Abstract
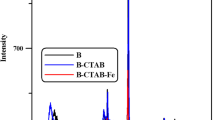
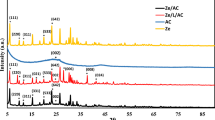
Clays are widely accepted for dye removal from wastewater. Here, in this work, nanobentonite was synthesized by a simple, rapid, and cost-effective chemithermal method. Additionally, nanobentonite was coupled with hematite nanoparticles via thermal coupling to prepare a nanocomposite with enhanced surface absorption. Surface morphology, compositional and structural analysis of synthesized samples were done using various characterization techniques. Results suggested that nanobentonite and nanocomposite having an average diameter of 35.36 and 21.85 nm respectively were synthesized. Nanocomposite exhibited increased surface area. Additionally, the ability of all samples as an adsorbent for removal of methylene blue (MB) and Congo red (CR), from an aqueous solution was investigated under various optimization conditions. Adsorption kinetics revealed that Pseudo II order equation fits well for the adsorption process following intraparticular diffusion mechanism for both the dyes. The adsorption equilibrium data of MB and CR was fitted well by the Langmuir and Freundlich adsorption isotherm model, respectively. The thermodynamic parameters suggested adsorption process is endothermic and spontaneous in nature with increased entropy value.






Similar content being viewed by others
References
Al-Essa K (2018) Activation of Jordanian bentonite by hydrochloric acid and its potential for olive mill wastewater enhanced treatment Khansaa. J Chem. https://doi.org/10.1155/2018/8385692
Alexander JA, Zaini MAA, Surajudeen A, Aliyu ENU, Omeiza AU (2019) Surface modification of low-cost bentonite adsorbents—a review. Part Sci Technol 37:538–549. https://doi.org/10.1080/02726351.2018.1438548
Almeida CAP, Debacher NA, Downs AJ, Cottet L, Mello CAD (2009) Removal of methylene blue from colored effluents by adsorption on montmorillonite clay. J Colloid Interface Sci 332:46–53
Beall GW (2003) The use of organo-clays in water treatment. Appl Clay Sci 24:11–20. https://doi.org/10.1016/j.clay.2003.07.006
Belachew N, Bekele G (2019) Synergy of magnetite intercalated bentonite for enhanced adsorption of congo red dye. SILICON 12:603–612. https://doi.org/10.1007/s12633-019-00152-2
Bukit N, Frida E, Harahap MH (2013) Preparation natural bentonite in nano particle material as filler nanocomposite high density poliethylene (Hdpe). Chem Mater Res 3:13. www.iiste.org
Bulut Y, Karaer H (2015) Adsorption of methylene blue from aqueous solution by crosslinked chitosan/bentonite composite. J Dispers Sci Technol 36:61–67. https://doi.org/10.1080/01932691.2014.888004
Chen AH, Huang YY (2010) Adsorption of remazol black 5 from aqueous solution by the templated crosslinked-chitosans. J Hazard Mater 177:668–675. https://doi.org/10.1016/j.jhazmat.2009.12.083
Darvishi Z, Morsali A (2011a) Synthesis and characterization of nano-bentonite by sonochemical method. Ultrason Sonochem 18:238–242
Darvishi Z, Morsali A (2011b) Synthesis and characterization of nano-bentonite by solvothermal method. Colloids Surf A Physicochem Eng Asp 377:15–19. https://doi.org/10.1016/j.colsurfa.2010.11.016
de Oliveira CIR, Rocha MCG, da Silva ALN, Bertolino LC (2016) Characterization of bentonite clays from Cubati, Paraíba (Northeast of Brazil). Cerâmica 62:272–277. https://doi.org/10.1590/0366-69132016623631970
Dhanasekaran V, Anandhavelu S, Polychroniadis EK, Mahalingam T (2014) Microstructural properties evaluation of Fe2O3 nanostructures. Mater Lett 126:288–290
Fu H, Quan X, Liu Z, Chen S (2004) Photoinduced transformation of gamma-HCH in the presence of dissolved organic matter and enhanced photoreactive activity of humate-coated alpha-Fe2O3. Langmuir 20(12):4867–4873. https://doi.org/10.1021/la0364486 (PMID: 15984243)
Ghosh D, Bhattacharyya KG (2002) Adsorption of methylene blue on kaolinite. Appl Clay Sci 20:295–300
Hashem FS (2013) Removal of methylene blue by magnetite-covered bentonite nanoparticles. Eur Chem Bull 2:524–529
Huang XY, Bu HT, Jiang GB, Zeng MH (2011) Cross-linked succinyl chitosan as an adsorbent for the removal of methylene blue from aqueous solution. Int J Biol Macromol 49:643–651. https://doi.org/10.1016/j.ijbiomac.2011.06.023
İȿlek C, Sinağ A, Akata I (2008) Investigation of biosorption behavior of methylene blue on Pleurotus ostreatus (Jacq.) P. Kumm. Clean 36:387–392. https://doi.org/10.1002/clen.200700167
Ismaeel SH, Mabrouk MS, Ali AAA, Abn-Elwalead K (2017) Synthesis and characterization of bentonite nanocomposites from Egyptian bentonite clay. Int J Nanotechnol Allied Sci 1:16–29
Landoulsi O, Megriche A, Calvet R, Espitalier F, Ferreira JMF, Mgaidi A (2013) Effects of heating and acid activation on the structure and surface properties of a kaolinite-illite-smectite clayey mixture. Open Miner Process J 6:13–20. https://doi.org/10.2174/1874841401306010013
Liu Y, Zheng Y, Wang A (2010) Enhanced adsorption of methylene blue from aqueous solution by chitosan-g-poly (acrylic acid)/vermiculite hydrogel composites. J Environ Sci 22:486–493. https://doi.org/10.1016/S1001-0742(09)60134-0
Lubis S, Sheilatina S, Sitompul DW (2019) Photocatalytic degradation of indigo carmine dye using α-Fe2O3/bentonite nanocomposite prepared by mechanochemical synthesis. IOP Conf Ser Mater Sci Eng 509:012142. https://doi.org/10.1088/1757-899X/509/1/012142
Mishra A, Mehta A, Sharma M, Basu S (2017) Enhanced heterogeneous photodegradation of VOC and dye using microwave synthesized TiO2/clay nanocomposites: a comparison study of different type of clays. J Alloys Compd 694:574–580. https://doi.org/10.1016/j.jallcom.2016.10.036
Motshekga SC, Ray SS, Onyango MS, Momba MNB (2013) Microwave-assisted synthesis, characterization and antibacterial activity of Ag/ZnO nanoparticles supported bentonite clay. J Hazard Mater 262:439–446. https://doi.org/10.1016/j.jhazmat.2013.08.074
Neaman A, Pelletier M, Villieras F (2003) The effects of exchanged cation, compression, heating and hydration on textural properties of bulk bentonite and its corresponding purified montmorillonite. Appl Clay Sci 22:153–168. https://doi.org/10.1016/S0169-1317(02)00146-1
Ngah WSW, Teong LC, Hanafiah MAKM (2011) Adsorption of dyes and heavy metal ions by chitosan composites : a review. Carbohydr Polym 83:1446–1456. https://doi.org/10.1016/j.carbpol.2010.11.004
Patil SP, Bethi B, Sonawane GH, Shrivastava VS, Sonawane S (2016) Efficient adsorption and photocatalytic degradation of Rhodamine B dye over Bi2O3 -bentonite nanocomposites : a kinetic study. J Ind Eng Chem 34:356–363
Raffatulah M, Sulaiman O, Hashim R, Ahmad A (2010) Adsorption of methylene blue on low-cost adsorbents: a review. J Hazard Mater 177:70–80. https://doi.org/10.1016/j.jhazmat.2009.12.047
Salah BA, Gaber MS, Kandil AHT (2019) The removal of uranium and thorium from their aqueous solutions by 8-hydroxyquinoline immobilized bentonite. Minerals 9:626. https://doi.org/10.3390/min9100626
Santhi T, Manonmani S (2011) Malachite green removal from aqueous solution by the peel of Cucumis sativa fruit. Clean 39:162–170. https://doi.org/10.1002/clen.201000077
Sharma P, Dhiman S, Kumari S, Rawat P, Srivastava C, Sato H, Akitsu T, Kumar S, Hassan I, Majumder S (2019) Revisiting the physiochemical properties of hematite ( α -Fe2O3) nanoparticle and exploring its bio-environmental application. Mater Res Express 6:095072. https://doi.org/10.1088/2053-1591/ab30ef
Sharma P, Kumari S, Ghosh D, Yadav V, Vij A, Rawat P, Kumar S, Sinha C, Saini S, Sharma V, Hassan MI, Srivastava C, Majumder S (2021) Capping agent-induced variation of physicochemical and biological properties of α-Fe2O3 nanoparticles. Mater Chem Phys 258:123899. https://doi.org/10.1016/j.matchemphys.2020.123899
Sonawane GH, Patil SP, Shrivastava VS (2017) Photocatalytic degradation of safranine by ZnO–bentonite: photodegradation versus adsorbability. J Inst Eng Ser E 98:55–63. https://doi.org/10.1007/s40034-016-0089-1
Tan IAW, Ahmad AL, Hameed BH (2008) Adsorption of basic dye on high-surface-area activated carbon prepared from coconut husk: equilibrium, kinetic and thermodynamic studies. J Hazard Mater 154:337–346. https://doi.org/10.1016/j.jhazmat.2007.10.031
Tran HV, Tran LD, Nguyen TN (2010) Preparation of chitosan/magnetite composite beads and their application for removal of Pb(ii) and Ni(ii) from aqueous solution. Mater Sci Eng C 30:304–310. https://doi.org/10.1016/j.msec.2009.11.008
Wang H, Mao J, Zhang Z, Zhang Q, Zhang L, Zhang W, Li P (2019) Photocatalytic degradation of deoxynivalenol over dendritic-like α-Fe2O3 under visible light irradiation. Toxins 11:105
Wang L, Zhang J, Wang A (2011) Fast removal of methylene blue from aqueous solution by adsorption onto chitosan-g-poly (acrylic acid)/attapulgite composite. Desalination 266:33–39. https://doi.org/10.1016/j.desal.2010.07.065
Zanjanchi MA, Sajjadi H, Arvand M, Mohammad-Khah A, Ghalami-Choobar B (2011) Modification of MCM-41 with anionic surfactant : a convenient design for efficient removal of cationic dyes from wastewater. Clean 39:1007–1013. https://doi.org/10.1002/clen.201000350
Acknowledgements
PS, and SM would like to thanks Amity University, Haryana for supporting this work. SM would like to acknowledge the support provided under the DST-FIST Grant No.SR/FST/PS-I/2019/68 of Govt. of India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there was no conflict of interest in this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, P., Yadav, V., Kumari, S. et al. Deciphering the potent application of nanobentonite and α-Fe2O3/bentonite nanocomposite in dye removal: revisiting the insights of adsorption mechanism. Appl Nanosci 13, 883–897 (2023). https://doi.org/10.1007/s13204-021-01927-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-021-01927-z