Abstract
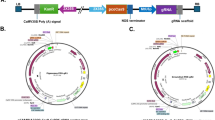
The gene editing using the CRISPR/Cas9 system has become an important biotechnological tool for studying gene function and improving crops. In this study, we have used CRISPR/Cas9 system for editing the phytoene desaturase gene (PDS) in popular Indian potato cultivar Kufri Chipsona-I. A construct (pHSE401) carrying two target gRNAs with glycine tRNA processing system under the control of Arabidopsis U6 promoter and the Cas9 protein was constructed and transformed in potato plants using Agrobacterium-mediated genetic transformations. The regeneration efficiency of 45% was observed in regenerated plants, out of which 81% of the putative transformants shoot lines exhibited mutant or bleached phenotype (albinism). The deletion mutations were detected within the StPDS gene in the genotyped plants and a mutation efficiency of 72% for gRNA1 and gRNA2 has been detected using Sanger sequencing. Hence, we set up a CRISPR/Cas9-mediated genome editing protocol which is efficient and generates mutations (deletions) within StPDS gene in potato. The bleached phenotype is easily detectable after only few weeks after Agrobacterium-mediated transformation. This is the first report as a proof of concept for CRISPR/Cas9-based editing of PDS gene in Indian potato cv. Kufri Chipsona-I. This study demonstrates that CRISPR/Cas9 can be used to edit genes at high frequency within the genome of the potato for various traits. Therefore, this study will aid in creating important mutants for modifying molecular mechanisms controlling traits of agronomic importance.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study have been included in this article.
References
Ashokkumar S, Jaganathan D, Ramanathan V et al (2020) Creation of novel alleles of fragrance gene OsBADH2 in rice through CRISPR/Cas9 mediated gene editing. PLoS ONE 15:e0237018. https://doi.org/10.1371/JOURNAL.PONE.0237018
Bouzroud S, Gasparini K, Hu G et al (2020) Down regulation and loss of auxin response factor 4 function using CRISPR/Cas9 alters plant growth, stomatal function and improves tomato tolerance to salinity and osmotic stress. Genes 11:272. https://doi.org/10.3390/genes11030272
Char SN, Neelakandan AK, Nahampun H et al (2017) An Agrobacterium-delivered CRISPR/Cas9 system for high-frequency targeted mutagenesis in maize. Plant Biotechnol J 15:257–268. https://doi.org/10.1111/pbi.12611
Doudna JA, Charpentier E (2014) The new frontier of genome engineering with CRISPR-Cas9. Science 346:1258096. https://doi.org/10.1126/science.1258096
Han E-H, Goo Y-M, Lee M-K, Lee S-W (2015) An efficient transformation method for a potato (Solanum tuberosum L. var. Atlantic). J Plant Biotechnol 42:77–82. https://doi.org/10.5010/JPB.2015.42.2.77
Han J, Guo B, Guo Y et al (2019) Creation of early flowering germplasm of soybean by CRISPR/Cas9 technology. Front Plant Sci 10:1446. https://doi.org/10.3389/fpls.2019.01446
Hooghvorst I, López-Cristoffanini C, Nogués S (2019) Efficient knockout of phytoene desaturase gene using CRISPR/Cas9 in melon. Sci Rep 9:17077. https://doi.org/10.1038/s41598-019-53710-4
Hunter CT, Ramachandran S (2021) CRISPR/Cas9 targeted mutagenesis for functional genetics in maize. Plants 10:723. https://doi.org/10.3390/PLANTS10040723
Ibrahim S, Saleem B, Rehman N et al (2021) CRISPR/Cas9 mediated disruption of Inositol Pentakisphosphate 2-Kinase 1 (TaIPK1) reduces phytic acid and improves iron and zinc accumulation in wheat grains. J Adv Res 37:33–41. https://doi.org/10.1016/J.JARE.2021.07.006
Jiang W, Zhou H, Bi H et al (2013) Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res 41:e188–e188. https://doi.org/10.1093/nar/gkt780
Kapusi E, Corcuera-Gómez M, Melnik S, Stoger E (2017) Heritable genomic fragment deletions and small indels in the putative ENGase gene induced by CRISPR/Cas9 in barley. Front Plant Sci 8:540. https://doi.org/10.3389/fpls.2017.00540
Kaur N, Alok A, Shivani J et al (2018) CRISPR/Cas9-mediated efficient editing in phytoene desaturase (PDS) demonstrates precise manipulation in banana cv. Rasthali genome. Funct Integr Genom 18:89–99. https://doi.org/10.1007/s10142-017-0577-5
Kaur A, Guleria S, Reddy MS, Kumar A (2020) A robust genetic transformation protocol to obtain transgenic shoots of Solanum tuberosum L. cultivar ‘Kufri Chipsona 1.’ Physiol Mol Biol Plants 26:367–377. https://doi.org/10.1007/s12298-019-00747-4
Komatsu H, Abdellatif IMY, Yuan S et al (2020) Genome editing in PDS genes of tomatoes by non-selection method and of Nicotiana benthamiana by one single guide RNA to edit two orthologs. Plant Biotechnol 37:213–221. https://doi.org/10.5511/plantbiotechnology.20.0527b
Ma X, Zhang Q, Zhu Q et al (2015) A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol Plant 8:1274–1284. https://doi.org/10.1016/j.molp.2015.04.007
Mao Y, Botella JR, Zhu J-K (2017) Heritability of targeted gene modifications induced by plant-optimized CRISPR systems. Cell Mol Life Sci 74:1075–1093. https://doi.org/10.1007/s00018-016-2380-1
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/ppl.1962.15.issue-3. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nasir MW, Toth Z (2022) Effect of drought stress on potato production: a review. Agronomy 12:635. https://doi.org/10.3390/agronomy12030635
Nishitani C, Hirai N, Komori S et al (2016) Efficient genome editing in apple using a CRISPR/Cas9 system. Sci Rep 6:31481. https://doi.org/10.1038/srep31481
Ohnuma M, Teramura H, Shimada H (2020) A simple method to establish an efficient medium suitable for potato regeneration. Plant Biotechnol 37:25–30. https://doi.org/10.5511/plantbiotechnology.19.1209a
Pan C, Ye L, Qin L et al (2016) CRISPR/Cas9-mediated efficient and heritable targeted mutagenesis in tomato plants in the first and later generations. Sci Rep 6:24765. https://doi.org/10.1038/srep24765
Pavese V, Moglia A, Corredoira E et al (2021) First report of CRISPR/Cas9 gene editing in Castanea sativa Mill. Front Plant Sci 12:728516. https://doi.org/10.3389/fpls.2021.728516
Qi W, Zhu T, Tian Z et al (2016) High-efficiency CRISPR/Cas9 multiplex gene editing using the glycine tRNA-processing system-based strategy in maize. BMC Biotechnol 16:58. https://doi.org/10.1186/s12896-016-0289-2
Qin G, Gu H, Ma L et al (2007) Disruption of phytoene desaturase gene results in albino and dwarf phenotypes in Arabidopsis by impairing chlorophyll, carotenoid, and gibberellin biosynthesis. Cell Res 17:471–482. https://doi.org/10.1038/cr.2007.40
Ren C, Liu Y, Guo Y et al (2021) Optimizing the CRISPR/Cas9 system for genome editing in grape by using grape promoters. Hortic Res 8:52. https://doi.org/10.1038/s41438-021-00489-z
Ren H, Xu Y, Zhao X et al (2022) Optimization of tissue culturing and genetic transformation protocol for Casuarina equisetifolia. Front Plant Sci 12:3096. https://doi.org/10.3389/fpls.2021.784566
Sharma N, Siddappa S, Malhotra N et al (2022) Advances in potato functional genomics: implications for crop improvement. Plant Cell Tissue Organ Cult 148:447–464
Singh B, Goutam U, Kukreja S et al (2021) Potato biofortification: an effective way to fight global hidden hunger. Physiol Mol Biol Plants 27:2297–2313. https://doi.org/10.1007/S12298-021-01081-4
Singh B, Sharma J, Bhardwaj V et al (2022) Genotypic variations for tuber nutrient content, dry matter and agronomic traits in tetraploid potato germplasm. Physiol Mol Biol Plants 28:1233–1248. https://doi.org/10.1007/S12298-022-01197-1
Távora FTPK, Meunier AC, Vernet A et al (2022) CRISPR/Cas9-targeted knockout of rice susceptibility genes OsDjA2 and OsERF104 reveals alternative sources of resistance to Pyricularia oryzae. Rice Sci 29:535–544. https://doi.org/10.1016/J.RSCI.2022.04.001
Tiwari JK, Devi S, Buckseth T et al (2020) Precision phenotyping of contrasting potato (Solanum tuberosum L.) varieties in a novel aeroponics system for improving nitrogen use efficiency: in search of key traits and genes. J Integr Agric 19:51–61. https://doi.org/10.1016/S2095-3119(19)62625-0
Tiwari JK, Buckseth T, Challam C et al (2022) CRISPR/Cas genome editing in potato: current status and future perspectives. Front Genet 13:82. https://doi.org/10.3389/fgene.2022.827808
Zhu C, Bortesi L, Baysal C et al (2017) Characteristics of genome editing mutations in cereal crops. Trends Plant Sci 22:38–52. https://doi.org/10.1016/j.tplants.2016.08.009
Authors are very thankful to Indian Council of Agricultural Research-National Agricultural Science Fund (Grant Number: NASF/GT-7024/2018-19) for providing the funds for current research. We are also very thankful to, ICAR-Central Potato Research Institute, Shimla, India for providing the facilities to conduct this research work.
Author information
Authors and Affiliations
Contributions
Concept and design: SS, SaS, VB, RM, KS. Experimental work: SS, NSh, NSa, KT, SP, BS, HS. Supervision: SS, VB, AKT. Writing—original draft: NSh, BS. Writing—review and editing: SS, VB, VM, RT. Resource: VB, VK. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Siddappa, S., Sharma, N., Salaria, N. et al. CRISPR/Cas9-mediated editing of phytoene desaturase (PDS) gene in an important staple crop, potato. 3 Biotech 13, 129 (2023). https://doi.org/10.1007/s13205-023-03543-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03543-w