Abstract
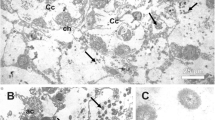
We studied the microbial communities associated with the Baikal ostracods, Candona sp. (Candonidae). Bacterial biofilms were identified on the shell valves of the ostracods localized near epibiotic ciliates of the genus Lagenophrys (Ciliophora: Lagenoprydae). In situ hybridization confirmed the presence of eubacterial cells in different parts of the ostracod’s body. Bacteria of two morphotypes were detected in bacterial biofilms, including filamentous bacteria. Next-generation sequencing of 16S rRNA bacterial gene fragment allowed us to identify representatives of five bacterial phyla. Members of the phyla Proteobacteria and Bacteroidetes typical of non-marine epibiotic associations of ostracods were dominant taxa in the ostracod microbiomes. Members of other taxa (Actinobacteria, Gracilibacteria and Cyanobacteria) are likely facultative and consumed by ostracods together with detritus. In the ostracod microbiome, a significant proportion (20% of all reads) belonged to the sequences of the genus Rickettsia related to intracellular parasites.






Similar content being viewed by others
References
Belkova NL, Denikina NN, Dzyuba EV (2015) Study of the microbiome of the intestine of the Comephorus dybowski (Korotneff, 1904). Biol Bull 42:463–469. https://doi.org/10.1134/S1062359015050039
Bernardet JF, Bowman JP (2006) The Genus Flavobacterium. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The prokaryotes. Springer, New York. https://doi.org/10.1007/0-387-30747-8_17
Bernardet JF, Kerouault B, Michel C (1994) Comparative study on Flexibacter maritimus strains isolated from farmed sea bass (Dicentrarchus labrax) in France. Fish Pathol 29:105–111. https://doi.org/10.3147/jsfp.29.105
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30(15):2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Carman KR, Dobbs FC (1997) Epibiotic microorganisms on copepods and other marine crustaceans. Microsc Res Tech 37:116–135. https://doi.org/10.1002/(sici)1097-0029(19970415)37:2<116::aid-jemt2>3.0.co;2-m
Caro A, Escalas A, Bouvier C, Grousset E (2012) Epibiotic bacterial community of Sphaeroma serratum (Crustacea, isopoda): relationship with molt status. Mar Ecol Prog Ser 457:11–27. https://doi.org/10.3354/meps09711
Cavanaugh CM, McKiness ZP, Newton ILG, Stewart FJ (2013) Marine Chemosynthetic Symbioses. In: Rosenberg E, DeLong EF, Lory S, Stackebrandt E, Thompson F (eds) The Prokaryotes. Springer, Berlin, pp 579–607. https://doi.org/10.1007/978-3-642-30194-0_21
Chigira A, Miura K (2005) Detection of ‘Candidatus Cardinium’ bacteria from the haploid host Brevipalpus californicus (Acari: Tenuipalpidae) and effect on the host. Exp Appl Acarol 37:107–116. https://doi.org/10.1007/s10493-005-0592-4
Dattagupta S, Schaperdoth I, Montanari A, Mariani S, Kita N, Valley JW (2009) A novel symbiosis between chemoautotrophic bacteria and a freshwater cave amphipod. ISME J 3:935–943. https://doi.org/10.1038/ismej.2009.34
Dubilier N, Bergin C, Lott C (2008) Symbiotic diversity in marine animals: the art of harnessing chemosynthesis. Nat Rev Microbiol 6:725–740. https://doi.org/10.1038/nrmicro1992
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Falkner KK, Measures CI, Herbelin SE, Edmond JM, Weiss RF (1991) The major and minor element geochemistry of Lake Baikal. Limnol Oceanogr 36:413–423. https://doi.org/10.4319/lo.1991.36.3.0413
Fernandez-Leborans G, Tato-Porto ML (2000) A review of the species of protozoan epibionts on crustaceans. I Peritrich ciliates. Crustaceana 73:643–683. https://doi.org/10.1163/156854000504705
Garza DR, Dutilh BE (2015) From cultured to uncultured genome sequences: metagenomics and modelling microbial ecosystems. Cell Mol Life Sci 72:4287–4308. https://doi.org/10.1007/s00018-015-2004-1
Gibson RA (1979) Protoraphis atlantica sp. nov., a new marine epizoic diatom. Bacillaria 2:109–126
Gillan DC, Ribesse J, De Ridder C (2004) The iron-encrusted microbial community of Urothoe poseidonis (Crustacea, Amphipoda). J Sea Res 52:21–32. https://doi.org/10.1016/j.seares.2003.08.009
Goffredi SK, Jones WJ, Erhlich H, Springer A, Vrijenhoek RC (2008) Epibiotic bacteria associated with the recently discovered Yeti crab, Kiwa hirsuta. Environ Microbiol 10:2623–2634. https://doi.org/10.1111/j.1462-2920.2008.01684.x
Goffredi SK, Gregory A, Jones WJ, Morella NM, Sakamoto RI (2014) Ontogenetic variation in epibiont community structure in the deep-sea yeti crab, Kiwa puravida: convergence among crustaceans. Mol Ecol 23:1457–1472. https://doi.org/10.1111/mec.12439
Green SL, Bouley DM, Tolwani RJ, Waggie KS, Lifland BD, Otto GM, Ferrell JJ (1999) Identification and management of an outbreak of Flavobacterium meningosepticum infection in a colony of south African clawed frogs (Xenopus laevis). J Am Vet Med Assoc 214:1833–1838
Hachikubo A, Khlystov O, Krylov A, Sakagami H, Minami H, Nunokawa Y, Kida M (2010) Molecular and isotopic characteristics of gas hydrate-bound hydrocarbons in southern and Central Lake Baikal. Geo-Mar Lett 30:321–329. https://doi.org/10.1007/s00367-010-0203-1
Jarett J, Fiore C, Mazel C, Lesser M (2013) Fluorescent epibiotic microbial community on the carapace of a Bahamian ostracod. Arch Microbiol 195:595–604
Kalmychkov GV, Egorov AV, Kuz'min MI, Khlystov OM (2006) Genetic types of methane from Lake Baikal. Dokl Earth Sci 411(5):672–675
Keyser D, Walter R (2004) Calcification in ostracodes. Rev Esp Micropaleontol 36:1–11
Khalzov IA, Mekhanikova I, Sitnikova TY (2018) First data on ectosymbiotic consortia of infusoria and prokaryotes associated with amphipods inhabiting the Frolikha underwater hydrothermal vent, Lake Baikal. Zoologichesky zhurnal 97:1525–1530. https://doi.org/10.1134/S0044513418120073
Khlystov OM, Zemskaya TI, Sitnikova TY, Mekhanikova IV, Kaigorodova IA, Gorshkov AG, Likhoshvai AV (2009) Bottom bituminous constructions and biota inhabiting them according to investigation of Lake Baikal with the Mir submersible. Dokl Earth Sci 429(8):1333–1336. https://doi.org/10.1134/s1028334x09080200
Killworth PD, Carmack EC, Weiss RF, Matear R (1996) Modeling deep-water renewal in Lake Baikal. Limnol Oceanogr 41:1521–1538
Kouris A, Juniper SK, Frebourg G, Gaill F (2007) Protozoan–bacterial symbiosis in a deep-sea hydrothermal vent folliculinid ciliate (Folliculinopsis sp.) from the Juan de Fuca ridge. Mar Ecol 28:63–71
Kozhov MM (1963) Lake Baikal and its life. DR. W. Junk Publishers, The Hague, Netherlands
Kozich JJ, Westcott SL, Baxter NT, Highlander SK, Schloss PD (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl Environ Microbiol 79:5112–5120. https://doi.org/10.1128/AEM.01043-13
Kuznetsov AP, Strizhov BP, Kuzin VS, Fialkov VA, Yastrebov VS (1991) The novelty in Baikal nature. A community based on bacterial chemosynthesis. Proceedings of the Russian Academy of Sciences, Series in Biology 5:766–772
Lazo FI (1980) Sulfur geochemistry in bottom sediments of Lake Baikal. Geokhimiya 1:109–115
Ma WJ, Schwander T (2017) Patterns and mechanisms in instances of endosymbiont induced parthenogenesis. J Evol Biol 30:868–888. https://doi.org/10.1111/jeb.13069
Martens K, Schön I, Meisch C, Horne DJ (2008) Global diversity of ostracods (Ostracoda, Crustacea) in freshwater. Hydrobiologia 595:185–193. https://doi.org/10.1007/978-1-4020-8259-7_20
Mazepova G (1990) Seed shripms (Ostracoda) of Lake Baikal. Nauka, Novosibirsk
Mesquita-Joanes F, Smith AJ, Viehberg FA (2012) The ecology of Ostracoda across levels of biological organisation from individual to ecosystem: a review of recent developments and future potential. In: Horne DJ, Holmes J, Rodriguez-Lazaro J, Viehberg FA (eds) Ostracoda as proxies for quaternary climate change. Elsevier, Amsterdam, pp 15–35. https://doi.org/10.1016/B978-0-444-53636-5.00002-0
Mioduchowska M, Czyż MJ, Gołdyn B, Kilikowska A, Namiotko T, Pinceel T, Łaciak M, Sell J (2018) Detection of bacterial endosymbionts in freshwater crustaceans: the applicability of non-degenerate primers to amplify the bacterial 16S rRNA gene. PeerJ 6:e6039. https://doi.org/10.7717/peerj.6039
Modeo L, Salvetti A, Rossi L, Castelli M, Szokoli F, Krenek S, Serra V, Sabaneyeva E, Di Giuseppe G, Fokin SI, Verni F, Petroni G (2020) "Candidatus Trichorickettsia mobilis", a Rickettsiales bacterium, can be transiently transferred from the unicellular eukaryote Paramecium to the planarian Dugesia japonica. PeerJ Apr 8:e8977. https://doi.org/10.7717/peerj.8977
Olmo C, Armengol X, Anto’n-Pardo M, Ortells R (2016) The environmental and zooplankton community changes in restored ponds over 4 years. J Plankton Res 38:490–501. https://doi.org/10.1093/plankt/fbw021
Olszewski P, Bruhn-Olszewska B, Namiotko L, Sell J, Namiotko T (2020) Co-cultured non-marine ostracods from a temporary wetland harbor host-specific microbiota of different metabolic profiles. Hydrobiologia 847:2503–2519. https://doi.org/10.1007/s10750-020-04269-z
Ondov BD, Bergman NH, Phillippy AM (2011) Interactive metagenomic visualization in a web browser. BMC Bioinform 12:385 http://www.biomedcentral.com/1471-2105/12/385
Ott MJ, Bulgheresi BS (2004) Marine microbial thiotrophic ectosymbioses, oceanograph. Mar Biol Annual Rev 42:95–118. https://doi.org/10.1201/9780203507810.ch4
Pogodaeva TV, Zemskaya TI, Khlystov OM (2012) Preliminary estimate of dissolved components flowing through water-bottom interfase at sites of oil and gas discharge in lake Baikal. 11th Conference of Gas in Marine Sediments 4–7 September 2012 Nice (France) 74
Rautian MS, Wackerow-Kouzova ND (2013) Phylogenetic placement of two previously described intranuclear bacteria from the ciliate Paramecium bursaria (Protozoa, Ciliophora): ‘Holospora acuminata’ and ‘Holospora curviuscula’. Int J Syst Evol Microbiol 63:1930–1933. https://doi.org/10.1099/ijs.0.046631-0
Rossi V, Albini D, Benassi G, Menozzi P (2012) To rest in hydration: hatching phenology of resting eggs of Heterocypris incongruens (Crustacea: Ostracoda). Fundam Appl Limnol 181:49–58. https://doi.org/10.1127/1863-9135/2012/0251
Rubin-Blum M, Chakkiath PA, Sayavedra L, Martínez-Pérez C, Birgel D, Peckmann J, Wu Y-C, Cardenas P, MacDonald I, Marcon Y, Sahling H, Hentschel U, Dubilier N (2019) Fueled by methane: deep-sea sponges from asphalt seeps gain their nutrition from methane-oxidizing symbionts. ISME J 13:1209–1225. https://doi.org/10.1038/s41396-019-0346-7
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541. https://doi.org/10.1128/aem.01541-09
Schön I, Kamiya T, Van den Berghe T, Van den Broecke L, Martens K (2019) Novel Cardinium strains in non-marine ostracod (Crustacea) hosts from natural populations. Mol Phylogenet Evol 130:406–415. https://doi.org/10.1016/j.ympev.2018.09.008
Sievert S, Vetriani C (2012) Chemoautotrophy at deep-sea vents: past, present, and future. Oceanography 25(1):218–233. https://doi.org/10.5670/oceanog.2012.21
Silknetter S, Creed RP, Brown BL, Frimpong EA, Skelton J, Peoples BK (2020) Positive biotic interactions in freshwaters: a review and research directive. Freshw Biol 65:811–832. https://doi.org/10.1111/fwb.13476
Sitnikova TY, Shirokaya AA (2013) New data on deep water Baikal limpets found in hydrothermal vents and oil-seeps. Archiv für Molluskenkunde: International Journal of Malacology 142:257–278. https://doi.org/10.1127/arch.moll/1869-0963/142/257-278
Sitnikova T, Kiyashko S, Bukshuk N, Zemskaya T, Khlystov O (2016) Moore MV stable isotope signatures and distribution of Deepwater sponges in Lake Baikal. Hydrobiologia 773(1):11–22. https://doi.org/10.1007/s10750-016-2674-1
Sitnikova TY, Mekhanikova IV, Sideleva VG, Kiyashko SI, Naumova TV, Zemskaya TI, Khlystov OM (2017a) Trophic relationships between macroinvertebrates and fish in St. Petersburg methane seep community in abyssal zone of Lake Baikal. Contemp Probl Ecol 10:147–156. https://doi.org/10.1134/S1995425517020123
Sitnikova TYA, Sideleva VG, Kiyashko SI, Zemskaya TI, Mekhanikova IV, Khlystov OM, Khalzov IA (2017b) Comparative analysis of communities of macroinvertebrates and fish associated with methane and oil-methane seeps in abyssal Lake Baikal. Uspekhi sovremennoi biologii 137(4):373–386. https://doi.org/10.7868/S0042132417040056
Starliper CE (2011) Bacterial Coldwater disease of fishes caused by Flavobacterium psychrophilum. J Adv Res 2:97–108. https://doi.org/10.1016/j.jare.2010.04.001
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 12:2725–2729. https://doi.org/10.1093/molbev/mst197
Tang KW, Dziallas C, Grossart HP (2011) Zooplankton and aggregates as refuge for aquatic bacteria: Protection from UV, heat and ozone stresses used for water treatment. Environ Microbiol 13:378–390. https://doi.org/10.1111/j.1462-2920.2010.02335.x
Tarasov VG, Gebruk AV, Mironov AN, Moskalev LI (2005) Deep-sea and shallow-water hydrothermal vent communities: two different phenomena? Chem Geol 224:5–39
Unz RF, Head IM (2005) Genus I, Thiothrix Winogradsky 1888, 39AL . In: Brenner DJ, Krieg NR, Staley JT, Garrity GM (eds) Bergey’s manual of systematic bacteriology, vol 2, 2nd edn. Springer, New York, pp 131–142
Vannier J, Abe K (1993) Functional morphology and behavior of Vargula Hilgendor Fii (Ostracoda: Myodocopida) from Japan, and discussion of its crustacean ectoparasites: preliminary results from video recordings. J Crustac Biol 13:51–76. https://doi.org/10.1163/193724093x00444
Vopel K, Reick CH, Arlt G, Pöhn M, Ott JA (2002) Flow microenvironment of two marine peritrich ciliates with ectobiotic chemoautotrophic bacteria. Aquat Microb Ecol 29:19–28. https://doi.org/10.3354/ame029019
Wahl M (1989) Marine epibiosis. I Fouling and antifouling: Some basic aspects. Mar Ecol Progr Ser 58:175–189. https://doi.org/10.3354/meps058175
Wahl M (2008) Ecological lever and interface ecology: epibiosis modulates the interactions between host and environment. Biofouling 24:427–438. https://doi.org/10.1080/08927010802339772
Wallner G, Amann R, Beisker W (1993) Optimizing fluorescent in situ hybridization with rRNA targeted oligonucleotide probes for flow cytometric identification of microorganisms. Cytometry. 14:136–143. https://doi.org/10.1002/cyto.990140205
Weissman P, Lonsdale DJ, Yen J (1993) The effect of peritrich ciliates on the production of Acartia hudsonica in Long Island sound. Limnol Oceanogr 38:613–622. https://doi.org/10.4319/lo.1993.38.3.0613
Wernegreen JJ (2012) Endosymbiosis. Curr Biol 22(14):R555–R561. https://doi.org/10.1016/j.cub.2012.06.010
Westcott SL, Schloss PD (2017) OptiClust, an improved method for assigning amplicon-based sequence data to operational taxonomic units. mSphere 2:e00073–e00017. https://doi.org/10.1128/mspheredirect.00073-17
Willey RL, Willey RB, Threlkeld ST (1993) Planktivore effects of zooplankton epibiont communities: Epibiont pigmentation effects. Limnol Oceanogr 38:1818–1822. https://doi.org/10.4319/lo.1993.38.8.1818
Winters AD, Marsh TL, Brenden TO, Faisal M (2015) Analysis of bacterial communities associated with the benthic amphipod Diporeia in the Laurentian Great Lakes Basin. Can J Microbiol 61:72–81. https://doi.org/10.1139/cjm-2014-0434
Zakharenko AS, Galachyants YP, Morozov IV, Shubenkova OV, Morozov AA, Ivanov VG, Pimenov NV, Krasnopeev AY, Zemskaya TI (2019) Bacterial Communities in Areas of Oil and Methane Seeps in Pelagic of Lake Baikal. Microb Ecol 78(2):269–285. https://doi.org/10.1007/s00248-018-1299-5
Zemskaya TI, Sitnikova TY, Kiyashko SI, Kalmychkov GV, Pogodaeva TV, Mekhanikova IV, Naumova TV, Shubenkova OV, Chernitsina SM, Kotsar OV, Chernyaev ES, Khlystov OM (2012) Faunal communities at sites of gas-and oil-bearing fluids in Lake Baikal. Geo-Mar Lett 32:437–451. https://doi.org/10.1007/s00367-012-0297-8
Zemskaya TI, Lomakina AV, Mamaeva EV, Zakharenko AS, Pogodaeva TV, Petrova DP, Galachyants YP (2015) Bacterial communities in sediments of Lake Baikal from areas with oil and gas discharge. Aquat Microb Ecol 76:95–109. https://doi.org/10.3354/ame01773
Zhang Y-K, Chen Y-T, Yang K, Hong X-Y (2016) A review of prevalence and phylogeny of the bacterial symbiont Cardinium in mites (subclass: Acari). Syst Appl Acarol 21(7):978–990. https://doi.org/10.11158/saa.21.7.11
Zinger L, Gobet A, Pommier T (2012) Two decades of describing the unseen majority of aquatic microbial diversity. Mol Ecol 21:1878–1896. https://doi.org/10.1111/j.1365-294X.2011.05362.x
Acknowledgements
The authors thank the crew of the research vessel “G.Yu. Vereschagin” for help in sampling. This study was carried out using computational resources of the HPC-cluster “Akademik V.M. Matrosov” of Irkutsk Supercomputer Center of SB RAS.
Microscopic studies were carried out in the Electron Microscopy Center of Collective Instrumental Center “Ultramicroanalysis” at Limnological Institute Siberian Branch of the Russian Academy of Sciences (http://www.lin.irk.ru/).
This work was financially supported by the State Task, project No. 0279-2021-0006 (LIN SB RAS).
We also acknowledge the valuable comments and suggestions of two anonymous reviewers.
Author information
Authors and Affiliations
Contributions
TZ conceived, designed and coordinated the study. Sample collection, material preparation and biomolecular experiments were performed by IK and TS. NGS data analysis was performed by SB, IK and YG. SB, SC and AZ prepared figures and tables. The first draft of the manuscript was written by TZ and IK; all authors commented on the previous versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
No specific permissions were required for the sampled locations, and the study did not involve endangered or protected species.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khalzov, I.A., Bukin, S.V., Zakharenko, A.S. et al. Microbial communities associated with the ostracods Candona sp. inhabiting the area of the methane seep Goloustnoye (Lake Baikal). Symbiosis 85, 163–174 (2021). https://doi.org/10.1007/s13199-021-00802-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-021-00802-3