Abstract
Rhizobium BICC 651, a fast-growing strain isolated from root nodule of chickpea (Cicer arietinum L.), produced a catechol siderophore to acquire iron under iron poor condition. A Tn5-induced mutant (B153) of the strain, BICC 651 impaired in siderophore biosynthesis was isolated and characterized. The mutant failed to grow on medium supplemented with iron chelator and grew less efficiently in deferrated broth indicating its higher iron requirement. The mutant produced less number of nodules than its parent strain. The Tn5 insertion in the mutant strain, B153, was located on a 2.8 kb SalI fragment of the chromosomal DNA. DNA sequence analysis revealed that the Tn5-adjoining genomic DNA region contained a coding sequence homologous to agbB gene of Agrobacterium tumefaciens MAFF301001. About 5 kb genomic DNA region of the strain BICC 651 was amplified using the primers designed from DNA sequence of agrobactin biosynthesis genes of A. tumefaciens MAFF 301001 found in the database. From the PCR product of the strain BICC 651, a 4,921 bp DNA fragment was identified which contained four open reading frames. These genes were designated as sid, after siderophore. The genes were identified to be located in the order of sidC, sidE, sidB, and sidA. Narrow intergenic spaces between the genes indicated that they constitute an operon. Phylogenetic analyses of deduced sid gene products suggested their sequence similarity with the sequences of the enzymes involved in biosynthesis of catechol siderophore in other bacteria.
Similar content being viewed by others
Introduction
In iron deficient environment, bacteria meet their iron requirement by producing low-molecular-mass iron-chelating compounds called siderophores. The compounds bind available Fe3+ with high affinity to form complexes which are internalized by the cells with the help of cognate membrane proteins. The structures of these compounds are quite variable. Many of them are catecholate in nature. Catecholate siderophores were isolated from a variety of bacterial species including Escherichia coli which produces enterobactin, the prototype of catecholate siderophore. Enterobactin is biosynthesized from 2,3-dihydroxybenzoic acid (DHBA) which, in turn is synthesized from chorismate via the consecutive actions of three enzymes, viz., isochorismate synthase (EntC), isochorismatase (EntB) and 2,3-dihydro-2,3-dihydroxybenzoate dehydrogenase (EntA) (Earhart 1996). Although, the enzymes involved in the pathway of DHBA biosynthesis are common to all catechol siderophore-producing organisms, the organization of the biosynthetic genes within the operon and the sequences of the genes are different.
The members of the genus Rhizobium are soil bacteria that enter into symbiotic relationship with plant hosts. This relationship is iron dependent, since iron is required for nodule formation, and synthesis of leghemoglobin, nitrogenase complex, ferredoxin and other electron transport proteins to energise the nitrogenase system during symbiosis (Guerinot 1991). Because a part of life cycle of Rhizobium requires invasion, growth and differentiation within its host plant tissue, the role of siderophore for iron acquisition is important for the plant–microbe interaction. Improved iron scavenging properties of Bradyrhizobium sp. and Sinorhizobium meliloti 1021 positively correlate with rhizospheric growth and nodulation effectiveness in peanut and in alfalfa (O’Hara et al. 1988; Gill et al. 1991). Moreover, siderophore-producing ability helps in the sustenance of rhizobia in iron-deficient soils (Lesueur et al. 1995). Benson et al. (2005) reported that the gene fegA required for utilization of ferrichrome of B. japonicum 61A152 was also required for symbiosis. When the gene with its native, promoter was cloned and transferred to Mesorhizobium sp. GN25, nodulating peanut (Joshi et al. 2008) and Rhizobium sp. ST1, nodulating pigeonpea (Joshi et al. 2009), the transconjugants became symbiotically more efficient indicating importance of iron uptake protein in rhizobium-legume symbiosis.
Siderophore biosynthesis genes were studied in Rhizobium leguminosarum bv. viciae (Carter et al. 2002) and in Sinorhizobium meliloti 1021 (Lynch et al. 2001). In both the systems, siderophore biosynthesis genes were located on plasmids and were clustered close to the genes encoding their cognate membrane proteins. Siderophores produced by these strains were not of catechol type. Catecholate siderophores were isolated from R. leguminosarum bv. trifolii (Skorupska et al. 1989), R. ciceri (Roy et al. 1994; Berraho et al. 1997), Bradyrhizobium (cowpea) (Modi et al. 1985), Bradyrhizobium (peanut) (Nambiar and Sivaramakrishnan 1987), but the biosynthetic genes have not been investigated yet.
A fast-growing Rhizobium strain BICC 651 was isolated from a nodule produced on root of a chickpea (Cicer arietinum L.) plant grown in the Experimental Farm of Bose Institute at Madhyamgram, West Bengal, India. The strain produced a catechol siderophore and its cognate membrane receptor in response to iron deficiency (Roy et al. 1994). Structurally, the siderophore produced by Rhizobium BICC 651 contains 2,3-dihydroxybenzoic acid as core compound with two moles of threonine as the ligand. This suggested that the strain BICC 651 is likely to possess enzymes for siderophore biosynthesis similar to those of other catechol siderophore-producing organisms and the biosynthetic mechanism of the siderophore could be similar to that in other catechol siderophore-producing organisms. In the present study, transposon Tn5-mob insertional mutagenesis of the Rhizobium strain BICC 651 was performed to generate mutants impaired in siderophore production. One of the mutants was exploited to find out siderophore biosynthetic genes of the Rhizobium strain BICC 651.
Materials and methods
Bacterial strains, plasmid and culture media
Bacterial strains and plasmid used in the study are listed in Table 1. Escherichia coli and Rhizobium strains were cultured in Luria–Bertani (LB) medium and yeast extract mannitol (YEM) medium, respectively. The complete medium described by Modi et al. (1985) [composition (g/l): K2HPO4, 0.5; MgSO4. 7H2O, 0.4; NaCl, 0.1; mannitol, 10; glutamine, 1; and NH4NO3, 1; pH 6.8] was used to study the growth and siderophore production of the strain BICC 651 and its mutants. For the purpose, the medium was deferrated with hydroxyquinoline (Meyer and Abdallah 1978). CAS-agar plate for detection of siderophore production by the organisms was prepared according to Schwyn and Neilands (1987) using the complete medium.
Tn5 mutagenesis
Transconjugates were generated by plate mating method (Mukhopadhyaya et al. 2000). Escherichia coli S17.1, harbouring the suicide plasmid pSUP5011::Tn5-mob (Simon 1984) was used as the donor and a rifampicin (100 μg/ml) resistant spontaneous mutant of the Rhizobium strain BICC 651, designated as Rhizobium BICC 651R was used as the recipient. Fresh cultures of both the donor and the recipient were mixed, centrifuged to collect the cells and spread on plates containing tryptone-yeast extract agar medium [composition (g/l): bactotryptone, 8; bactoyeast, 5; NaCl, 5; and agar 20; pH 6.8] (Beringer 1974). The mating mixture was incubated overnight at 30 °C. The cells were then collected, suspended in YEM broth and plated on Rhizobium medium (Himedia) containing neomycin (50 μg/ml) and rifampicin (100 μg/ml). The plates were incubated at 28 °C for the development of isolated colonies of transconjugants. The colonies were picked to prepare master plates for post mating selection. Each master plate was replica plated on CAS plate. The mutants which did not produce orange halo were selected as siderophore negative (Sid−) mutants.
Bacterial growth and siderophore production
The parent strain BICC 651R, and its Sid− mutant, B153, were examined for their growth on Rhizobium medium in presence of increasing concentrations of bipyridine, a synthetic iron chelator. Subsequently, the parent strain and the mutant were studied for their growth and siderophore production in deferrated complete medium. The cultures were incubated on a shaker at 28 °C and growth was measured by monitoring OD590 of the culture. Siderophore in the culture supernatant was assayed using the CAS reagent (Schwyn and Neilands 1987).
Plant inoculation and nodulation assay
For nodulation study, surface-sterilized seeds of chickpea were inoculated with Rhizobium strains and planted in sea sand in pots. Sea sand was treated with HCl, washed with distilled water until the pH of the washing was neutral. It was then air dried and fumigated with chloroform overnight under airtight condition, autoclaved and finally kept at 200 °C in a hot air oven for 6 h. Five hundreds gram of sterile sand in an earthen pot was moistened with 100 ml of nitrogen-free plant nutrient medium for sowing the seeds. The composition of the nitrogen-free medium was (g/l): CaSO4. 2H2O, 0.34; K2HPO4, 0.17; MgSO4. 7H2O, 0.25; Fe Citrate, 0.002; KCl, 0.075; trace element solution, 0.5 ml; pH 7.2 (Norris and Date 1976). The composition of the trace element solution was (g/l): ZnSO4. 7H2O, 2.25; CuSO4. 5H2O, 1.00; MnSO4. 5H2O, 0.50; CaCl2. 2H2O, 2.00 Na2B4O7. 10H2O, 0.23; (NH4)6Mo7O24, 0.10. Surface sterilization of chickpea seeds was carried out by soaking in concentrated H2SO4 for 10 min, then washing the seeds several times with sterile distilled water. The parent strain BICC 651R and its Sid− mutant B153, were grown in YEM broth till late log phase, and the harvested cells were mixed with sand-charcoal (1:3) containing 2 % aqueous sodium carboxymethyl cellulose. The mixture was used to coat the surface-sterilized seeds. The coated seeds (~108 bacteria/seed) were kept in dark for overnight and the next day, five seeds were transferred to each pot containing the sterile sand soaked in nitrogen-free plant nutrient medium. A control set of seeds which did not receive any inoculum was also included in this study. The pots were kept under well-illuminated condition and watered when necessary to moisten the sand. At 1 week interval, 50 ml of 1/10th dilution of nitrogen-free plant nutrient medium was added to each pot. At 35 days of inoculation, plants were uprooted and observed for development of nodules.
DNA preparations and Southern hybridization
Total DNA of the parent, BICC 651R and the mutant, B153, was isolated, digested with SalI, electrophoresed on 1 % agarose gel according to standard protocol (Sambrook et al. 1989). DNA fragments from agarose gel were blotted on nylon membrane and hybridized with probe prepared from Tn5 fragment containing neomycin resistance (neor) gene. The probe was labelled by biotinylation following the manufacturer’s instruction (NE Blot Phototope Kit, Biolabs).
DNA ligation and inverse PCR
The SalI digested DNA fragments showing positive signal following hybridization with labelled probe were eluted from the gel and allowed to self-ligate by incubating the DNA at a concentration of 0.3–0.5 μg/ml in presence of 3 U of T4 DNA ligase (Promega)/ml overnight at 4 °C (Huang et al. 2000). The ligation mixture was extracted by phenol: chloroform, the DNA was precipitated with ethanol, and dissolved in sterile distilled water to a concentration of 20 μg/ml. The inverse PCR was carried out using the self-ligated product containing a portion of the Tn5 as template and Tn5Int and NeoF as primers (Table 2). The amplifications were performed using Perkin-Elmer PCR system 2,400 in 50 μl of reaction mixture containing 1 × enzyme buffer, 1.5 mM MgCl2, 200 μM dNTPs (each), 500 nM of each primers and 1.5 U Taq DNA polymerase (Fermentas) and 50 ng of purified self-ligated product. The thermal programme used for PCR was as follows: initial denaturation for 10 min at 94 °C; 35 cycles of denaturation for 30 s at 94 °C, annealing for 30 s at 55 °C, and extension at 72 °C for 1 min, followed by a final extension at 72 °C for 10 min. The identity of the PCR product was confirmed by nested PCR and also by ascertaining the profile of bands resulting from its specific endonuclease digestion. The PCR product was purified and sequenced by ABI PRISM 377 automated DNA sequencer (Perkin-Elmer, Applied Biosystem, Inc.).
Amplification of agb homologues from genomic DNA of BICC 651 and site of Tn5 insertion in Sid− mutant
To amplify the homologues of agrobactin biosynthetic genes from the strain BICC 651, PCR amplifications were carried out using the genomic DNA of BICC 651 as template, and agbCF and agbAR as primers (Table 2). A long PCR amplification kit was used for the purpose following the manufacturer’s instruction (Genei, India). To identify the position of Tn5 insertion in the genomic DNA of the mutant, B153, two sets of PCR were carried out, one with the primer pair, agbCF and Tn5Int and another with the primer pair agbAR and Tn5Int. The reaction mixture for amplification contained 100 ng of genomic DNA as template and the thermal programme remained the same as described earlier for IPCR. To amplify larger (>2 kb) DNA fragment, extension time was increased by 1 min for each 1 kb of desired amplicon during final extension. The PCR products were analysed by agarose gel electrophoresis, purified and sequenced.
Analysis of the Tn5 adjacent DNA sequence
Genomic DNA sequence was analyzed using BLAST version 2.2.1 of National Center for Biotechnology Information. Nucleotide BLAST and BLASTX were used to search for nucleotide sequences and derivative amino acid sequences, respectively.
Genomic context analysis
Gene context analysis of sidB gene of Rhizobium strain BICC 651 was carried out using the GeConT programme. The program allowed comparison of adjacent genes of sidB and visualization of genomic context of sidB homologue in the genomes of other organisms. The program is available at: http://www.ibt.unam.mx/biocomputo/gecont.html (Ciria et al. 2004).
Phylogenetic analysis
Evolutionary analyses were conducted using the software MEGA5 (Tamura et al. 2011). Multiple sequence alignment was carried out using a CLUSTALW and evolutionary history of the sequence was inferred using the neighbor-joining method (Saitou and Nei 1987). The optimal tree with the sum of branch length is equal to 4.31786458 is shown. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances are computed using the Poisson correction method (Zuckerkandl and Pauling 1965) and are in units of the number of amino acid substitutions per site.
Results and discussion
Transposon mutagenesis, and isolation of Sid− mutants
Transposon insertion mutagenesis in a siderophore producing Rhizobium sp. of chickpea was carried out to select the siderophore negative mutants to identify the structural genes required for biosynthesis of siderophore in the organism. Random mutagenesis of the Rhizobium strain BICC 651R by mobilization of Tn5-mob from the suicide vector pSUP5011 to the recipient cells occurred at a frequency of 10−5 per donor as revealed by the number of neomycin-resistant transconjugants. This is about 1,000 times greater than the rate of spontaneous resistance of the recipients to neomycin (10−8). Similar transposition frequency had also been reported for other bacteria (Mukhopadhyaya et al. 2000; Manjanatha et al. 1992). Upon screening of the 1,000 of transconjugants, five Sid− mutants were isolated. Among these, one mutant (B153) was characterized in the present study.
Study of iron requirement
The amount of iron required for optimal growth of many Gram-negative bacteria ranged from 0.36 to 1.8 μM (Lankford 1973). The strain BICC 651R was able to grow on Rhizobium medium containing bipyridine up to a concentration of 400 μM but the Sid− mutant, B153, could not grow even in the presence of 50 μM of bipyridine (Table 3). When the organisms were grown in the complete medium deferrated with hydroxyquinoline, the highest level of growth of the Sid− mutant B153 (OD590 = 1) was almost half as that of the parent strain (OD590 > 2). These observations suggested that because of impaired biosynthesis of siderophore, the mutant required more iron for its growth than the parent strain. Siderophore was not detected in the culture filtrate of the mutant at all stages of its growth, whereas the parent strain was observed to produce as much as 70 nmole of siderophore per ml of culture filtrate during late log phase of its growth (at 48 h) in the deferrated complete medium (Fig. 1).
Plant nodulation study
Table 4 presents the data of symbiotic performance of the siderophore non producing mutant B153 and its parent strain BICC 651R. The control plants receiving no inoculum produced no nodules. The plants inoculated with B153 produced less number of nodules than those inoculated with the parent strain BICC 651R. The wet weight of nodules produced by the mutant B153 was almost half as compared to those of its parents. Gill et al. (1991) reported that mutants of Sinorhizobium meliloti which were unable to produce siderophore were able to nodulate the plants but the efficiency of the nodules in nitrogen fixation was less as compared to wild type incited nodules indicating the importance of iron in symbiotic N2 fixation.
Identification of the DNA fragments containing Tn5
SalI cuts the 7.7 kb Tn5-mob DNA into two parts: a 2.7 kb part with neor gene and a 5 kb part containing the 1.9 kb mob region. When the Tn5 probe prepared from Tn5 fragment containing neor gene was allowed to hybridize with SalI restricted genomic DNAs of BICC 651R and its Sid− mutant, B153, it showed positive signal only as a single band of 2.8 kb genomic DNA of the mutant indicating insertion of a single copy of Tn5 in the genome of the mutant and no hybridization of the probe was detected with the genomic DNA of the parent, BICC 651R. In the mutant, the 2.8 kb SalI restricted genomic DNA fragment contained the 2.7 kb Tn5 fragment adjoining nearly a 0.1 kb genomic DNA fragment. The 2.8 kb SalI fragment was used to self-ligate more easily than using a larger (>7.7 kb) EcoRI restricted fragment. The Tn5 adjoining genomic DNA fragments were amplified by inverse PCR using the Tn5Int and NeoF primer pair (Table 2). A 1.3 kb amplicon containing an almost 0.1 kb genomic DNA was obtained (Fig. 2a). The genomic DNA fragment was sequenced and the sequence showed 94 % identity with the agbB gene sequence of Agrobacterium tumefaciens MAFF301001 (GenBank accession no. AB083344). The agbB gene product, isochorismatase, catalyzes the synthesis of 2,3-dihydro 2,3-dihydroxybenzoic acid from isochorismate (Sonoda et al. 2002). Thus, in the mutant, B153, Tn5 interrupted one of the DHBA biosynthetic genes and resulted in impaired synthesis of siderophore.
Ethidium bromide stained agarose gel showing amplified DNA fragments. a 1.3 kb fragment containing part of Tn5 and part of sidB gene of the mutant, B153. b 5 kb fragment containing four sid genes of Rhizobium BICC 651. c 3.5 kb fragment containing whole of the sidC, sidE genes, and part of the sidB gene of B153. d 1.5 kb fragment containing part of the sidB and whole of the sidA gene of B153
Nucleotide sequence analysis of the siderophore biosynthesis region
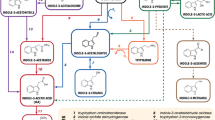
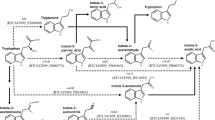
Using the agbCF and agbAR primer pair, a 5 kb DNA fragment was amplified from the genomic DNA of BICC 651 (Fig. 2b). From the fragment, 4,921 bp was sequenced using appropriate primers and the DNA sequence was submitted to GenBank under accession no. GU251056. Upon nucleotide BLAST search, the 4,921 bp DNA region of BICC 651 was found to match (90 % identity) with the agrobactin biosynthetic (agb) genes of A. tumefaciens MAFF301001 (accession no. AB083344). Nucleotide sequence analysis of the 4,921 bp DNA region of BICC 651 revealed the presence of four open reading frames (ORFs) (Fig. 3a). ORF1 (sidC) was 1,029 bp long (positions 213–1,241), potentially coding for a 342-amino-acid-residue-long protein. ORF2 (sidE), identified between positions 1,406–3,034, was 1,629 bp long and coded for a putative protein of 542 amino acid residues. At 97 bp, downstream from the TGA stop codon of ORF2 was found the ORF3 (sidB), 870 bp long, potentially coding for a 289-amino-acid-residue protein (positions 3,131–4,000). Finally, at 14 bp from the TGA stop codon of ORF3, the ATG start codon of ORF4 (sidA) (positions 4,014–4,772) was located. It was only 759 bp long, with a predicted translation product of 252-amino acid residues. The four ORFs identified in the 5 kb region were closely connected with narrow intergenic spaces indicating their polycistronic organization within an operon. In the sequence of 4,921 bp, a probable ribosome-binding site (AGGAGG) was identified six bp upstream of the ATG start codon of sidC of BICC 651 using Promoter prediction search tool (www.softberry.com). A presumable iron box (ACAAAACATGATTAGC) was also identified 56 bp upstream of the sidC start codon similar to the one found in A. tumefaciens MAFF301001 (accession no. AB083344) and in Pseudomonas fluorescens (accession no. Y09356). Presence of the potential Fur box sequence indicated iron regulation of the functions of sid genes in the strain BICC 651.
Genetic organization and genomic context of the siderophore biosynthesis DNA region of Rhizobium mutant, B153. a The physical map showing organization of sid genes, transcriptional directions of four ORFs, site of Tn5 insertion, SalI (S) restriction sites, alignment site of the primers (agbCF, agbAR and Tn5Int) and hybridization site of the Tn5 probe (marked as white). b Genomic context and organization conservation of the sidB gene region in the genome of Rhizobium BICC 651 in comparison to the genomes of related catechol-producing organisms. Arrows represent genes with their relative orientation in the genomes. Genes are coloured according to their functional categories and labelled according to their original gene annotation in the database. Orange/black (isochorismatase); red (2,3-dihydroxybenzoate-AMP-ligase); green (isochorismate synthase); sky blue/grey (2,3-dihydro-2,3-dihydroxybenzoate) and yellow (entF)
Site of Tn5 insertion
From the genomic DNA of the mutant, B153, a 3.5 kb product was obtained using agbCF and Tn5Int primers (Fig. 2c) and a 1.5 kb product was amplified using agbAR and Tn5Int primers (Fig. 2d). Matching the sequences, it was found that Tn5 was inserted next to the 210th nucleotide position from the start codon of the sidB gene in the mutant B153 (Fig. 3a).
Comparison of genetic organization of sid genes of BICC 651 with other species
The genomic context of sid DNA region in Rhizobium BICC 651, when compared to those in other catechol siderophore-producing members revealed that the organization of the region was not conserved; relative orientation and arrangement of the genes in the region of the genomes of different members varied considerably (Fig. 3b). Genetic organization of sidC, sidE, sidB, and sidA of Rhizobium BICC 651 was similar to that of entCEBA in E. coli for enterobactin biosynthesis (Crosa and Walsh 2002), in A. tumefaciens MAFF301001 for agrobactin (Sonoda et al. 2002), and siderophore biosynthetic genes in Brucella melitensis, Chromobacterium violaceum, and Klebsiella pneumoniae. In Aeromonas hydrophila, entA was missing immediately after entB, whereas in Erwinia carotovora, entF was present in between entB and entA. The four biosynthesis genes were arranged as dhbACEB cluster in Bacillus subtilis (Rowland et al. 1996) as well as in Acinetobactor baumannii (Dorsey et al. 2003). In Vibrio cholerae for vibriobactin biosynthesis, these were organised as vibACEB cluster in which vibC and vibE were co-transcribed, while vibA and vibB belonged to two independent transcriptional units (Wyckoff et al. 1997). Since organization of catechol siderophore genes of any Rhizobium spp. was not found in the database, gene context analysis was carried out from available data of catechol siderophore-producing organisms.
Deduced functions of sid gene products
Comparison of the derivative amino acid sequences of the genes with those of related organisms was made by BLASTX analysis (Table 5). The derivative amino acid sequence of ORF1 (sidC) of Rhizobium BICC 651 showed highest similarity (92 %) to isochorismate synthase of Agrobacterium tumefaciens (accession no. BAC16757). The enzyme converts chorismate to isochorismate. The derivative amino acid sequence of ORF2 (sidE) showed highest homology (89 %) to that of 2,3-dihydroxybenzoate-AMP-ligase of A. tumefaciens (BAC16758). The enzyme activates 2,3-dihydroxybenzoate by forming a (2,3-dihydroxybenzoyl) adenylate-enzyme complex (Rusnak et al. 1989). Consequently, the deduced gene product of ORF3 (sidB) showed highest similarity (97 %) with 2,3-dihydro-2,3-dihydroxybenzoate synthetase (isochorismatase) of A. tumefaciens (BAC16759). The enzyme removes the enolpyruvyl side chain of isochorismate (Rusnak et al. 1990). The ORF4 (sidA) translational product showed 90 % homology with 2,3-dihydro-2,3-dihydroxybenzoate dehydrogenase of A. tumefaciens (BAC16760). The enzyme catalyzes the NAD-coupled oxidation of 2,3-dihydro-2,3-dihydroxybenzoate (Liu et al. 1989). The deduced functions of the derivative amino acid sequence of sid gene products are presented in the Fig. 4.
Phylogenetic analysis
On the basis of the amino acid sequence identities data given in Table 5, phylogenetic tree of each of the four sid translational products was constructed using neighbor-joining method (Fig. 5). In all the trees, the strain BICC 651 was placed in the same evolutionary branch as of Agrobacterium tumefaciens. This result along with 16S Rdna sequence analysis of the strain BICC 651 (accession no. DQ839132) demonstrates that the strain BICC 651 has a chromosomal background similar to that of A. tumefaciens. Since the strain BICC 651 was isolated from a nodule on the root of a legume and genes responsible for symbiosis including nodulation are located on Sym plasmids of rhizobia it might be possible that the strain BICC 651 had its ancestry in Agrobacterium but through horizontal gene transfer acquired the Sym plasmid from some hitherto unknown source. A somewhat similar observation was made with a strain T1K, reported to be Rhizobium trifolii (R. leguminosarum biovar trifolii), which nodulated red and white clover and shared many physiological properties with Agrobacteriumtumefaciens. The strain was more closely related to Agrobacterium than to R.trifolii (Skotnicki and Rolfe 1978). Based on the gene sequence similarity, it is concluded that the stain BICC 651 might acquire the siderophore gene cluster from other bacteria via horizontal gene transfer during evolutionary process as suggested by Sonoda et al. (2002) who proposed that Agrobacterium tumefaciens MAFF301001 had obtained the agb gene cluster from Brucella melitensis, Pseudomonas fluorescens, Escherichia coli or Salmonella typhimurium in the same process.
Phylogenetic trees based on comparison of the translational products of the sid genes. Trees were constructed by using the neighbour-joining method. Horizontal branch lengths are proportional to the estimated number of nucleotide substitutions. a SidC and its homologues, b SidE and its homologues, c SidB and its homologues, d SidA and its homologues
Nucleotide sequence analysis revealed that the genes, sidC, sidE, sidB, sidA constitute an operon. Disruption of sidB by Tn5 insertion results in poor growth under iron-limiting condition, absence of siderophore production and production of less number of nodules by the Sid− mutant, B153 as compared to the wild type strain. The data indicate that the siderophore biosynthetic genes are essential for growth and production of catechol siderophore of the Rhizobium BICC 651, which in turn influences its symbiotic efficiency.
References
Benson HP, Boncompagni E, Guerinot ML (2005) An iron uptake operon required for proper nodule development in the Bradyrhizobium japonicum-soybean symbiosis. Mol Plant Microbe Interact 18:950–959
Beringer JE (1974) R factor transfer in Rhizobium leguminosarum. J Gen Microbiol 84:188–198
Berraho E, Lesuere D, Diem HG, Sasson A (1997) Iron requirement and siderophore production in Rhizobium ciceri during growth on an iron-deficient medium. World J Microbiol Biotechnol 13:501–510
Carter RA, Worsley PS, Sawers G, Challis GL, Dilworth MJ, Carson KC et al (2002) The vbs genes that direct synthesis of the siderophore vicibactin in Rhizobium leguminosarum: their expression in other genera requires ECF sigma factor RpoI. Mol Microbiol 44(5):1153–1166
Ciria R, Abreu-Goodger C, Morett E, Merino E (2004) GeConT: gene context analysis. Bioinformatics 14:2307–2308
Crosa JH, Walsh TC (2002) Genetics and assembly line enzymology of siderophore biosynthesis in bacteria. Microbiol Mol Biol Rev 66:223–249
Dorsey CW, Tolmasky ME, Crosa JH, Actis LA (2003) Genetic organization of an Acinetobacter baumannii chromosomal region harbouring genes related to siderophore biosynthesis and transport. Microbiology 149:1227–1238
Earhart CF (1996) Uptake and metabolism of iron and molybdenum. In: Neidhardt FC et al. (eds) Escherichia coli and Salmonella: Cellular and Molecular Biology. American Society for Microbiology, Washington, DC, pp 1075–1090
Gill PR, Barton LL, Scoble MD, Neilands JB (1991) A high affinity iron transport system of Rhizobium meliloti may be required for efficient nitrogen fixation in planta. In: Chen Y, Hadar Y (eds) Iron nutrition and interactions in plants. Kluwer Academic Publishers, Netherlands, pp 251–257
Guerinot ML (1991) Iron uptake and metabolism in the rhizobia/legume symbioses. Plant Soil 130:199–209
Huang G, Zhang L, Birch RG (2000) Rapid amplification and cloning of Tn5 flanking fragments by inverse PCR. Lett Appl Microbiol 31:149–153
Joshi FR, Chaudhari A, Joglekar P, Archana G, Desai AJ (2008) Effect of expression of Bradyrhizobium japonicum 61A152 fegA gene in Mesorhizobium sp., on its competitive survival and nodule occupancy on Arachis hypogea. Applied Soil Ecology 40:338–347
Joshi FR, Desai DK, Archana G, Desai AJ (2009) Enhanced survival and nodule occupancy of pigeon pea nodulating Rhizobium sp. ST1 expressing fegA gene of Bradyrhizobium japonicum 61A152. J Biol Sci 9:40–51
Lankford CE (1973) Bacterial assimilation of iron. Crit Rev Microbiol 2:273–331
Lesueur D, Carr Del Rio M, Dien HG (1995) Modification of the growth and the competitiveness of Bradyrhizobium strain obtained through affecting its siderophore-producing ability. In: Abadia J (ed) Iron nutrition in soil and plants. Kluwer Academic Publishers, PaysBas, pp 59–66
Liu J, Duncan K, Walsh C (1989) Nucleotide sequence of a cluster of Escherichia coli enterobactin biosynthesis genes: identification of entA and purification of its product 2,3-dihydro-2,3-dihydroxybenzoate dehydrogenase. J Bacteriol 171:791–798
Lynch D, O’Brien J, Welch T, Clarke P, Cuiv P, Crosa JH, O’Connell M (2001) Genetic organization of the region encoding regulation biosynthesis and transport of rhizobactin 1021, a siderophore produced by Sinorhizobium meliloti. J Bacteriol 183:2576–2585
Manjanatha MG, Loynachan TE, Atherly AG (1992) Tn5 mutagenesis of Chinese Rhizobium fredii for siderophore over production. Soil Biol Biochem 24:151–155
Meyer JM, Abdallah MA (1978) The florescent pigment of Pseudomonas fluorescens biosynthesis, purification and physical-chemical properties. J Gen Microbiol 107:319–328
Modi M, Shah KS, Modi VV (1985) Isolation and characterization of catechol-like siderophore from cowpea Rhizobium RA-1. Arch Microbiol 141:156–158
Mukhopadhyaya P, Deb C, Lahiri C, Roy P (2000) A soxA gene, encoding a diheme cytochrome c, and a sox locus, essential for sulfur oxidation in a new sulfur lithotrophic bacterium. J Bacteriol 182:4278–4287
Nambiar PTC, Sivaramakrishnan S (1987) Detection and assay of siderophores in Cowpea Rhizobia (Bradyrhizobium) using radioactive Fe (59Fe). Appl Microbiol Lett 4:37–40
Norris DO, Date RA (1976) Legume bacteriology. In: Shaw NH, Bryan WW (eds) Tropical Pasture Research – Principles and Methods, Commonwealth Bureau of Pastures and Field Crops Bulletin no. 51. Commonwealth Agricultural Bureau Publishers: Slough, England, pp 134–174
O’Hara GW, Hartzook A, Bell RW, Loneragan JF (1988) Response to Bradyrhizobium strain of peanut cultivars grown under iron stress. J Plant Nutr 11:6–11
Rowland BM, Grossman TH, Osburne MS, Taber HW (1996) Sequence and genetic organization of a Bacillus subtilis operon encoding 2,3-dihydroxybenzoate biosynthetic enzymes. Gene 178:119–123
Roy N, Bhattacharyya P, Chakrabartty PK (1994) Iron acquisition during growth in an iron-deficient medium by Rhizobium sp. isolated from Cicer arietinum. Microbiology 140:2811–2820
Rusnak F, Faraci W, Walsh C (1989) Subcloning, expression, and purification of the enterobactin biosynthetic enzyme 2,3-dihydroxybenzoate-AMP ligase: demonstration of enzyme-bound (2,3-dihydroxybenzoyl)adenylate product. Biochemistry 28:6827–6835
Rusnak F, Liu J, Quinn N, Berchtold G, Walsh C (1990) Subcloning of the enterobactin biosynthetic gene entB: expression, purification, characterization, and substrate specificity of isochorismatase. Biochemistry 29:1425–1435
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Press, New York
Schwyn B, Neiland JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56
Simon R (1984) High frequency mobilization of gram-negative bacterial replicons by the in vitro constructed Tn5-Mob transposon. Mol General Genet 196:413–420
Skorupska A, Derylo M, Lorkiewiez Z (1989) Siderophore production and utilization by Rhizobium trifolii. Biol Met 2:45–49
Skotnicki ML, Rolfe BG (1978) Interaction between the fumarate reductase system of Escherichia coli and the nitrogen fixation genes of Klebsiella pneumoniae. J Bacteriol 133:518–526
Sonoda H, Suzuki K, Yoshida K (2002) Gene cluster for ferric iron uptake in Agrobacterium tumefaciens MAFF301001. Genes Genet Syst 77:137–146
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Wyckoff EE, Stoebner JA, Reed KE, Payne SM (1997) Cloning of a Vibrio cholerae vibriobactin gene cluster: identification of genes required for early steps in siderophore biosynthesis. J Bacteriol 179:7055–7062
Zuckerkandl E, Pauling L (1965) Evolutionary divergence and convergence in proteins. In: Bryson V, Vogel HJ (eds) Evolving genes and proteins. Academic Press, New York, pp 97–166
Acknowledgments
We thank late Dr. Pradosh Roy and Prof. Sujoy Kumar Dasgupta, Department of Microbiology, Bose Institute, Kolkata for their active support. The work was funded by Department of Biotechnology, India and a fellowship (to B. Datta) was provided by the University Grant Commission, India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under license to BioMed Central Ltd. Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Datta, B., Chakrabartty, P.K. Siderophore biosynthesis genes of Rhizobium sp. isolated from Cicer arietinum L.. 3 Biotech 4, 391–401 (2014). https://doi.org/10.1007/s13205-013-0164-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13205-013-0164-y