Abstract
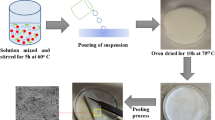
Current study deals with an agriculture waste paddy straw (PS) incorporated with chitosan (CS) to prepare photocatalytic paddy straw chitosan nanocomposite (PS-CSNC) with cross-linking agent sodium tripolyphosphate was investigated and catalysis-based photocatalytic adsorptions of Rhodamine B (RhB) and Malachite green (MG) in aqueous solution under UV irradiation were also examined. Moreover, this study examined that the factors influence the adsorption process, such as catalyst concentration, pH and temperature, using kinetic models. Additionally, the composite characterization was achieved by several analysis, such as UV–visible spectroscopy, Fourier-transform infrared spectroscopy, X-ray diffraction analysis, thermogravimetry/differential thermal analysis and field effect scanning electron microscopy. The study shown that the optimized concentration of PS-CSNC catalyst (3.0 g L−1), below neutral pH (6.5) of the solution and temperature 60 °C achieved excellent removal of both dyes. Initial sorption process involved the removal mechanism of both RhB and MG followed by photocatalytic adsorption. The process of adsorption was well adapted with pseudo second-order kinetics. Overall results suggested that chitosan-based agriculture waste paddy straw nanocomposite could be an eco-friendly and cost-effective bio-adsorbent for removal of RhB and MG in wastewater treatment.
















Similar content being viewed by others
Data Availability Statement
Not applicable.
References
AbdElhady MM (2012) Preparation and characterization of chitosan/zinc oxide nanoparticles for imparting antimicrobial and UV protection to cotton fabric. Int J Carbohydr Chem 2012:1–6. https://doi.org/10.1155/2012/840591
Ado A, Tukur AI, Ladan M, Gumel SM, Muhammad AA, Habibu S, Koki IB (2015) A review on industrial effluents as major sources of water pollution in a review on industrial effluents as major sources of water pollution in Nigeria. Chem J 1(5):159–164. http://www.aiscience.org/journal/cj. http://creativecommons.org/licenses/by-nc/4.0/
Ahmed W, Iqbal J (2022) Mn doped ZrO2 nanoparticles: an optically tuned photocatalyst with superior structural, magnetic and dielectric characteristics. J Phys Chem Solids 160:110285
Akar ST, Yilmazer D, Celik S, Balk YY, Akar T (2013) On the utilization of a lignocellulosic waste as an excellent dye remover: modification, characterization and mechanism analysis. Chem Eng J 229:257–266. https://doi.org/10.1016/j.cej.2013.06.009
Alakhras I, Ouachtak F, Alhajri H, Rehman E, Al-Mazaideh R, Anastopoulos G, Lima EC (2021) Adsorptive removal of cationic rhodamine B dye from aqueous solutions using chitosan-derived schiff base. Sep Sci Technol (philadelphia). https://doi.org/10.1080/01496395.2021.1931326
Ali I, Burakova I, Galunin E, Burakov A, Mkrtchyan E, Melezhik A, Kurnosov D, Tkachev A, Grachev V (2019) High-speed and high-capacity removal of methyl orange and malachite green in water using newly developed mesoporous carbon: kinetic and isotherm studies. ACS Omega 4(21):19293–19306. https://doi.org/10.1021/acsomega.9b02669
Alshabanat MN, Al-Anazy MM (2018) An experimental study of photocatalytic degradation of Congo red using polymer nanocomposite films. J Chem. https://doi.org/10.1155/2018/9651850
Anitha A, Deepa N, Chennazhi KP, Nair SV, Tamura H, Jayakumar R (2011) Development of mucoadhesive thiolated chitosan nanoparticles for biomedical applications. Carbohydr Polym 83(1):66–73. https://doi.org/10.1016/j.carbpol.2010.07.028
Ariff SAY, Yusoff K, Masarudin MJ (2017) Encapsulation of miRNA in chitosan nanoparticles as a candidate for an anti-metastatic agent in cancer therapy. Malays Appl Biol 46(1):165–170
Arikal D, Kallingal A (2019) Photocatalytic degradation of azo and anthraquinone dye using TiO2/MgO nanocomposite immobilized chitosan hydrogels. Environ Technol (united Kingdom). https://doi.org/10.1080/09593330.2019.1701094
Arumugam TK, Krishnamoorthy P, Rajagopalan NR, Nanthini S, Vasudevan D (2019) Removal of malachite green from aqueous solutions using a modified chitosan composite. Int J Biol Macromol 128:655–664. https://doi.org/10.1016/j.ijbiomac.2019.01.185
Aslam Z, Qaiser M, Ali R, Abbas A, Ihsanullah, Zarin S (2019) Al2O3/MnO2/CNTs nanocomposite: synthesis, characterization and phenol adsorption. Fuller Nanotubes Carbon Nanostruct 27(7):591–600. https://doi.org/10.1080/1536383X.2019.1622528
Banik N, Hussain A, Ramteke A, Sharma HK, Maji TK (2012) Preparation and evaluation of the effect of particle size on the properties of chitosan-montmorillonite nanoparticles loaded with isoniazid. RSC Adv 2(28):10519–10528. https://doi.org/10.1039/c2ra20702h
Chanzu HA, Onyari JM, Shiundu PM (2012) Biosorption of malachite green from aqueous solutions onto polylactide/spent brewery grains films: kinetic and equilibrium studies. J Polym Environ 20(3):665–672. https://doi.org/10.1007/s10924-012-0479-5
Chiou MS, Li HY (2003) Adsorption behavior of reactive dye in aqueous solution on chemical cross-linked chitosan beads. Chemosphere 50(8):1095–1105. https://doi.org/10.1016/S0045-6535(02)00636-7
Chong MN, Jin B, Chow CW, Saint C (2010) Recent developments in photocatalyticwater treatment technology: a review. Water Res 44:2997
Chowdhury S, Saha P (2010) Sea shell powder as a new adsorbent to remove basic green 4 (malachite green) from aqueous solutions: equilibrium, kinetic and thermodynamic studies. Chem Eng J 164(1):168–177. https://doi.org/10.1016/j.cej.2010.08.050
da Silva Alves DC, Healy B, Pinto LA, Cadaval TR, Breslin CB (2021) Recent developments in chitosan-based adsorbents for the removal of pollutants from aqueous environments. Molecules 26(3):594. https://doi.org/10.3390/molecules26030594
Daifullah AAM, Yakout SM, Elreefy SA (2007) Adsorption of fluoride in aqueous solutions using KMnO4-modified activated carbon derived from steam pyrolysis of rice straw. J Hazard Mater 147(1–2):633–643. https://doi.org/10.1016/j.jhazmat.2007.01.062
El Haddad M, Mamouni R, Saffaj N, Lazar S (2012) Removal of a cationic dye—basic red 12—from aqueous solution by adsorption onto animal bone meal. J Assoc Arab Univ Basic Appl Sci 12(1):48–54. https://doi.org/10.1016/j.jaubas.2012.04.003
Fukuoka E, Makita M, Yamamura S (1993) Pattern fitting procedure for the characterization of crystals and/or crystallites in tablets. Chem Pharm Bull 41(12):2166–2171. https://doi.org/10.1248/cpb.41.2166
Ghorai S, Sarkar A, Raoufi M, Panda AB, Schönherr H, Pal S (2014) Enhanced removal of methylene blue and methyl violet dyes from aqueous solution using a nanocomposite of hydrolyzed polyacrylamide grafted xanthan gum and incorporated nanosilica. ACS Appl Mater Interfaces 6(7):4766–4777. https://doi.org/10.1021/am4055657
Gokila S, Gomathi T, Sudha PN, Anil S (2017) Removal of the heavy metal ion chromiuim(VI) using chitosan and alginate nanocomposites. Int J Biol Macromol 104:1459–1468. https://doi.org/10.1016/j.ijbiomac.2017.05.117
Hasmath Farzana M, Meenakshi S (2014) Synergistic effect of chitosan and titanium dioxide on the removal of toxic dyes by the photodegradation technique. Ind Eng Chem Res 53(1):55–63. https://doi.org/10.1021/ie402347g
Hassaan MA, Nemr AE (2017) Advanced oxidation processes (AOPs) for wastewater treatment advanced oxidation processes of some organic pollutants in fresh and sea water. Am J Environ Sci Eng 1(3):64–67. http://www.sciencepublishinggroup.com/j/ajese
Hassan SED, Boon E, St-Arnaud M, Hijri M (2011) Molecular biodiversity of arbuscular mycorrhizal fungi in trace metal-polluted soils. Mol Ecol 20(16):3469–3483. https://doi.org/10.1111/j.1365-294X.2011.05142.x
Hassan ML, Fadel SM, El-Wakil NA, Oksman K (2012) Chitosan/rice straw nanofibers nanocomposites: preparation, mechanical, and dynamic thermomechanical properties. J Appl Polym Sci 125(S2):E216–E222. https://doi.org/10.1002/app.36606
Hassan UA, Hussein MZ, Alitheen NB, Ariff SAY, Masarudin MJ (2018) In vitro cellular localization and efficient accumulation of fluorescently tagged biomaterials from monodispersed chitosan nanoparticles for elucidation of controlled release pathways for drug delivery systems. Int J Nanomed 13:5075–5095. https://doi.org/10.2147/IJN.S164843
Ho YS (2004) Citation review of Lagergren kinetic rate equation on adsorption reactions. Scientometrics 59(1):171–177. https://doi.org/10.1023/B:SCIE.0000013305.99473.cf
Hu A, Liang R, Zhang X, Kurdi S, Luong D, Huang H, Peng P, Marzbanrad E, Oakes KD, Zhou Y, Servos MR (2013) Enhanced photocatalytic degradation of dyes by TiO2 nanobelts with hierarchical structures. J Photochem Photobiol A 256:7–15. https://doi.org/10.1016/j.jphotochem.2013.01.015
Inyinbor AA, Adekola FA, Olatunji GA (2017) Liquid phase adsorptions of rhodamine B dye onto raw and chitosan supported mesoporous adsorbents: isotherms and kinetics studies. Appl Water Sci 7(5):2297–2307. https://doi.org/10.1007/s13201-016-0405-4
Li J, Ma Q, Shao H, Zhou X, Xia H, Xie J (2017) Biosynthesis, characterization, and antibacterial activity of silver nanoparticles produced from rice straw biomass. BioResources 12(3):4897–4911. https://doi.org/10.15376/biores.12.3.4897-4911
Lin C, Ritter JA, Popov BN (1999) Correlation of double-layer capacitance with the pore structure of sol-gel derived carbon xerogels. J Electrochem Soc 146(10):3639–3643. https://doi.org/10.1149/1.1392526
Liu X, Tian J, Li Y, Sun N, Mi S, Xie Y, Chen Z (2019) Enhanced dyes adsorption from wastewater via Fe3O4 nanoparticles functionalized activated carbon. J Hazard Mater 373:397–407. https://doi.org/10.1016/j.jhazmat.2019.03.103
López Arbeloa I, Ruiz Ojeda P (1982) Dimeric states of rhodamine B. Chem Phys Lett 87(6):556–560. https://doi.org/10.1016/0009-2614(82)83177-1
Nga NK, Thuy Chau NT, Viet PH (2020) Preparation and characterization of a chitosan/MgO composite for the effective removal of reactive blue 19 dye from aqueous solution. J Sci Adv Mater Devices 5(1):65–72. https://doi.org/10.1016/J.JSAMD.2020.01.009
Qasem NAA, Mohammed RH, Lawal DU (2021) Removal of heavy metal ions from wastewater: a comprehensive and critical review. NPJ Clean Water 4(1):1–15
Qiu B, Xu XF, Deng RH, Xia GQ, Shang XF, Zhou PH (2019) Construction of chitosan/ZnO nanocomposite film by in situ precipitation. Int J Biol Macromol 122:82–87. https://doi.org/10.1016/j.ijbiomac.2018.10.084
Rana VK, Pandey AK, Singh RP, Kumar B, Mishra S, Ha CS (2010) Enhancement of thermal stability and phase relaxation behavior of chitosan dissolved in aqueous l-lactic acid: using “silver nanoparticles” as nano filler. Macromol Res 18(8):713–720. https://doi.org/10.1007/s13233-010-0801-9
Ray AK, Beenackers AACM (1998) Novel photocatalytic reactor for water purification. AIChE J 44(2):477–483. https://doi.org/10.1002/aic.690440224
Ruiz HA, Cerqueira MA, Silva HD, Rodríguez-Jasso RM, Vicente AA, Teixeira JA (2013) Biorefinery valorization of autohydrolysis wheat straw hemicellulose to be applied in a polymer-blend film. Carbohydr Polym 92(2):2154–2162. https://doi.org/10.1016/j.carbpol.2012.11.054
Sadiq AC, Rahim NY, Suah FBM (2020) Adsorption and desorption of malachite green by using chitosan-deep eutectic solvents beads. Int J Biol Macromol 164:3965–3973. https://doi.org/10.1016/j.ijbiomac.2020.09.029
Samuel MS, Suman S, Venkateshkannan, Selvarajan E, Mathimani T, Pugazhendhi A (2020) Immobilization of Cu3(btc)2 on graphene oxide–chitosan hybrid composite for the adsorption and photocatalytic degradation of methylene blue. J Photochem Photobiol B Biol. https://doi.org/10.1016/j.jphotobiol.2020.111809
Sathiyavimal S, Vasantharaj S, Kaliannan T, Pugazhendhi A (2020) Eco-biocompatibility of chitosan coated biosynthesized copper oxide nanocomposite for enhanced industrial (azo) dye removal from aqueous solution and antibacterial properties. Carbohydr Polym 241:116243. https://doi.org/10.1016/j.carbpol.2020.116243
Scialla S, Barca A, Palazzo B, D’Amora U, Russo T, Gloria A, De Santis R, Verri T, Sannino A, Ambrosio L, Gervaso F (2019) Bioactive chitosan-based scaffolds with improved properties induced by dextran-grafted nano-maghemite and l-arginine amino acid. J Biomed Mater Res Part A 107(6):1244–1252. https://doi.org/10.1002/jbm.a.36633
Sharifianjazi F, Esmaeilkhanian A, Moradi M, Pakseresht A, Asl MS, Karimi-Maleh H, Jang HW, Shokouhimehr M, Varma RS (2021) Biocompatibility and mechanical properties of pigeon bone waste extracted natural nano-hydroxyapatite for bone tissue engineering. Mater Sci Eng B Solid-State Mater Adv Technol 264:114950. https://doi.org/10.1016/j.mseb.2020.114950
Sharma SK (2015) Green chemistry for dyes removal from waste water: research trends and applications. Green chemistry for dyes removal from waste water: research trends and applications. Wiley, New York
Shrestha R, Ban S, Devkota S, Sharma S, Joshi R, Tiwari AP, Kim HY, Joshi MK (2021) Technological trends in heavy metals removal from industrial wastewater: a review. J Environ Chem Eng 9(4). https://doi.org/10.1016/j.jece.2021.105688
Slimani R, El Ouahabi I, Abidi F, El Haddad M, Regti A, Laamari MR, Antri SE, Lazar S (2014) Calcined eggshells as a new biosorbent to remove basic dye from aqueous solutions: thermodynamics, kinetics, isotherms and error analysis. J Taiwan Inst Chem Eng 45(4):1578–1587. https://doi.org/10.1016/j.jtice.2013.10.009
Su JF, Yuan XY, Huang Z, Wang XY, Lu XZ, Zhang LD, Wang SB (2012) Physicochemical properties of soy protein isolate/carboxymethyl cellulose blend films crosslinked by Maillard reactions: Color, transparency and heat-sealing ability. Mater Sci Eng C 32(1):40–46. https://doi.org/10.1016/j.msec.2011.09.009
Subramanian V, Kamat PV, Wolf EE (2003) Mass-transfer and kinetic studies during the photocatalytic degradation of an azo dye on optically transparent electrode thin film. Ind Eng Chem Res 42(10):2131–2138. https://doi.org/10.1021/ie020636u
Suneetha RB (2018) Spectral, thermal and morphological characterization of biodegradable graphene oxide-chitosan nanocomposites. J Nanosci Technol 04(02):342–344. https://doi.org/10.30799/jnst.sp208.18040201
Szeto C-C, Li PK-T, Johnson DW, Bernardini J, Dong J, Figueiredo AE, Ito Y, Kazancioglu R, Moraes T, Van Esch S (2017) ISPD catheter-related infection recommendations: 2017 update. Perit Dial Int 37(2):141–154
Unnikrishnan L, Mohanty S, Nayak SK, Ali A (2011) Preparation and characterization of poly(methyl methacrylate)-clay nanocomposites via melt intercalation: effect of organoclay on thermal, mechanical and flammability properties. Mater Sci Eng A 528(12):3943–3951. https://doi.org/10.1016/j.msea.2011.01.071
Wan Ngah WS, Teong LC, Hanafiah MAKM (2011) Adsorption of dyes and heavy metal ions by chitosan composites: a review. Carbohydr Polym 83(4):1446–1456. https://doi.org/10.1016/j.carbpol.2010.11.004
Wu FC, Tseng RL, Juang RS (2010) A review and experimental verification of using chitosan and its derivatives as adsorbents for selected heavy metals. J Environ Manag 91(4):798–806. https://doi.org/10.1016/j.jenvman.2009.10.018
Xu Z, Zhang Q, Li X, Huang X (2021) A critical review on chemical analysis of heavy metal complexes in water/wastewater and the mechanism of treatment methods. Chem Eng J 429:131688. https://doi.org/10.1016/j.cej.2021.131688
Zeng L, Chen Y, Zhang Q, Guo X, Peng Y, Xiao H, Chen X, Luo J (2015) Adsorption of Cd(II), Cu(II) and Ni(II) ions by cross-linking chitosan/rectorite nano-hybrid composite microspheres. Carbohydr Polym 130:333–343. https://doi.org/10.1016/j.carbpol.2015.05.015
Funding
There is no funding support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sakthivel, S., Periakaruppan, R., Vallinayagam, S. et al. Synthesis and characterization of paddy straw chitosan nanocomposite as an efficient photocatalytic bio-adsorbent for the removal of rhodamine B and malachite green dye from aqueous solution. Appl Nanosci 13, 2555–2569 (2023). https://doi.org/10.1007/s13204-021-02141-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-021-02141-7