Abstract
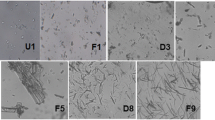
Drug stability and sustained release issues are important areas in drug-delivery research. The sustained release of proteins can be achieved by their encapsulation with a hydrophobic polymer, but this requires the proteins to be protected from the harsh processing environments of organic solvents and mechanical force. Preencapsulation with poly(vinyl alcohol) (PVA) using a freeze/thaw method has been shown to allow successful protection and sustained release. This study examined the effects of freezing/thawing on PVA encapsulation in the preparation of PVA-PLGA composite particles. Freezing/thawing slightly improved the crystalline peaks and the heat of fusion of PVA, but more distinct differences were observed when the properties of the surface layers were probed by AFM. The hardness of particles’ surfaces increased with increasing number of freezing/thawing cycles, whereas the adhesion force with an AFM cantilever tip decreased. The mean particle size and entrapment efficiency decreased. These results suggest that surface hardening is the major mechanism responsible for the sustained release characteristics.
Similar content being viewed by others
References
V. Sinha and A. Trehan, J. Control. Release, 90, 261 (2003).
J. Cleland, Protein Delivery, 10, 1 (2002).
J. Herrmann and R. Bodmeier, J. Control. Release, 36, 63 (1995).
H. Zhao, K. Saatchi, and U. Hafeli, J. Magn. Magn. Mater., 321, 1356 (2009).
O. Farokhzad, Expert Opinion on Drug Delivery, 5, 927 (2008).
S. Putney and P. Burke, Nature Biotechnology, 16, 153 (1998).
J. Li, N. Wang, and X. Wu, J. Control. Release, 56, 117 (1998).
N. Wang, X. Wu, and J. Li, Pharm. Res., 16, 1430 (1999).
K. Hong and G. Sun, J. Appl. Polym. Sci., 116, 2418 (2010).
N. Vrana, et al., J. Tissue Eng. Regen. M., 3, 567 (2009).
M. M. Simöes and M. G. de Oliveira, J. Biomed. Mater. Res. Part B: Appl. Biomater., 93B, 416 (2010).
A. Szücs, T. Haraszti, I. Dékány, and J. H. Fendler, J. Phys. Chem. B, 105, 10579 (2001).
E. R. Beach, G. W. Tormoen, J. Drelich, and R. Han, J. Colloid Interface Sci., 247, 84 (2002).
M. Tortonese and M. Kirk, Proceeding of International Society for Optics and Photonics, 1997, vol. 3009, p53–60.
U. Sindel and I. Zimmermann, Powder Technol., 117, 247 (2001).
M. Fuji, K. Machida, T. Takei, T. Watanabe, and M. Chikazawa, Langmuir, 15, 4584 (1999).
X. Xiao and L. Qian, Langmuir, 16, 8153 (2000).
V. Bérard, E. Lesniewska, C. Andrés, D. Pertuy, C. Laroche, and Y. Pourcelot, Int. J. Pharm., 232, 213 (2002).
W. Ducker, T. Senden, and R. Pashley, Nature, 353, 239 (1991).
I. Larson, C. J. Drummond, D. Y. C. Chan, and F. Grieser, J. Am. Chem. Soc., 115, 11885 (1993).
J. Villarrubia, Surf. Sci., 321, 287 (1994).
M. Louey, P. Mulvaney, and P. Stewart, J. Pharmaceut. Biomed. Anal., 25, 559 (2001).
J. K. Eve, N. Patel, S. Y. Luk, S. J. Ebbens, and C. J. Roberts, Int. J. Pharm., 238, 17 (2002).
J. E. Sader, I. Larson, P. Mulvaney, and L. R. White, Rev. Sci. Instrum., 66, 3789 (1995).
J. P. Cleveland, S. Manne, D. Bocek, and P. K. Hansma, Rev. Sci. Instrum., 64, 403 (1993).
C. Hassan, J. Stewart, and N. Peppas, Eur. J. Pharm. Biopharm., 49, 161 (2000).
C. Hassan and N. Peppas, Macromolecules, 33, 2472 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, M., Bae, H., Lee, S. et al. Freezing/thawing processing of PVA in the preparation of structured microspheres for protein drug delivery. Macromol. Res. 19, 130–136 (2011). https://doi.org/10.1007/s13233-011-0203-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-011-0203-7