Abstract
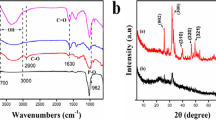
Calcium hydroxyapatatite (CHAP) has been fabricated from waste material as an egg shell towards the applicability of glyphosate (GLY) removal from aqueous phase via biosorption method. However, regarding parameters, which have been obtained to influence adsorption capability to perform the optimization study by Box–Behnken design (BBD). The materials were characterized by BET surface, field-emission scanning electron microscope (FESEM) equipped with an energy-dispersive X-ray spectrometer (FESEM-EDX), Fourier transformed infrared spectroscopy (FT-IR study), zero-point charge (ZPC), X-ray patterns (XRD), etc. The highest optimization was recorded as 99.9532% adsorption, regarding adsorption dose of 0.05 g, GLY concentration of 49.99 mg/L and pH of 4.22, respectively. The adsorption kinetics study was demonstrated with pseudo-first order, pseudo-second order and intraparticle diffusion modelling, as compared to maximum fitted with pseudo-second order. The study of equilibrium was employed through Langmuir, Freudlich, Dubinin–Radushkevitch (D–R), Tempkin isotherms, leads maximum with Freundlich (KF = 177.47 Lmg−1) and Temkin to derive the mechanism as usually engaged chemisorption. The thermodynamic parameters revealed that ΔHo (5E-13 kJ/mol) has positive showed with negative on ΔSo (− 0.0572 kJ/mol) to conform endothermic reaction facilitated with spontaneous response. The regeneration study was examined by three types of eluting agents, the NaOH: NaCl medium is the best recommended in stage of six cycles with 80% efficiency. This adsorbent utility could be beneficial to GLY adsorption in sustainable ways.





Similar content being viewed by others
References
Asfaram A, Ghaedi M, Azqhandi MHA et al (2016) Statistical experimental design, least squares-support vector machine (LS-SVM) and artificial neural network (ANN) methods for modeling the facilitated adsorption of methylene blue dye. RSC Adv 6:40502–40516. https://doi.org/10.1039/C6RA01874B
Babić BM, Milonjić SK, Polovina MJ, Kaludierović BV (1999) Point of zero charge and intrinsic equilibrium constants of activated carbon cloth. Carbon 37:477–481. https://doi.org/10.1016/S0008-6223(98)00216-4
Bai SH, Ogbourne SM (2016) Glyphosate: environmental contamination, toxicity and potential risks to human health via food contamination. Environ Sci Pollut Res Int 23:18988–19001. https://doi.org/10.1007/s11356-016-7425-3
Banerjee S, Chattopadhyaya MC (2017) Adsorption characteristics for the removal of a toxic dye, tartrazine from aqueous solutions by a low cost agricultural by-product. Arab J Chem 10:S1629–S1638. https://doi.org/10.1016/j.arabjc.2013.06.005
Benbrook CM (2016) Trends in glyphosate herbicide use in the United States and globally. Environ Sci Eur. https://doi.org/10.1186/s12302-016-0070-0
Bhaskara BL, Nagaraja P (2006) Direct sensitive spectrophotometric determination of glyphosate by using Ninhydrin as a chromogenic reagent in formulations and environmental water samples. Helv Chim Acta 89:2686–2693. https://doi.org/10.1002/hlca.200690240
Bhattacharyya S, Bhattacharjee S, Mondal NK (2015) A quantum backpropagation multilayer perceptron (QBMLP) for predicting iron adsorption capacity of calcareous soil from aqueous solution. Appl Soft Computing 27:299–312. https://doi.org/10.1016/j.asoc.2014.11.019
Bhaumik R, Mondal NK (2016) Optimizing adsorption of fluoride from water by modified banana peel dust using response surface modelling approach. Appl Water Sci 6:115–135
Boukhalfa N, Boutahala M, Djebri N, Idris A (2019) Kinetics, thermodynamics, equilibrium isotherms, and reusability studies of cationic dye adsorption by magnetic alginate/oxidized multiwalled carbon nanotubes composites. Int J Biol Macromol 123:539–548. https://doi.org/10.1016/j.ijbiomac.2018.11.102
Carneiro RTA, Taketa TB, Gomes Neto RJ et al (2015) Removal of glyphosate herbicide from water using biopolymer membranes. J Environ Manage 151:353–360. https://doi.org/10.1016/j.jenvman.2015.01.005
Chattoraj S, Mondal NK, Sadhukhan B et al (2016) Optimization of adsorption parameters for removal of carbaryl insecticide using neem bark dust by response surface methodology. Water Conserv Sci Eng 1:127–141. https://doi.org/10.1007/s41101-016-0008-9
Chen F, Zhou C, Li G, Peng F (2016) Thermodynamics and kinetics of glyphosate adsorption on resin D301. Arab J Chem 9:S1665–S1669. https://doi.org/10.1016/j.arabjc.2012.04.014
Chen Q, Zheng J, Yang Q et al (2019) Insights into the glyphosate adsorption behavior and mechanism by a MnFe2O4@cellulose-activated carbon magnetic hybrid. ACS Appl Mater Interfaces 11:15478–15488. https://doi.org/10.1021/acsami.8b22386
De Angelis G, Medeghini L, Conte AM, Mignardi S (2017) Recycling of eggshell waste into low-cost adsorbent for Ni removal from wastewater. J Clean Prod 164:1497–1506. https://doi.org/10.1016/j.jclepro.2017.07.085
De Gisi S, Lofrano G, Grassi M, Notarnicola M (2016) Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: a review. Sustain Mater Technol 9:10–40. https://doi.org/10.1016/j.susmat.2016.06.002
Di Guardo A, Finizio A (2018) A new methodology to identify surface water bodies at risk by using pesticide monitoring data: the glyphosate case study in Lombardy region (Italy). Sci Total Environ 610–611:421–429. https://doi.org/10.1016/j.scitotenv.2017.08.049
Dissanayake Herath GA, Poh LS, Ng WJ (2019) Statistical optimization of glyphosate adsorption by biochar and activated carbon with response surface methodology. Chemosphere 227:533–540. https://doi.org/10.1016/j.chemosphere.2019.04.078
Dzigbor A, Chimphango A (2019) Production and optimization of NaCl-activated carbon from mango seed using response surface methodology. Biomass Conv Bioref 9:421–431. https://doi.org/10.1007/s13399-018-0361-3
Ferro AC, Guedes M (2019) Mechanochemical synthesis of hydroxyapatite using cuttlefish bone and chicken eggshell as calcium precursors. Mater Sci Eng C 97:124–140. https://doi.org/10.1016/j.msec.2018.11.083
Ghaedi M, Sadeghian B, Pebdani AA et al (2012) Kinetics, thermodynamics and equilibrium evaluation of direct yellow 12 removal by adsorption onto silver nanoparticles loaded activated carbon. Chem Eng J 187:133–141. https://doi.org/10.1016/j.cej.2012.01.111
Ghaedi M, Ansari A, Bahari F et al (2015) A hybrid artificial neural network and particle swarm optimization for prediction of removal of hazardous dye brilliant green from aqueous solution using zinc sulfide nanoparticle loaded on activated carbon. Spectrochim Acta Part A Mol Biomol Spectrosc 137:1004–1015. https://doi.org/10.1016/j.saa.2014.08.011
Goloshchapov DL, Kashkarov VM, Rumyantseva NA et al (2013) Synthesis of nanocrystalline hydroxyapatite by precipitation using hen’s eggshell. Ceram Int 39:4539–4549. https://doi.org/10.1016/j.ceramint.2012.11.050
Hokkanen S, Bhatnagar A, Repo E et al (2016) Calcium hydroxyapatite microfibrillated cellulose composite as a potential adsorbent for the removal of Cr(VI) from aqueous solution. Chem Eng J 283:445–452. https://doi.org/10.1016/j.cej.2015.07.035
Ingaramo P, Alarcón R, Muñoz-de-Toro M, Luque EH (2020) Are glyphosate and glyphosate-based herbicides endocrine disruptors that alter female fertility? Mol Cell Endocrinol. https://doi.org/10.1016/j.mce.2020.110934
Jiang X, Ouyang Z, Zhang Z et al (2018) Mechanism of glyphosate removal by biochar supported nano-zero-valent iron in aqueous solutions. Colloids Surf A 547:64–72. https://doi.org/10.1016/j.colsurfa.2018.03.041
Khan S, Zhou JL, Ren L, Mojiri A (2020) Effects of glyphosate on germination, photosynthesis and chloroplast morphology in tomato. Chemosphere 258:127350. https://doi.org/10.1016/j.chemosphere.2020.127350
Kordatos K, Gavela S, Ntziouni A et al (2008) Synthesis of highly siliceous ZSM-5 zeolite using silica from rice husk ash. Microporous Mesoporous Mater 115:189–196. https://doi.org/10.1016/j.micromeso.2007.12.032
Le DT, Le TPT, Do HT, et al (2019) Fabrication of porous hydroxyapatite granules as an effective adsorbent for the removal of aqueous Pb(II) ions. J Chem https://www.hindawi.com/journals/jchem/2019/8620181/. Accessed 19 Jul 2020
Lin K, Chang J (2015) 1: Structure and properties of hydroxyapatite for biomedical applications. In: Mucalo M (ed) Hydroxyapatite (Hap) for Biomedical Applications. Woodhead Publishing 3–19
Marbawi H, Khudri MAMRS, Othman AR et al (2019) Isothermal modelling of the adsorption of glyphosate onto palm oil fronds activated carbon. J Environ Microbiol Toxicol 7:21–26
Matozzo V, Fabrello J, Masiero L et al (2018) Ecotoxicological risk assessment for the herbicide glyphosate to non-target aquatic species: a case study with the mussel Mytilus galloprovincialis. Environ Pollut 233:623–632. https://doi.org/10.1016/j.envpol.2017.10.100
Mayakaduwa SS, Kumarathilaka P, Herath I et al (2016) Equilibrium and kinetic mechanisms of woody biochar on aqueous glyphosate removal. Chemosphere 144:2516–2521. https://doi.org/10.1016/j.chemosphere.2015.07.080
Meski S, Ziani S, Khireddine H (2010) Removal of lead ions by hydroxyapatite prepared from the egg shell. J Chem Eng Data 55:3923–3928. https://doi.org/10.1021/je901070e
Nakano T, Ikawa N, Ozimek L (2003) Chemical composition of chicken eggshell and shell membranes. Poult Sci 82:510–514. https://doi.org/10.1093/ps/82.3.510
Okada E, Allinson M, Barral MP et al (2020) Glyphosate and aminomethylphosphonic acid (AMPA) are commonly found in urban streams and wetlands of Melbourne. Aust Water Res 168:115139. https://doi.org/10.1016/j.watres.2019.115139
Pollegioni L, Schonbrunn E, Siehl D (2011) Molecular basis of glyphosate resistance: different approaches through protein engineering. FEBS J 278:2753–2766. https://doi.org/10.1111/j.1742-4658.2011.08214.x
Rehman I, Bonfield W (1997) Characterization of hydroxyapatite and carbonated apatite by photo acoustic FTIR spectroscopy. J Mater Sci Mater Med 8:1–4. https://doi.org/10.1023/A:1018570213546
Sadhukhan B, Mondal NK, Chattoraj S (2016) Optimisation using central composite design (CCD) and the desirability function for sorption of methylene blue from aqueous solution onto Lemna major. Karbala Int J Mod Sci 2:145–155
Santos TRT, Andrade MB, Silva MF et al (2019) Development of α- and γ-Fe2O3 decorated graphene oxides for glyphosate removal from water. Environ Technol 40:1118–1137. https://doi.org/10.1080/09593330.2017.1411397
Schönbrunn E, Eschenburg S, Shuttleworth WA et al (2001) Interaction of the herbicide glyphosate with its target enzyme 5-enolpyruvylshikimate 3-phosphate synthase in atomic detail. Proc Natl Acad Sci USA 98:1376–1380
Sen K, Mondal NK (2020) Facile fabrication of amino-functionalized silicon flakes for removal of organophosphorus herbicide. In silico optimization. Water Conserv Sci Eng 5:67–80. https://doi.org/10.1007/s41101-020-00085-7
Sen K, Mondal NK, Chattoraj S, Datta JK (2016) Statistical optimization study of adsorption parameters for the removal of glyphosate on forest soil using the response surface methodology. Environ Earth Sci 76:22. https://doi.org/10.1007/s12665-016-6333-7
Sen K, Datta JK, Mondal NK (2019) Glyphosate adsorption by Eucalyptus camaldulensis bark-mediated char and optimization through response surface modeling. Appl Water Sci 9:162. https://doi.org/10.1007/s13201-019-1036-3
Silva V, Montanarella L, Jones A et al (2018) Distribution of glyphosate and aminomethylphosphonic acid (AMPA) in agricultural topsoils of the European Union. Sci Total Environ 621:1352–1359. https://doi.org/10.1016/j.scitotenv.2017.10.093
Tarazona JV, Court-Marques D, Tiramani M et al (2017) Glyphosate toxicity and carcinogenicity: a review of the scientific basis of the European Union assessment and its differences with IARC. Arch Toxicol 91:2723–2743. https://doi.org/10.1007/s00204-017-1962-5
Thang NH, Dang PT (2020) Characterization of hydroxyapatite synthesized from calcium hydroxide and phosphoric acid as adsorbents of lead in wastewater. In: materials science forum. https://www.scientific.net/MSF.991.159. Accessed 19 Jul 2020
Vega ED, Pedregosa JC, Narda GE, Morando PJ (2003) Removal of oxovanadium(IV) from aqueous solutions by using commercial crystalline calcium hydroxyapatite. Water Res 37:1776–1782. https://doi.org/10.1016/S0043-1354(02)00565-1
Acknowledgements
The authors would like to thank USIC, the University of Burdwan for the FESEM-EDX measurement and staff. Member of the Department of Environmental Sciences (BU), for providing facilities and help continue the research work. Other device facilities are provided by DST-FIST (SR/FST/ESI-141/2015, dt: 30.09.2019) and WBDST-BOOST, Govt. of West Bengal (39/WBBDC/1p-2/2013, dt: 25.03.2015).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no interest in competition or controversy.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sen, K., Datta, J.K. & Mondal, N.K. Box–Behnken optimization of glyphosate adsorption on to biofabricated calcium hydroxyapatite: kinetic, isotherm, thermodynamic studies. Appl Nanosci 11, 687–697 (2021). https://doi.org/10.1007/s13204-020-01612-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-020-01612-7