Abstract
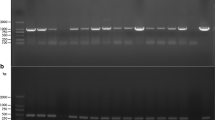
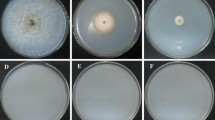
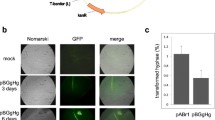
Gene recombination has been widely used in bacteria, yeast and other lower organisms for gene function research, but its application in filamentous fungi is uncommon because long homologous sequences are required. In this study, first, the task1 gene encoding a mitogen-activated protein kinase (MAPK) during fungal growth, mycoparasitic interaction, and biocontrol was cloned, and then we devised a novel and efficient PCR-based technique to amplify unknown regions adjacent to known genes using a single, specific primer. The feasibility of this technique was demonstrated by isolating sequences flanking the task1 gene of Trichoderma asperellum. The flanking regions obtained were used to construct a binary vector to knock out T. asperellum task1 gene by a Agrobacterium tumefaciens-mediated transformation method.






Similar content being viewed by others
References
An YF, Wu WF, Lv AG (2010) A PCR-after-ligation method for cloning of multiple DNA inserts. Anal Biochem 402:203–205
Cardoza RE, Vizcaíno JA, Hermosa MR, Sousa S, González FJ, Llobell A, Monte E, Gutiérrez S (2006) Cloning and characterization of the erg1 gene of Trichoderma harzianum: Effect of the erg1 silencing on ergosterol biosynthesis and resistance to terbinafine. Fungal Genet Biol 43:164–178
Figueiredo JG, Goulin EH, Stringari TD F, Cordeiro VK, Terasawa LV, Staats CC, Schrank A, Glienke C (2010) Agrobacterium tumefaciens-mediated transformation of Guignardia citricarpa. J Microb Meth 80:143–147
Guigon-Lopez C, Carvajal-Millan E, de Leon-Renova NP, Vargas-Albores F, Bravo-Luna L, Guerrero-Prieto VM (2010) Microcalorimetric measurement of Trichoderma spp. growth at different temperatures. Thermochim Acta 509:40–45
Hanada RE, Pomella WV, Soberanis W, Loguercio LL, Pereira JO (2009) Biocontrol potential of Trichoderma martiale against the black-pod disease (Phytophthora palmivora) of cacao. Biol Control 50:143–149
Hishida T, Iwasaki H, Ishioka K, Shinagawa H (1996) Molecular analysis of the Pseudomonas aeruginosa genes, ruvA, ruvB and ruvC, involved in processing of homologous recombination intermediates. Gene 182:63–70
Khrebtukova I, Michaud EJ, Foster CM, Stark KL, Garfinkel DJ, Woychik RP (1998) Utilization of microhomologous recombination in yeast to generate targeting constructs for mammalian genes. Mutat Res-Fund Mol M 401:11–25
Lewis JA, Lumsden RD (2001) Biocontrol of damping- off greenhouse-grown crops caused by Rhizoctonia solani with a formulation of Trichoderma spp. Crop Prot 20:49–56
Limansky AS, Viale AM (2002) Can composition and structural features of oligonucleotides contribute to their wide-scale applicability as random PCR primers in mapping bacterial genome diversity? J Microb Meth 50:291–297
Nthangeni MB, Ramagoma F, Tlou MG, Litthauer D (2005) Development of a versatile cassette for directional genome walking using cassette ligation-mediated PCR and its application in the cloning of complete lipolytic genes from Bacillus species. J Microb Meth 61:225–234
Olson HA, Benson DM (2007) Induced systemic resistance and the role of binucleate Rhizoctonia and Trichoderma hamatum 382 in biocontrol of Botrytis blight in geranium. Biol Control 42:233–241
Prod’hom G, Lagier B, Pelicic V, Hance AJ, Gicque B, Guilhot C (1998) A reliable amplification technique for the characterization of genomic DNA sequences flanking insertion sequences. FEMS Microbiol Lett 158:75–81
Rabeendran N, Jones EE, Moot DJ, Stewart A (2006) Biocontrol of Sclerotinia lettuce drop by Coniothyrium minitans and Trichoderma hamatum. Biol Control 39:352–362
Rafat A, Aziz MA, Rashid AA, Abdullah S, Na KH, Sirchi M, Ht JMB (2010) Optimization of Agrobacterium tumefaciens-mediated transformation and shoot regeneration after co-cultivation of cabbage (Brassica oleracea subsp. capitata) cv. KY Cross with AtHSP101 gene. Sci Hortic-Amsterdam 124:1–8
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Sun WL, Liu LX, Hu XL, Tang J, Liu P, Chen J, Chen YP (2009) Generation and identification of DNA sequence flanking T-DNA integration site of Trichoderma atroviride mutants with high dichlorvos-degrading capacity. Bioresour Technol 100:5941–5946
Sun WL, Chen YP, Liu LX, Tang J, Chen J, Liu P (2010) Conidia immobilization of T-DNA inserted Trichoderma atroviride mutant AMT-28 with dichlorvos degradation ability and exploration of biodegradation mechanism. Bioresour Technol 101:9197–9203
Vizcaíno JA, Sanz L, Cardoza RE (2005) Detection of putative peptide synthetase genes in Trichoderma species: Application of this method to the cloning of a gene from T. harzianum CECT 2413. FEMS Microbiol Lett 244:139–148
Wang DP, Sun W, Li MC, Wei DS, Zhang YH, Xing LJ (2006) A method using long primers for cloning the upstream sequence of Δ-6 fatty acid Desaturases Gene of Thamnidium elegans by Nested Inverse PCR. Chin J Biotechnol 4:581–586
Zhang PY, Xu B, Wang YZ, Li YQ, Qian Z, Tang SR, Huan S, Ren SX (2008) Agrobacterium tumefaciens-mediated transformation as a tool for insertional mutagenesis in the fungus Penicillium marneffei. Mycol Res 112:943–949
Acknowledgements
This project was supported by the National Science and Technology Pillar Program of China (2006BAD07A01), the National High Technology Research and Development Program 863 of China (2006AA10Z424) and a sub-project of National Science and Technology Support Programme of China (2007BAD65B 03–02). We are sincerely thankful to professor Seogchan Kang at the Department of Plant Pathology of the Pennsylvania State University, USA, for sharing vectors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, P., Yang, Q., Song, J. et al. Isolation of sequences flanking the Trichoderma asperellum task1 gene using a single specific primer PCR and their use for gene knockout. Ann Microbiol 62, 1557–1564 (2012). https://doi.org/10.1007/s13213-011-0410-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-011-0410-7