitaxel (PTX) for application as an oral delivery system was developed, by combining the pH sensitivity of polyelectrolyte complex nanoparticles (CNPs) and the active targeting of hyaluronic acid (HA). Chitosan/hyaluronic acid-paclitaxel (CS/HA-PTX) CNPs were prepared by coating the CS onto the HA-PTX nanoparticles (NPs), and characterized by Fourier-transform infrared spectroscopy (FTIR), nuclear magnetic resonance (1H NMR), transmission electron microscopy (TEM) and high-performance liquid chromatography (HPLC). HA-PTX conjugates could self-assemble into NPs in aqueous solution with an average size of 100±5 nm, and the PTX content of HA-PTX conjugates was 10.6 wt%. The CS/HA-PTX CNPs had a smaller size and higher PTX content when the ratio of positive charge to negative charge was 2:1. The in vitro release of PTX from CNPs was pH-responsive, suggesting that the CS shell could prevent the breakage of the ester bond in HA-PTX NPs in acidic pH conditions. HA-PTX NPs exhibited higher cellular uptake than free PTX against HepG2 cells via receptor-mediated endocytosis. PTX could accumulate remarkably into tumor sites after oral administration of CNPs. These results indicate that the CNP drug delivery system has great potential for applications in the oral administration of hydrophobic drugs.

Similar content being viewed by others
References
L. Bromberg, J. Control. Release, 128, 99 (2008).
Y.-H. Lin, H.-F. Liang, C.-K. Chung, M.-C. Chen, and H.-W. Sung, Biomaterials, 26, 2105 (2005).
Y.-H. Lin, F.-L. Mi, C.-T. Chen, W.-C. Chang, S.-F. Peng, H.-F. Liang, and H.-W. Sung, Biomacromolecules, 8, 146 (2007).
G. Gaucher, P. Satturwar, M.-C. Jones, A. Furtos, and J.-C. Leroux, Eur. J. Pharm. Biopharm., 76, 147 (2010).
L. M. Ensign, R. Cone, and J. Hanes, Adv. Drug Deliv. Rev., 64, 557 (2012).
F. Leonelli, A. La Bella, L. Migneco, and R. Bettolo, Molecules, 13, 360 (2008).
M. M. Malingre, J. H. Beijnen, and J. H. M. Schellens, Invest. New Drugs, 19, 155 (2001).
P. Breedveld, J. H. Beijnen, and J. H. M. Schellens, Trends Pharmacol. Sci., 27, 17 (2006).
C. Fonseca, S. Simoes, and R. Gaspar, J. Control. Release, 83, 273 (2002).
L. Vicari, T. Musumeci, I. Giannone, L. Adamo, C. Conticello, R. De Maria, R. Pignatello, G. Puglisi, and M. Gulisano, BMC Cancer, 8 (2008).
G. Bachar, K. Cohen, R. Hod, R. Feinmesser, A. Mizrachi, T. Shpitzer, O. Katz, and D. Peer, Biomaterials, 32, 4840 (2011).
I. Rivkin, K. Cohen, J. Koffler, D. Melikhov, D. Peer, and R. Margalit, Biomaterials, 31, 7106 (2010).
H. Lee, C.-H. Ahn, and T.G. Park, Macromol. Biosci., 9, 336 (2009).
D. Yang, X. Liu, X. Jiang, Y. Liu, W. Ying, H. Wang, H. Bai, W. D. Taylor, Y. Wang, J.-P. Clamme, E. Co, P. Chivukula, K. Y. Tsang, Y. Jin, and L. Yu, J. Control. Release, 161, 124 (2012).
Y. J. Jun, J. H. Min, D. E. Ji, J. H. Yoo, J. H. Kim, H. J. Lee, B. Jeong, and Y. S. Sohn, Bioorg. Med. Chem. Lett., 18, 6410 (2008).
D. Kalaria, G. Sharma, V. Beniwal, and M. R. Kumar, Pharm. Res., 26, 492 (2009).
E. Lee, J. Lee, I.-H. Lee, M. Yu, H. Kim, S. Y. Chae, and S. Jon, J. Med. Chem., 51, 6442 (2008).
L. Milas, K. A. Mason, N. Hunter, C. Li, and S. Wallace, Int. J. Radiat. Oncol., 55, 707 (2003).
E. Lee, H. Kim, I.-H. Lee, and S. Jon, J. Control. Release, 140, 79 (2009).
X. Zhang, Y. Li, X. Chen, X. Wang, X. Xu, Q. Liang, J. Hu, and X. Jing, Biomaterials, 26, 2121 (2005).
C. Li, R. A. Newman, Q. P. Wu, S. Ke, W. Chen, T. Hutto, Z. X. Kan, M. D. Brannan, C. Charnsangavej, and S. Wallace, Cancer Chemother. Pharmacol., 46, 416 (2000).
A. K. Jain, N. K. Swarnakar, C. Godugu, R. P. Singh, and S. Jain, Biomaterials, 32, 503 (2011).
C. E. Schante, G. Zuber, C. Herlin, and T. F. Vandamme, Carbohydr. Polym., 85, 469 (2011).
I. De Stefano, A. Battaglia, G. F. Zannoni, M. G. Prisco, A. Fattorossi, D. Travaglia, S. Baroni, D. Renier, G. Scambia, C. Ferlini, and D. Gallo, Cancer Chemother. Pharmacol., 68, 107 (2011).
S. Ogino, N. Nishida, R. Umemoto, M. Suzuki, M. Takeda, H. Terasawa, J. Kitayama, M. Matsumoto, H. Hayasaka, M. Miyasaka, and I. Shimada, Structure, 18, 649 (2010).
S. Manju and K. Sreenivasan, J. Colloid Interface Sci., 359, 318 (2011).
J. Klostergaard, E. Auzenne, S. C. Ghosh, M. Khodadadian, B. Rivera, D. Farquhar, R. E. Price, M. Ravoori, V. Kundra, and R. S. Freedman, Mol. Cancer Ther., 6, 3619S (2007).
H.-J. Cho, I.-S. Yoon, H. Y. Yoon, H. Koo, Y.-J. Jin, S.-H. Ko, J.-S. Shim, K. Kim, I. C. Kwon, and D.-D. Kim, Biomaterials, 33, 1190 (2012).
T. Jiang, Z. Zhang, Y. Zhang, H. Lv, J. Zhou, C. Li, L. Hou, and Q. Zhang, Biomaterials, 33, 9246 (2012).
R. Paliwal, S. R. Paliwal, G. P. Agrawal, and S. P. Vyas, Int. J. Pharm., 422, 179 (2012).
W. Ke, Y. Zhao, R. Huang, C. Jiang, and Y. Pei, J. Pharm. Sci., 97, 2208 (2008).
J. H. Hamman, Mar. Drugs, 8, 1305 (2010).
B. Sarmento, A. Ribeiro, F. Veiga, P. Sampaio, R. Neufeld, and D. Ferreira, Pharm. Res., 24, 2198 (2007).
H. Yoon and G. Kim, Macromol. Res., 20, 402 (2012).
J. M. Smith, M. Dornish, and E. J. Wood, Biomaterials, 26, 3269 (2005).
D. F. Evans, G. Pye, R. Bramley, A. G. Clark, T. J. Dyson, and J. D. Hardcastle, Gut, 29, 1035 (1988).
C. Thouzeau, G. Peters, C. Le Bohec, and Y. Le Maho, J. Expe. Biol., 207, 2715 (2004).
C. T. Tsao, C. H. Chang, Y. Y. Lin, M. F. Wu, J.-L. Wang, J. L. Han, and K. H. Hsieh, Carbohydr. Res., 345, 1774 (2010).
C. Chun, S. M. Lee, S. Y. Kim, H. K. Yang, and S.-C. Song, Biomaterials, 30, 2349 (2009).
X. Feng, Y.-J. Yuan, and J.-C. Wu, Bioorg. Med. Chem. Lett., 12, 3301 (2002).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Li, J., Huang, P., Chang, L. et al. Tumor targeting and pH-responsive polyelectrolyte complex nanoparticles based on hyaluronic acid-paclitaxel conjugates and Chitosan for oral delivery of paclitaxel. Macromol. Res. 21, 1331–1337 (2013). https://doi.org/10.1007/s13233-013-1171-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-013-1171-x