Abstract
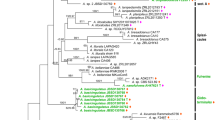
The recognition of taxonomic ranks in the Linnean classification system is largely arbitrary. Some authors have proposed the use of divergence time as a universally standardized criterion. Agaricus (Agaricaceae, Agaricales) is a mushroom genus that contains many species of high commercial value. Recent studies using ITS sequence data discovered 11 new phylogenetic lineages within the genus, however their taxonomic ranks were uncertain due to the lack of criteria to define them within traditional taxonomy. In this study, we analyzed ITS sequence data from 745 collections (nearly 600 being newly generated) including 86 from type specimens of previously recognized subgenera and sections. Many monophyletic groups were recognized, but most basal relationships were unresolved. One hundred and fourteen representatives of the identified ITS clades were selected in order to produce a multi-gene phylogeny based on combined LSU, tef-1α, and rpb2 sequence data. Divergence times within the multi-gene phylogeny were estimated using BEAST v1.8. Based on phylogenetic relationships and with respect to morphology, we propose a revised taxonomic system for Agaricus that considers divergence time as a standardized criterion for establishing taxonomic ranks. We propose to segregate Agaricus into five subgenera and 20 sections. Subgenus Pseudochitonia is substantially emended; circumscription of the subgenera Agaricus and Flavoagaricus is restricted to taxa of sections Agaricus and Arvenses, respectively; and two new subgenera (Minores and Spissicaules) are introduced. Within Pseudochitonia, sections Bivelares, Brunneopicti, Chitonioides, Nigrobrunnescentes, Sanguinolenti and Xanthodermatei are maintained, but the latter two are reduced because we raise subsection Bohusia to sectional rank and a clade within section Xanthodermatei is formally introduced as section Hondenses; and sections Rubricosi, Crassispori, Flocculenti, and Amoeni are introduced. Section Laeticolores is placed in the subgenus Minores and sections Rarolentes and Subrutilescentes are placed in the subgenus Spissicaules. Twenty-two new species belonging to various sections are described. This work exemplifies that ITS data, while useful at lower taxonomic levels (i.e., detection of species and species groups), are of limited value for inferring deeper phylogenetic relationships. Finally, we suggest that the establishment of a standardized taxonomic system based on divergence times could result in a more objective, and biologically more meaningful, taxonomic ranking of fungi.


































Similar content being viewed by others
References
Ariyawansa HA, Tanaka K, Thambugala KM, Phookamsak R, Tian Q, Camporesi E, Hongsanan S, Monkai J, Wanasinghe DN, Chukeatirote E, Kang JC, Xu JC, McKenzie EHC, Jones EBG, Hyde KD (2014) A molecular phylogenetic reappraisal of the Didymosphaeriaceae (= Montagnulaceae). Fungal Divers 68:69–104
Avise JC, John GC (1999) Proposal for a standardized temporal scheme of biological classification for extant species. Proc Natl Acad Sci U S A 96:7358–7363
Berbee ML, Taylor JW (2010) Dating the molecular clock in fungi – how close are we? Fungal Biol Rev 24:1–16
Bohus G (1995) Agaricus studies XIII. Key to the subgenus Agaricus from Europe. Mikol Közlem 34(1):5–35
Budd G, Jensen S (2000) A critical reappraisal of the fossil of the bilaterian phyla. Biol Rev 75:253–295
Budd G (2001) Climbing life’s tree. Nature 412:487
Callac P, Guinberteau J (2005) Morphological and molecular characterization of two novel species of Agaricus section Xanthodermatei. Mycologia 97:416–424
Cappelli A (1984) Agaricus L.: Fr. (Psalliota Fr.). Libreria editrice Biella Giovanna, Saronno, p 558
Challen MP, Kerrigan RW, Callac P (2003) A phylogenetic reconstruction and emendation of Agaricus section Duploannulatae. Mycologia 95(1):61–73
Chen J, Zhao RL, Karunarathna S, Callac P, Raspé O, Bahkali AH, Hyde KD (2012) Agaricus megalosporus: a new species in section Minores. Cryptogamie Mycol 33:145–155
Chen J, Zhao RL, Parra LA, Guelly AK, Kesel AD, Rapior S, Hyde KD, Chukeatirote E, Callac P (2015) Agaricus section Brunneopicti: a phylogenetic reconstruction with descriptions of four new taxa. Phytotaxa 192:145–168
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772
Dentinger BM, Margaritescu S, Moncalvo J-M (2010) Rapid and reliable high-throughput methods of DNA extraction for use in barcoding and molecular systematics of mushrooms. Mol Ecol Resour 10:628–633
Didukh M, Vilgalys R, Wasser SP, Isikhuemhen OS, Nevo E (2005) Notes on Agaricus section Duploannulati using molecular and morphological data. Mycol Res 109:729–740
Drummond AJ, Ho SYW, Phillips MJ, Rambaut A (2006) Relaxed phylogenetics and dating with confidence. PLoS Biol 4(5), e88
Drummond AJ, Suchard MA, Xie D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and BEAST 1.7. Mol Biol 29:1969–1973
Edgar RC (2004a) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Edgar RC (2004b) MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinforma 5:113
Fungal Names (2013). http://fungalinfo.im.ac.cn/fungalname/fungalname.html
Geml J, Geiser DM, Royse DJ (2004) Molecular evolution of Agaricus species based on ITS and LSU rDNA sequences. Mycol Prog 3(2):157–176
Geml J, Laursen GA, Nusbaum HC, Taylor DL (2007) Two new species of Agaricus from the Subantarctic. Mycotaxon 100:193–208
Gui Y, Zhu GS, Callac P, Hyde KD, Parra LA, Chen J, Yang TJ, Huang WB, Gong GL, Liu ZY (2015) Agaricus section Arvenses: three new species in highland subtropical Southwest China. Fungal Biol 119(2–3):79–94
Heinemann P (1956a) Champignons récoltés au Congo Belge par Mme M Goossens-Fontana, II Agaricus Fr ss. Bull Jard Bot État 26:1–127
Heinemann P (1956b) Champignons récoltés au Congo Belge par Mme M Goossens-Fontana, II Agaricus, Note complémentaire. Bull Jard Bot État 26:325–333
Heinemann P (1961) Agarici Austroamericani I, Agarics of Trinidad. Kew Bull 15:231–248, pl. 1
Heinemann P (1978) Essai d’une clé de determination des genres Agaricus et Micropsalliota. Sydowia 30:6–37
Heinemann P (1980) Les genres AgaricusetMicropsalliota en Malaisie et en Indonésie. Bull Jard Bot Belg 50:3–68
Hennig W (1966) Phylogenetic systematics. Univ. of Illinois Press, Urbana
Hibbett DS, Matheny PB (2009) The relative ages of ectomycorrhizal mushrooms and their plant hosts estimated using Bayesian relaxed molecular clock analyses. BMC Biol 7:13
Hibbett DS, Grimaldi D, Donoghue MJ (1997) Fossil mushrooms from Miocene and Cretaceous ambers and the evolution of Homobasidiomycetes. Am J Bot 84(8):981–991
Huelsenbeck JP, Ronquist F (2001) MrBayes: Bayesian inferance of phylogeny. Biometrics 17:754–755
Hunag Y (2012) Molecular phylogenetics. Science Press, Beijing, p 533
Hyde KD, Jones EBG, Liu JK, Ariyawansa H, Boehm E, Boonmee S, Braun U, Chomnunti P, Crous PW, Dai DQ, Diederich P, Dissanayake A, Doilom M, Doveri F, Hongsanan S, Jayawardena R, Lawrey JD, Li YM, Liu YX, Lücking R, Monkai J, Muggia L, Nelsen MP, Pang KL, Phookamsak R, Senanayake I, Shearer CA, Suetrong S, Tanaka K, Thambugala KM, Wijayawardene NN, Wikee S, Wu HX, Zhang Y, Aguirre-Hudson B, Alias SA, Aptroot A, Bahkali AH, Bezerra JL, Bhat DJ, Camporesi E, Chukeatirote E, Gueidan C, Hawksworth DL, Hirayama K, Hoog SD, Kang JC, Knudsen K, Li WJ, Li XH, Liu ZY, Mapook A, McKenzie EHC, Miller AN, Mortimer PE, Phillips AJL, Raja HA, Scheuer C, Schumm F, Taylor JE, Tian Q, Tibpromma S, Wanasinghe DN, Wang Y, Xu JC, Yan JY, Yacharoen S, Zhang M (2013) Families of Dothideomycetes. Fungal Divers 63:1–313
István N (2009) Az Agaricus biberi Azonosítása és elöfordulása magyarországon. Mikol Közlem - Clusiana 48(1):37–44
Jayasiri SC, Hyde KD, Ariyawansa HA, Bhat J, Buyck B, Cai L, Dai YC, Abd-Elsalam KA, Ertz D, Hidayat I, Jeewon R, Jones EBG, Bahkali AH, Karunarathna SC, Liu JK, Luangsa-ard JJ, Lumbsch HT, Maharachchikumbura SSN, McKenzie EHC, Moncalvo JM, Ghobad-Nejhad M, Nilsson H, Pang KL, Pereira OL, Phillips AJL, Raspé O, Rollins AW, Romero AI, Etayo J, Selçuk F, Stephenson SL, Suetrong S, Taylor JE, Tsui CKM, Vizzini A, Abdel-Wahab MA, Wen TC, Boonmee S, Dai DQ, Daranagama DA, Dissanayake AJ, Ekanayaka AH, Fryar SC, Hongsanan S, Jayawardena RS, Li WJ, Perera RH, Phookamsak R, de Silva NI, Thambugala KM, Tian Q, Wijayawardene NN, Zhao RL, Zhao Q, Kang JC, Promputtha I (2015) The faces of fungi database: fungal names linked with morphology, molecular and human attributes. Fungal Divers 74:3–18
Kerrigan RW (1986) Agaricales of California. Vol. 6. Agaricaceae. Mad River Press, Eureka, pp 1–62
Kerrigan RW (2016) Agaricus of North America. New York Botanical Garden Press, Bronx, (in press)
Kerrigan RW, Callac P, Challen M, Guinberteau J, Parra LA (2005) Agaricus section Xanthodermatei: a phylogenetic reconstruction with commentary on taxa. Mycologia 97(6):1292–1315
Kerrigan RW, Callac P, Parra LA (2008) New and rare taxa in Agaricus section Bivelares (Duploannulati). Mycologia 100(6):876–892
Karunarathna SC, Guinberteau J, Chen J, Vellinga EC, Zhao RL, Chukeatirote E, Yan JY, Hyde KD, Callac P (2014) Two new species in Agaricus Tropical Clade I. Chiang Mai J Sci 41:771–780
Konrad P, Maublanc A (1952) Les Agaricales. Lechevalier, Paris
Kornerp A, Wanscher JH (1978) Methuen handbook of colour, 3rd edn. Eyre Methuen, London
Kühner P, Romagnesi H (1953) Flore analytique des champignons superieurs (Agarics, Boletes, Chanteralles). Masson et CIE, Paris
Largent DL (1986a) How to identify mushrooms to genus vol. 1. Macroscopic Features. Mad River Press, Eureka, p 166
Largent DL (1986b) How to identify mushrooms to genus vol. 3. Microscopic Features. Mad River Press, Eureka, p 148
Largeteau ML, Llarena-Hernández RC, Regnault-Roger C, Savoie J-M (2011) The medicinal Agaricus mushroom cultivated in Brazil: biology, cultivation and non-medicinal valorization. Appl Microbiol Biotechnol 92:897–907
Lebel T (2013) Two new species of sequestrate Agaricus (section Minores) from Australia. Mycol Prog 12:699–707
Lebel T, Syme A (2012) Sequestrate species of Agaricus and Macrolepiota from Australia: new species and combinations and their position in a calibrated phylogeny. Mycologia 104:496–520
Lepage T, Bryant D, Philippe H, Lartillot NA (2007) General comparison of relaxed molecular clock models. Mol Biol Evol 24(12):2669–2680
Li SF, Xi YL, Qi CX, Liang QQ, Wei SL, Li GJ, Zhao D, Li SJ, Wen HA (2014) Agaricus taeniatus sp. nov., a new member of Agaricus sect. Bivelares from northwest China. Mycotaxon 129:187–196
Maddison W, Maddison D (2007) Mesquite 2. http://mesquiteproject.org
Maharachchikumbura SSN, Hyde KD, Jones EBG, McKenzie EHC, Huang S-K, Abdel-Wahab MA, Daranagama DA, Dayarathne M, D’souza MJ, Goonasekara ID, Hongsanan S, Jayawardena RS, Kirk PM, Konta S, Liu J-K, Liu Z-Y, Norphanphoun C, Shenoy BD, Xiao Y, Bahkali AH, Kang J, Somrothipol S, Suetrong S, Wen T, Xu J (2015) Towards a natural classification and backbone tree for Sordariomycetes. Fungal Divers 72:199–301
Matheny PB, Wang Z, Binder M, Curtis JM, Lim YM, Nilsson RH, Hughes KW, Hofstetter V, Ammirati JF, Schoch CL, Langer E, Langer G, McLaughlin DJ, Wilson AW, FrØslev T, Ge ZW, Kerrigan RW, Slot GC, Yang ZL, Baroni TJ, Fischer M, Hosaka K, Matsuura K, Seidl MT, Vauras J, Hibbett DS (2007) Contributions of rpb2 and tef-1α to the phylogeny of mushrooms and allies (Basidiomycota, Fungi). Mol Phylogenet Evol 43:430–451
Mayden RL (1997) A hierarchy of species concepts: the denouement in the saga of the species problem. In: Claridge MF, Dawah HA, Wilson MP (eds) In species: the units of biodiversity. Chapman & Hall, London, pp 381–424
McPeek MA, Brown JM (2007) Clade age and not diversification rate explains species richness among animal taxa. Am Nat 169, E000
Mitchell AD (1999) Phylogenetic relationships of Agaricus species based on ITS-2 and 28S ribosomal DNA sequences. Mycologia 91(5):811–819
Möller FH (1950) Danish Psalliota species I. Friesia, IV, pp 1–60
Möller FH (1952) Danish Psalliota species II. Friesia, IV, pp 135–220
Moncalvo J-M, Lutzoni FM, Rehner SA, Johnson J, Vilgalys R (2000) Phyligenetic relationships of Agaric fungi based on nuclear large subunit ribosomal DNA sequences. Syst Biol 49:278–305
Moncalvo JM, Vilgalys R, Redhead SA, Johnson JE, James TY, Aime MC, Hofstetter V, Verduin SJW, Larsson E, Baroni TJ, Thorn RG, Jacobsson S, Clémençon H, Miller OK Jr (2002) One Hundred and Seventeen Clades of Euagarics. Mol Phylogenet Evol 23:357–400
Morehouse EA, James TY, Ganley ARD, Vilgalys R, Berger L, Murphy PJ, Longcore JE (2003) Multilocus sequence typing suggests that the chytrid pathogen of amphibians is a recently emerged clone. Mol Ecol 12:395–403
Moser M (1967–1983) Kleine Kryptogamenflora: Die Röhrlinge und Blatterpilze (polyporales, Boletales, Agaricales, Russulales). Gustav Fischer Verlag, Stuttgart
Nauta MM (2001) Agaricus L. and Allopsalliota Nanta et Bas. In: Noordeloos ME, Kuyper TW, Vellinga EC (eds) Flora Agaricina Neerlandica, Critical monographs on families of Agarics and Boleti occurring in the Netherlands. Vol. 5 Family Agaricaceae. A.A. Balkema Publishers, Lisse, pp 23–62
Parra LA (2008) Agaricus L. Allopsalliota, Nauta & Bas, Fungi Europaei, Volume 1. Edizioni Candusso, Alassio, p 824
Parra LA (2013) Agaricus L. Allopsalliota, Nauta & Bas, Fungi Europaei, Volume 1A. Candusso Edizioni s.a.s, Alassio, p 1168
Parra LA, Mua A, Cappelli A, Callac P (2011) Agaricus biannulatus sp. nov., a new species of the section Xanthodermatei collected in Sardinia and Sicily. Micol Veget Medit 26:3–20
Parra LA, Muñoz G, Callac P (2014) Agaricus caballeroi sp. nov., una nueva especie de la sección Nigrobrunnescentes recolectada en España. Micol Veget Medit 29:21–38
Peterson KR, Desjardin ED, Hemmes DE (2000) Agaricales of the Hawaiian Islands. 6. Agaricxaceae I. Agariceae: Agaricus and Melanophyllum. Sydowia 52(2):204–257
Phookamsak R, Liu JK, McKenzie EHC, Manamgoda DS, Ariyawansa HA, Thambugala KM, Dai DQ, Camporesi E, Chukeatirote E, Wijayawardene NN, Bahkali AH, Mortimer PEXJC, Hyde KD (2014) Revision of Phaeosphaeriaceae. Fungal Divers 68:159–238
Pilát A (1951) The Bohemian species of the genus Agaricus. Acta musei Nationalis Pragae, VII B
Rambaut A, Suchard M, Drummond AJ (2013) Tracer 1.6 http://tree.bio.ed.ac.uk/software/tracer/
Robinson NE, Robinson AB (2001) Molecular clocks. Proc Natl Acad Sci U S A 98(3):944–949
Ronquist F, Heulsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Sánchez-Ramírez S, Tulloss RE, Amalfi M, Moncalvo JM (2015) Palaeotropical origins, boreotropical distribution and increased rated of diversification in a clade of edible ectomycorrhizal mushrooms (Amanita section Caesareae). J Biogeogr 42(2):351–363
Singer R (1986) The Agaricales in modern taxonomy, 4th edn. Koeltz Scientific Books, Koenigsternm
Smith SY, Currah RS, Stockey RA (2004) Cretaceous and Eocene poroid hymenophores fromVancouver Island, British Columbia. Mycologia 96(1):180–186
Stadler T, Rabosky DL, Ricklefs RE, Bokma F (2014) On age and species richness of higher taxa. Am Nat 184:447–455
Stamatakis A (2006) RaxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22(21):2688–2690
Talavera G, Lukhtanov LA, Pierce NE, Vila R (2012) Establishing criteria for higher-level classification using molecular data: the systematics of Polyommatus blue butterflies (Lepidoptera, Lycaenidae). Cladistics 29(2):166–192
The Marie Curie SPECIATION Network (2012) What do we need to know about speciation? Trends Ecol Evol 27:27–39
Thongklang N, Nawaz R, Khalid AN, Chen J, Hyde KD, Zhao RL, Parra LA, Hanif M, Moinard M, Callac P (2014) Morphological and molecular characterization of three Agaricus species from tropical Asia (Pakistan, Thailand) reveals a new group in section Xanthodermatei. Mycologia 106:1220–1232
van Tuinen M, Torres CR (2015) Potential for bias and low precision in molecular divergence time estimation of the Canopy of Life: an example from aquatic bird families. Front Genet 6:203. doi:10.3389/fgene.2015.00203
Vellinga EC, Sysouphanthong P, Hyde KD (2011) The family of Agaricaceae: phylogenies and two new white-spored genera. Mycologia 103(3):494–509
Wang ZR, Parra LA, Callac P, Zhou JL, Fu WJ, Dui SH, Hyde KD, Zhao RL (2015) Edible species of Agaricus (Agaricaceae) from Xinjiang Province (Western China). Phytotaxa 202(3):185–197
Wasser SP (1980) Flora Fungorum RSS Ucrainicae: Agaricaceae Cohn. Naukova Dumka, Kiev
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a guide to methods and applications. Academic, New York, pp 315–322
Wisitrassameewong K, Karunarathna SC, Thongklang N, Zhao RL, Callac P, Chukeatirote E, Bahkali AH, Hyde KD (2012) Agaricus subrufescens: new to Thailand. Chiang Mai J Sci 39(2):281–291
Zhao RL, Desjardin DE, Callac P, Parra LA, Guinbereau J, Soytong K, Karunarathna S, Zhang Y, Hyde KD (2013) Two species of Agaricus sect. Xanthodermatei from Thailand. Mycotaxon 122:187–195
Zhao RL, Hyde KD, Desjardin DE, Raspé O, Soytong K, Guinberteau J, Karunarathna SC, Callac P (2012) Agaricus flocculosipes sp. nov., a new potentially cultivatable species from the palaeotropics. Mycoscience 53:300–311
Zhao RL, Karunarathna S, Raspé O, Parra LA, Guinberteau J, Moinard M, Kesel AD, Barroso G, Courtecuisse R, Hyde KD, Guelly AK, Desjardin DE, Callac P (2011) Major clades in tropical Agaricus. Fungal Divers 51:279–296
Zhou JL, Su SY, Su HY, Wang B, Hyde KD, Zhao RL (2016) A description of eleven new species of Agaricus sections Xanthodermatei and Hondenses collected from Tibet and the surrounding areas. Phytotaxa (in press)
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China to RLZ (Project IDs 31000013, 31360014 and 31470152), the Thailand Research Fund to KDH (grant BRG 5580009), and the Natural Sciences and Engineering Research Council of Canada and the ROM Governors to JMM. Dr. Richard Kerrigan made valuable comments and suggestions to improve this paper. Drs. Wang Xiang-Hua, Su Hong-Yang, István Nagy, Samantha C. Karunarathna and Phongeun Sysouphanthong lended specimens.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
(PDF 459 kb)
Rights and permissions
About this article
Cite this article
Zhao, RL., Zhou, JL., Chen, J. et al. Towards standardizing taxonomic ranks using divergence times – a case study for reconstruction of the Agaricus taxonomic system. Fungal Diversity 78, 239–292 (2016). https://doi.org/10.1007/s13225-016-0357-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13225-016-0357-x