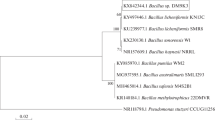
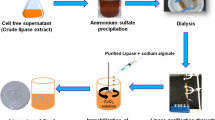
Abstract
The present study was conducted to purify lipase from indigenous Bacillus subtilis strain Kakrayal_1 (BSK-L) for enantioselective resolution of racemic-ketoprofen. The production of lipase (BSK-L) was optimized using Plackett–Burman and central composite design of response surface methodology (RSM). The optimized media containing olive oil (3.5%), MnSO4 (8 mM), CaCl2 (5 mM), peptone (20 g/l), pH (8), agitation (180 rpm) and temperature (37 °C) resulted in maximum lipase production of 7500 U/g of cell biomass. The lipase was purified using sequential method to an overall purification fold of 13% with 20% recovery, 882 U/mg specific activity and a molecular weight of 45 kDa. Optimal pH and temperature of purified lipase were found to be 8 and 37 °C, respectively. Furthermore, BSK-L displayed good stability with various organic solvents, surfactants and metal ions. Km and Vmax values of lipase were observed to be 2.2 mM and 6.67 mmoles of product formed/min/mg, respectively. The racemic ketoprofen butyl ester was hydrolyzed using lipase with 49% conversion efficiency and 69% enantiomeric excess (ee) which was superior to the commercially procured lipase (Candida antarctica lipase). Thus, this enzyme could be considered as a promising candidate for the pharmaceutical industry.




Similar content being viewed by others
References
Abdel-Aziz AA-M, Al-Badr AA, Hafez GA (2012) Flurbiprofen. In: Brittain HG (ed) Profiles of drug substances, excipients and related methodology. Academic Press, Cambridge, pp 113–181
Bakir ZB, Metin K (2016) Purification and characterization of an alkali-thermostable lipase from thermophilic Anoxybacillus flavithermus HBB 134. J Microbiol Biotechnol 26:1087–1097. https://doi.org/10.4014/jmb.1512.12056
Bento HBS, de Castro HF, de Oliveira PC, Freitas L (2017) Magnetized poly(STY-co-DVB) as a matrix for immobilizing microbial lipase to be used in biotransformation. J Magn Magn Mater 426:95–101. https://doi.org/10.1016/J.JMMM.2016.11.061
Bhosale HJ, Uzma SZ, Bismile PC (2015) Optimization of lipase production by thermo-alkalophilic Bacillus sp. 8C. Res J Microbiol 10:523–532. https://doi.org/10.3923/jm.2015.523.532
Bhushan I, Parshad R, Nabi G et al (2008) Lipase enzyme immobilization on synthetic beaded macroporous copolymers for kinetic resolution of chiral drugs intermediates. Process Biochem 43:321–330. https://doi.org/10.1016/j.procbio.2007.11.019
Borkar PS, Bodade RG, Rao SR, Khobragade CN (2009) Purification and characterization of extracellular lipase from a new strain: Pseudomonas aeruginosa SRT 9. Braz J Microbiol 40:358–366. https://doi.org/10.1590/S1517-838220090002000028
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cadirci BH, Yasa I (2010) An organic solvents tolerant and thermotolerant lipase from Pseudomonas fluorescens P21. J Mol Catal B Enzym 64:155–161. https://doi.org/10.1016/J.MOLCATB.2009.09.009
Caroline A, Melo L, Fonseca TDS et al (2015) Recent Advances in lipase-mediated preparation of pharmaceuticals and their intermediates. Int J Mol Sci. https://doi.org/10.3390/ijms161226191
Casas-Godoy L, Duquesne S, Bordes F et al (2012) Lipases: an overview. In: Clifton N (ed) Methods in molecular biology. Humana Press, Guadalajara, Mexico, pp 3–30. https://doi.org/10.1007/978-1-61779-600-5_1
Castro-Ochoa LD, Rodríguez-Gómez C, Valerio-Alfaro G, Oliart Ros R (2005) Screening, purification and characterization of the thermoalkalophilic lipase produced by Bacillus thermoleovorans CCR11. Enzyme Microb Technol 37:648–654. https://doi.org/10.1016/j.enzmictec.2005.06.003
Chang S-W, Shaw J-F, Yang K-H et al (2008) Studies of optimum conditions for covalent immobilization of Candida rugosa lipase on poly(γ-glutamic acid) by RSM. Bioresour Technol 99:2800–2805. https://doi.org/10.1016/j.biortech.2007.06.020
Chen S, Qian L, Shi B (2007) Purification and properties of enantioselective lipase from a newly isolated Bacillus cereus C71. Process Biochem 42:988–994. https://doi.org/10.1016/j.procbio.2007.03.010
Cubides-Roman DC, Pérez VH, de Castro HF et al (2017) Ethyl esters (biodiesel) production by Pseudomonas fluorescens lipase immobilized on chitosan with magnetic properties in a bioreactor assisted by electromagnetic field. Fuel 196:481–487. https://doi.org/10.1016/j.fuel.2017.02.014
Dlugy C, Wolfson A (2007) Lipase catalyse glycerolysis for kinetic resolution of racemates. Bioprocess Biosyst Eng 30:327–330. https://doi.org/10.1007/s00449-007-0128-x
Duong-Ly KC, Gabelli SB (2014) Salting out of Proteins Using Ammonium Sulfate Precipitation. In: Methods in enzymology. pp 85–94
Esmaeili M, Yolmeh M, Shakerardakani A, Golivari H (2015) A central composite design for the optimizing lipase and protease production from Bacillus subtilis PTCC 1720. Biocatal Agric Biotechnol 4:349–354. https://doi.org/10.1016/j.bcab.2015.05.002
Golaki BP, Aminzadeh S, Karkhane AA et al (2015) Cloning, expression, purification, and characterization of lipase 3646 from thermophilic indigenous Cohnella sp. A01. Protein Expr Purif 109:120–126. https://doi.org/10.1016/j.pep.2014.10.002
Gururaj P, Ramalingam S, Devi GN et al (2016) Process optimization for production and purification of a thermostable, organic solvent tolerant lipase from Acinetobacter sp. AU07. Brazil J Microbiol 47:647–657. https://doi.org/10.1016/j.bjm.2015.04.002
Joseph B, Ramteke PW (2013) Extracellular solvent stable cold-active lipase from psychrotrophic Bacillus sphaericus MTCC 7526: Partial purification and characterization. Ann Microbiol 63:363–370. https://doi.org/10.1007/s13213-012-0483-y
Kambourova M, Kirilova N, Mandeva R (2003) Purification and properties of thermostable lipase from a thermophilic Bacillus stearothermophilus MC 7. J Mol Catal B Enzym 22:307–313
Kantor TG (1986) Ketoprofen: A Review of Its Pharmacologic and Clinical Properties. Pharmacother J Hum Pharmacol Drug Ther 6:93–102. https://doi.org/10.1002/j.1875-9114.1986.tb03459.x
Kaur G, Singh A, Sharma R et al (2016) Cloning, expression, purification and characterization of lipase from Bacillus licheniformis, isolated from hot spring of Himachal Pradesh, India. 3 Biotech 6:49–59. https://doi.org/10.1007/s13205-016-0369-y
Köse Ö, Tüter M, Aksoy HA (2002) Immobilized Candida antarctica lipase-catalyzed alcoholysis of cotton seed oil in a solvent-free medium. Bioresour Technol 83:125–129. https://doi.org/10.1016/S0960-8524(01)00203-6
Kumar S, Kikon K, Upadhyay A et al (2005) Production, purification, and characterization of lipase from thermophilic and alkaliphilic Bacillus coagulans BTS-3. Protein Expr Purif 41:38–44. https://doi.org/10.1016/J.PEP.2004.12.010
Kumar D, Nagar S, Bhushan I et al (2013) Covalent immobilization of organic solvent tolerant lipase on aluminum oxide pellets and its potential application in esterification reaction. J Mol Catal B Enzym 87:51–61. https://doi.org/10.1016/J.MOLCATB.2012.10.002
Kumar D, Parshad R, Gupta VK (2014) Application of a statistically enhanced, novel, organic solvent stable lipase from Bacillus safensis DVL-43. Int J Biol Macromol 66:97–107. https://doi.org/10.1016/j.ijbiomac.2014.02.015
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lesuisse E, Schanck K (1993) Purification and preliminary characterization of the extracellular lipase of Bacillus subtilis 168, an extremely basic pH tolerant enzyme. Eur J Biochem 216:155–160
Liebeton K, Zacharias A, Jaeger K-E (2001) Disulfide Bond in Pseudomonas aeruginosa Lipase Stabilizes the Structure but Is Not Required for Interaction with Its Foldase. J Bacteriol 183:597–603. https://doi.org/10.1128/JB.183.2.597-603.2001
Ma J, Ma Y, Wei W, Wei D (2015) In vivo functional expression of an extracellular Ca 2 + -independent Bacillus pumilus lipase in Bacillus subtilis WB800N. Ann Microbiol 65:1973–1983. https://doi.org/10.1007/s13213-015-1035-z
MarketsandMarkets (2018) Microbial Lipase Market: Global Forecast until 2023. In: ReportLinker. https://www.reportlinker.com/p05376210/Microbial-Lipase-Market-by-Application-Form-Source-And-Region-Global-Forecast-to.html. Accessed 23 Apr 2018
Mobarak-Qamsari E, Kasra-Kermanshahi R, Moosavi-Nejad Z (2011) Isolation and identification of a novel, lipase-producing bacterium, Pseudomnas aeruginosa KM110. Iran J Microbiol 3:92–98
Qin B, Liang P, Jia X et al (2013) Directed evolution of Candida antarctica lipase B for kinetic resolution of profen esters. Catal Commun 38:1–5. https://doi.org/10.1016/J.CATCOM.2013.03.040
Rigo E, Ninow JL, Di Luccio M et al (2010) Lipase production by solid fermentation of soybean meal with different supplements. LWT - Food Sci Technol 43:1132–1137. https://doi.org/10.1016/J.LWT.2010.03.002
Sajna KV, Sukumaran RK, Jayamurthy H et al (2013) Studies on biosurfactants from Pseudozyma sp. NII 08165 and their potential application as laundry detergent additives. Biochem Eng J 78:85–92. https://doi.org/10.1016/j.bej.2012.12.014
Saraswat R, Verma V, Sistla S, Bhushan I (2017) Evaluation of alkali and thermotolerant lipase from an indigenous isolated Bacillus strain for detergent formulation. Electron J Biotechnol. https://doi.org/10.1016/j.ejbt.2017.08.007
Saxena RK, Davidson WS, Sheoran A, Giri B (2003) Purification and characterization of an alkaline thermostable lipase from Aspergillus carneus. Process Biochem 39:239–247. https://doi.org/10.1016/S0032-9592(03)00068-2
Seddigi ZS, Malik MS, Ahmed SA et al (2017) Lipases in asymmetric transformations: recent advances in classical kinetic resolution and lipase–metal combinations for dynamic processes. Coord Chem Rev 348:54–70. https://doi.org/10.1016/J.CCR.2017.08.008
Shah R, Bhatt S (2011) Purification and characterization of lipase from Bacillus subtilis Pa2. J Biochem Technol 3:292–295
Sharma R, Soni S, Vohra R et al (2002) Purification and characterisation of a thermostable alkaline lipase from a new thermophilic Bacillus sp. RSJ-1. Process Biochem 37:1075–1084
Sharma D, Kumbhar BK, Verma AK, Tewari L (2014) Optimization of critical growth parameters for enhancing extracellular lipase production by alkalophilic Bacillus sp. Biocatal Agric Biotechnol 3:205–211. https://doi.org/10.1016/j.bcab.2014.04.004
Shi J, Deng Q, Wan C et al (2017) Fluorometric probing of the lipase level as acute pancreatitis biomarkers based on interfacially controlled aggregation-induced emission (AIE). Chem Sci 8:6188–6195. https://doi.org/10.1039/c7sc02189e
Sivaramakrishnan R, Incharoensakdi A (2016) Purification and characterization of solvent tolerant lipase from Bacillus sp. for methyl ester production from algal oil. J Biosci Bioeng 121:517–522. https://doi.org/10.1016/J.JBIOSC.2015.09.005
Sivaramakrishnan R, Muthukumar K (2012) Isolation of thermo-stable and solvent-tolerant Bacillus sp. lipase for the production of biodiesel. Appl Biochem Biotechnol 166:1095–1111. https://doi.org/10.1007/s12010-011-9497-3
Treichel H, de Oliveira D, Mazutti MA et al (2010) A review on microbial lipases production. Food Bioprocess Technol 3:182–196. https://doi.org/10.1007/s11947-009-0202-2
Tripathi R, Singh J, Bharti R, Thakur IS (2014) Isolation, purification and characterization of lipase from Microbacterium sp. and its application in biodiesel production. Energy Procedia 54:518–529. https://doi.org/10.1016/J.EGYPRO.2014.07.293
Unni KN, Priji P, Sajith S et al (2016) Pseudomonas aeruginosa strain BUP2, a novel bacterium inhabiting the rumen of Malabari goat, produces an efficient lipase. Biologia (Bratisl) 71:378–387. https://doi.org/10.1515/biolog-2016-0057
van Dijl JM, Hecker M (2013) Bacillus subtilis: from soil bacterium to super-secreting cell factory. Microb Cell Fact 12:3. https://doi.org/10.1186/1475-2859-12-3
Wang H, Zhong S, Ma H et al (2012) Screening and characterization of a novel alkaline lipase from Acinetobacter calcoaceticus 1–7 isolated from Bohai Bay in china for detergent formulation. Braz J Microbiol 43:148–156. https://doi.org/10.1590/S1517-838220120001000016
Zhang W, Jia J, Wang N et al (2015) Improved activity of lipase immobilized in microemulsion-based organogels for (R, S) -ketoprofen ester resolution : Long-term stability and reusability. Biotech Rep 7:1–8
Acknowledgements
The authors are obliged for the work supported by Shri Mata Vaishno Devi University, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no conflict of interest and the article also complies with the ethical standard.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saraswat, R., Bhushan, I., Gupta, P. et al. Production and purification of an alkaline lipase from Bacillus sp. for enantioselective resolution of (±)-Ketoprofen butyl ester. 3 Biotech 8, 491 (2018). https://doi.org/10.1007/s13205-018-1506-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1506-6