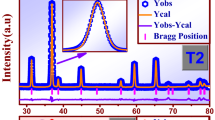
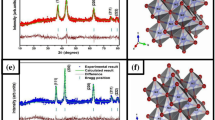
Abstract
Sol–gel method was used to synthesize pure and doped (Ni, Co, and Fe) Cr2O3 nanoparticles (NixCr2-xO3, CoxCr2-xO3, and FexCr2-xO3, where x = 0.00, 0.01, 0.03, 0.05, and 0.07). The structural properties of the prepared samples were determined using x-ray powder diffraction (XRD) with an aim to investigate the influence of doping concentration on the behavior of pure and doped Cr2O3 nanoparticles (NPs). The average crystallite size was estimated with Debye Scherrer’s formula. Morphology of pure and doped (Ni, Co, and Fe) Cr2O3 nanoparticles was examined using field emission scanning electron microscope (FESEM). Fourier transform infrared spectroscopy (FT-IR) was employed to ascertain surface group species of the samples. UV–Vis spectroscopy was used to determine the energy band gap of samples through optical absorption spectrum. Photoluminescence spectroscopy was undertaken to acquire emission and absorption spectra and determine defects in the structures of all synthesized nanopowder samples. Antibacterial activity of pure and doped (Ni, Co, and Fe) Cr2O3 nanoparticles was investigated against Gram-negative bacteria such as Escherichia coli (E. coli) and Gram-positive bacteria Staphylococcus aureus (S. aureus) using paper disc diffusion method and pour plate procedure. Enhanced anti-bactericidal activity was shown against Gram-negative bacteria compared to Gram-positive bacteria. The minimum and maximum inhibition of E. coli and S. aureus was recorded with 40 and 100 μg/ml of nanoparticles, respectively. Furthermore, iron-doped Cr2O3 was more effective in restricting growth of bacterial cell compared to cobalt- and nickel-doped Cr2O3. Superior activity observed against E. coli and S. aureus in terms of inhibition make these samples suitable and potential candidates in medical and paint industries.
















Similar content being viewed by others
References
Abdullah MM, Rajab FM, Al-Abbas SM (2014) Structural and optical characterization of Cr2O3 nanostructures: evaluation of its dielectric properties. AIP Adv. https://doi.org/10.1063/1.4867012
Ackerley DF, Barak Y, Lynch SV, Curtin J, Matin A (2006) Effect of chromate stress on Escherichia coli K-12 effect of chromate stress on Escherichia coli K-12. J Bacteriol 188:3371–3381. https://doi.org/10.1128/JB.188.9.3371
Aghaie-Khafri M, Lafdani MHK (2012) A novel method to synthesize Cr2O3 nanopowders using EDTA as a chelating agent. Powder Technol 222:152–159. https://doi.org/10.1016/j.powtec.2012.02.024
Ahmad S, Farrukh MA, Khan M, Khaleeq-ur-Rahman M, Tahir MA (2014) Synthesis of iron oxide–tin oxide nanoparticles and evaluation of their activities against different bacterial strains. Can Chem Trans 2:122–133. https://doi.org/10.13179/canchemtrans.2014.02.02.0071
Akbarzadeh A, Samiei M, Davaran S (2012) Magnetic nanoparticles: preparation, physical properties, and applications in biomedicine. Nanoscale Res Lett 7:1–13. https://doi.org/10.1186/1556-276X-7-144
Alahmadi NS, Betts JW, Cheng F, Francesconi MG, Kelly SM (2017) Synthesis and antibacterial effects of cobalt–cellulose magnetic nanocomposites. RSC Adv. https://doi.org/10.1039/c7ra00920h
Almontasser A, Parveen A (2020) Preparation and characterization of chromium oxide nanoparticles. In: 3rd International conference condensed matter applied physics, vol 2220, p 020010. https://doi.org/10.1063/5.0001685
Almontasser A, Parveen A, Azam A (2019) Synthesis, characterization and antibacterial activity of magnesium oxide (MgO) nanoparticles. IOP Conf Ser Mater Sci Eng. https://doi.org/10.1088/1757-899X/577/1/012051
Ananda S, Gowda NMM (2013) Synthesis of chromium(III) oxide nanoparticles by electrochemical method and Mukia maderaspatana decomposition and antibacterial study. Mod Res Catal 2:127–135
Balachandran U, Siegel RW, Liao YX, Askew TR (1995) Synthesis, sintering, and magnetic of nanophase Cr2O3 properties. Nanostruct Mater 5:505–512
Berry CC, Curtis ASG (2003) Functionalisation of magnetic nanoparticles for applications in biomedicine. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/36/13/203
Bumajdad A (2018) Non-noble, efficient catalyst of unsupported α-Cr2O3 nanoparticles for low temperature CO oxidation. Sci Rep. https://doi.org/10.1038/s41598-017-14779-x
Cava RJ (2000) Oxide superconductors. J Am Ceram Soc 83:5–28. https://doi.org/10.1111/j.1151-2916.2000.tb01142.x
Dhas NA, Koltypin Y, Gedanken A (1997) Sonochemical preparation and characterization of ultrafine chromium oxide and manganese oxide powders. Chem Mater 9:3159–3163. https://doi.org/10.1021/cm9704645
Fahim RB, Zaki MI, Yacoub NH (1982) Water sorption in relation to surface defect structure of calcined chromia gel. J Colloid Interface Sci 88:502–511. https://doi.org/10.1016/0021-9797(82)90278-8
Gibot P, Vidal L (2010) Original synthesis of chromium(III) oxide nanoparticles. J Eur Ceram Soc 30:911–915. https://doi.org/10.1016/j.jeurceramsoc.2009.09.019
Gibot P, Comet M, Eichhorn A, Schnell F, Muller O, Ciszek F, Boehrer Y, Spitzer D (2011) Highly insensitive/reactive thermite prepared from Cr2O3 nanoparticles. Propellants Explos Pyrotech 36:80–87. https://doi.org/10.1002/prep.201000080
Han R, Zou L, Zhao X, Xu Y, Xu F, Li Y, Wang Y (2009) Characterization and properties of iron oxide-coated zeolite as adsorbent for removal of copper(II) from solution in fixed bed column. Chem Eng J 149:123–131. https://doi.org/10.1016/j.cej.2008.10.015
Hwang J-Y, Seo D-S (2010) Liquid crystal alignment at low temperatures in flexible liquid crystal displays. J Electrochem Soc 157:J351. https://doi.org/10.1149/1.3473785
Jordan A, Scholz R, Wust P, Fähling H (1999) Roland Felix: magnetic fluid hyperthermia (MFH): cancer treatment with AC magnetic field induced excitation of biocompatible superparamagnetic nanoparticles. J Magn Magn Mater 201:413–419. https://doi.org/10.1016/S0304-8853(99)00088-8
Julia B, Ma F, Mun R, Maso N, Cordoncillo E (2004) Environmental study of Cr2O3–Al2O3 green ceramic pigment synthesis. J Eur Ceram Soc 24:2087–2094. https://doi.org/10.1016/S0955-2219(03)00360-1
Kawabata A, Yoshinaka M, Hirota K, Yamaguchi O (1995) Hot isostatic pressing and characterization of sol-gel-derived chromium(II) oxide. J Am Ceram Soc. https://doi.org/10.1111/j.1151-2916.1995.tb08655.x
Lee C, Kim JEEY, Lee WONIL, Nelson KL (2008) Bactericidal effect of zero-valent iron nanoparticles on Escherichia coli. J Environ Sci Technol. https://doi.org/10.1021/es800408u
Li CL, Zhao HX, Takahashi T, Matsumura M (2001) Improvement of corrosion resistance of materials coated with a Cr2O2/NiCr dilayer using a sealing treatment. Mater Sci Eng A 308:268–276. https://doi.org/10.1016/S0921-5093(00)01976-6
Li L, Zhu Z, Yao X, Lu G, Yan Z (2008) Synthesis and characterization of chromium oxide nanocrystals via solid thermal decomposition at low temperature. Microporous Mesoporous Mater 112:621–626. https://doi.org/10.1016/j.micromeso.2007.10.044
Li L, Fan L, Sun M, Qiu H, Li X, Duan H, Luo C (2013) Adsorbent for chromium removal based on graphene oxide functionalized with magnetic cyclodextrin–chitosan. Colloids Surf B Biointerfaces 107:76–83. https://doi.org/10.1016/j.colsurfb.2013.01.074
Madi C, Tabbal M, Christidis T, Isber S, Nsouli B, Zahraman K (2007) Microstructural characterization of chromium oxide thin films grown by remote plasma assisted pulsed laser deposition. J Phys Conf Ser 59:600–604. https://doi.org/10.1088/1742-6596/59/1/128
Mishra SR, Dubenko I, Griffis J, Ali N, Marasinghe K (2009) Exchange bias effect in ball milled Co-Cr2O3 FM-AFM nanocomposites. J Alloys Compd 485:667–671. https://doi.org/10.1016/j.jallcom.2009.06.046
Mohammadtaheri M, Yang Q, Li Y, Corona-Gomez J (2018) The effect of deposition parameters on the structure and mechanical properties of chromium oxide coatings deposited by reactive magnetron sputtering. Coatings 8:111. https://doi.org/10.3390/coatings8030111
Mornet S, Vasseur S, Grasset F, Duguet E (2004) Magnetic nanoparticle design for medical diagnosis and therapy. Forunal Mater Chem 14:2161–2175. https://doi.org/10.1039/b402025a
Noguchi S, Mizuhashi M (1981) Optical properties of CrCo oxide films obtained by chemical spray deposition: substrate temperature effects. Thin Solid Films 77:99–106. https://doi.org/10.1016/0040-6090(81)90364-3
Pang X, Gao K, Luo F, Yang H, Qiao L, Wang Y, Volinsky AA (2008) Annealing effects on microstructure and mechanical properties of chromium oxide coatings. Thin Solid Films 516:4685–4689. https://doi.org/10.1016/j.tsf.2007.08.083
Pankhurst QA, Connolly J, Jones SK, Dobson J (2003) Applications of magnetic nanoparticles in biomedicine. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/36/13/201
Parveen A, Agrawal S (2020) Regulated electromagnetic behavior of transition metal-doped lead sulfide Pb0.9A0.1S (A: Fe Co, and Ni) nanoparticles. J Supercond Nov Magn 33:1159–1165. https://doi.org/10.1007/s10948-019-05338-8
Pillai V, Kumar P, Hou MJ, Ayyub P, Shah DO (1995) Preparation of nanoparticles of silver halides, superconductors and magnetic materials using water-in-oil microemulsions as nano-reactors. Adv Colloid Interface Sci 55:241–269. https://doi.org/10.1016/0001-8686(94)00227-4
Polman EA, Fransen T, Gellings PJ (1989) The reactive element effect: ionic processes of grain-boundary segregation and diffusion in chromium oxide scales. J Phys Condens Matter 1:4497–4510
Rahimi-Nasarabadi M, Ahmadi F, Hamdi S, Eslami N, Didehban K, Ganjali MR (2016) Preparation of nanosized chromium carbonate and chromium oxide green pigment through direct carbonation and precursor thermal decomposition. J Mol Liq 216:814–820. https://doi.org/10.1016/j.molliq.2016.01.065
Rahman MM, Balkhoyor HB, Asiri AM (2017) Phenolic sensor development based on chromium oxide-decorated carbon nanotubes for environmental safety. J Environ Manag 188:228–237. https://doi.org/10.1016/j.jenvman.2016.12.008
Ramesh C, Kumar KM, Senthil M, Ragunathan V (2012) Antibacterial activity of Cr2O3 nanoparticles against E. coli; reduction of chromate ions by Arachis hypogaea leaves. Arch Appl Sci Res 4:1894–1900
Raza M, Kanwal Z, Nassem RS (2016) Antibacterial performance of chromium nanoparticles against Escherichia coli, and Pseudomonas aeruginosa. Adv Civ Environ Mater Res
Ren ZA, Che GC, Dong XL, Yang J, Lu W, Yi W, Shen XL, Li ZC, Sun LL, Zhou F, Zhao ZX (2008) Superconductivity and phase diagram in iron-based arsenic-oxides ReFeAsO1-δ (Re = rare-earth metal) without fluorine doping. EPL. https://doi.org/10.1209/0295-5075/83/17002
Roca AG, Costo R, Rebolledo AF, Veintemillas-Verdaguer S, Tartaj P, González-Carreño T, Morales MP, Serna CJ (2009) Progress in the preparation of magnetic nanoparticles for applications in biomedicine. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/42/22/224002
Sangwan P, Kumar H (2017) Synthesis, characterization and antibacterial activities of chromium oxide nanoparticles against Klebsiella pneumoniae. Asian J Pharm Clin Res 10:206. https://doi.org/10.22159/ajpcr.2017.v10i2.15189
Senthil M, Ramesh C (2012) Biogenic synthesis of Fe3O4 nanoparticles using tridax procumbens leaf extract and its antibacterial activity. Digest J Nanomater Biostruct 7:1655–1660
Singh G, Vajpayee P, Khatoon I, Jyoti A (2011) Chromium oxide nano-particles induce stress in bacteria: probing cell viability. J Biomed Nanotechnol. https://doi.org/10.1166/jbn.2011.1252
Song C, Gui Y, Xing X, Zhang W (2016) Well-dispersed chromium oxide decorated reduced graphene oxide hybrids and application in energy storage. Mater Chem Phys 173:460–466. https://doi.org/10.1016/j.matchemphys.2016.02.039
Tartaj P, Morales MP, Gonzalez-Carreño T, Veintemillas-Verdaguer S, Serna CJ (2011) The iron oxides strike back: from biomedical applications to energy storage devices and photoelectrochemical water splitting. Adv Mater 23:5243–5249. https://doi.org/10.1002/adma.201101368
Tsuzuki T, McCormick PG (2000) Synthesis of metal-oxide nanoparticles by mechanochemical processing. J Metastable Nanocryst Mater 8:383–388. https://doi.org/10.4028/www.scientific.net/JMNM.8.383
Vollath D, Szabó DV, Willis JO (1996) Magnetic properties of nanocrystalline Cr2O3 synthesized in a microwave plasma. Mater Lett 29:271–279. https://doi.org/10.1016/S0167-577X(96)00158-9
Wang Y, Yuan X, Liu X, Ren J, Tong W, Wang Y, Lu G (2008) Mesoporous single-crystal Cr2O3: synthesis, characterization, and its activity in toluene removal. Solid State Sci 10:1117–1123. https://doi.org/10.1016/j.solidstatesciences.2007.11.018
Wang X, Feng Z, Shi J, Jia G, Shen S (2010) Trap states and carrier dynamics of TiO2 studied by photoluminescence spectroscopy under weak excitation condition. Phys Chem Chem Phys. https://doi.org/10.1039/b925277k
Wang H, Zhang Y, Zhang L, Guo Y, Liu S, Gao F, Han Y, Feng G, Liang X, Ge L (2016) Synthesis of C–N dual-doped Cr2O3 visible light-driven photocatalysts derived from metalorganic framework (MOF) for cyclohexane oxidation. RSC Adv. https://doi.org/10.1039/c6ra09908d
Wu L, Gao J, Liu Z, Liang L, Xia F, Cao H (2013) Solar energy materials and solar cells thermal aging characteristics of CrNxOy solar selective absorber coating for FL at plate solar thermal collector applications. Sol Energy Mater Sol Cells 114:186–191. https://doi.org/10.1016/j.solmat.2013.03.005
Yang X, Peng X, Xu C, Wang F (2009) Electrochemical assembly of Ni-xCr-yAl nanocomposites with excellent high-temperature oxidation resistance. J Electrochem Soc 156:C167–C175. https://doi.org/10.1149/1.3082378
Zekaik A, Benhebal H, Benrabah B, Chibout A, Tayebi N, Kharroubi A, Ammari A, Dalache C (2018) Sol-gel synthesis of nickel-doped Cr2O3 thin films. J Mol Eng Mater 5:1–6. https://doi.org/10.1142/S2251237317500125
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that there is no conflict of interest regarding the publication of this paper.
Ethical approval
This paper does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Almontasser, A., Parveen, A., Hashim, M. et al. Structural, optical, and antibacterial properties of pure and doped (Ni, Co, and Fe) Cr2O3 nanoparticles: a comparative study. Appl Nanosci 11, 583–604 (2021). https://doi.org/10.1007/s13204-020-01590-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-020-01590-w