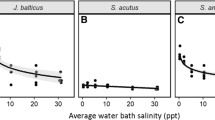
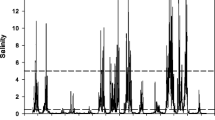
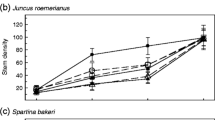
Abstract
In a tidal freshwater marsh (TFM) in lower Chesapeake Bay, salt marsh cordgrass (Spartina alterniflora) recently has become established where TFM vegetation previously grew. To determine the potential contribution of saltwater intrusion to the observed species transition, we conducted a mesocosm study subjecting Spartina and two TFM perennials (Peltandra virginica and Leersia oryzoides) to sub-lethal levels of salinity (0, 2, 4 and 6). We measured plant performance as carbon dioxide flux over the leaf stomata as a proxy for net photosynthesis (Anet), aboveground and belowground biomass and tissue carbon (C), nitrogen (N) and phosphorus. For Spartina, all metrics were unchanged across the range of tested salinity. The TFM species, however, had lower Anet than Spartina and decreased with added salinity. TFM biomass was reduced up to 86 % in both the aboveground and belowground compartments, and aboveground nutrient allocation was altered: N increased in Leersia and P increased in Peltandra with increasing salinity. Under this simulation of increased salt intrusion associated with climate change and sea level rise, TFM species responded with decreased biomass and decreased tissue C:N. Although other factors must contribute, the abiotic salt stressor leads to plant responses consistent with the observed replacement of TFM species by Spartina.



Similar content being viewed by others
References
Baldwin AH, Egnotovich MS, Clarke E (2001) Hydrologic change and vegetation of tidal freshwater marshes: field, greenhouse, and seed-bank experiments. Wetlands 21:519–531
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society: Series B: Methodological 57:289–300
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Annals of Statistics 29:1165–1188
Boon JD (2012) Evidence of sea level acceleration at U.S. and Canadian tidal stations, Atlantic coast, North America. Journal of Coastal Research 28:1437–1445
Chambers RM, Fourqurean JM (1991) Alternative criteria for assessing nutrient limitation of a wetland macrophyte (Peltandra virginica (L.) Kunth). Aquatic Botany 40:305–320
Crain CM, Silliman BR, Bertness SL, Bertness MD (2004) Physical and biotic drivers of plant distribution across estuarine salinity gradients. Ecology 85:2539–2549
Davies SD (2004) Vegetation dynamics of a tidal freshwater marsh: long-term and inter-annual variability and their relationship to salinity. Master’s thesis. The College of William and Mary
Elser JJ, Fagan WF, Denno RF, Dobberfuhl DR, Folarin A et al (2000) Nutritional contraints in terrestrial and freshwater food webs. Nature 408:578–580
Engels JG, Jensen K (2010) Role of biotic interactions and physical factors in determining the distribution of marsh species along an estuarine salinity gradient. Oikos 119:679–685
Eskalinen A, Harrison S, Tuomi M (2012) Plant traits mediate consumer and nutrient control on plant community productivity and diversity. Ecology 93:2705–2718
Flynn KM, McKee KL, Mendelssohn IA (1995) Recovery of freshwater marsh vegetation after a saltwater intrusion event. Oecologia 103:63–72
Geng Y, Pan X, Xu C, Zhang W, Li B, Chen J (2007) Plasticity and ontogenetic draft of biomass allocation in response to above- and below-ground resource availabilities in perennial herbs: a case study of Alternanthera philoxeroides. Ecological Research 22:255–260
Greenwood ME, MacFarlane GR (2006) Effects of salinity and temperature on the germination of Phragmites australis, Juncus kraussii, and Juncus acutus: Implications for estuarine restoration initiatives. Wetlands 26:854–861
Guo H, Pennings SC (2012) Mechanisms mediating plant distributions across estuarine landscapes in a low-latitude tidal estuary. Ecology 93:90–100
Hester MW, Mendelssohn IA, McKee KL (1998) Intraspecific variation in salt tolerance and morphology in Panicum hemitomon and Spartina alterniflora (Poaceae). International Journal of Plant Science 159:127–138
Hester MW, Mendelssohn IA, McKee KL (2001) Species and population variation to salinity stress in Panicum hemitomom, Spartina patens and Spartina alterniflora: morphological and physiological constraints. Environmental and Experimental Botany 46:277–297
Holdahl SR, Morrison NL (1974) Regional investigations of vertical crustal movements in the US, using precise relevelings and mareograph data. Tectonophysics 23:373–390
Howard RJ, Mendelssohn IA (2000) Structure and composition of oligohaline marsh plant communities exposed to salinity pulses. Aquatic Botany 68:143–164
Jordan TE, Cornwell JC, Boynton WR, Anderson JT (2008) Changes in phosphorus biogeochemistry along an estuarine salinity gradient: The iron conveyor belt. Limnology and Oceanography 53(1):172–184
Marsh AS, Rasse DP, Drake BG, Megonigal JP (2005) Effect of elevated CO2 on carbon pools and fluxes in a brackish marsh. Estuaries 28:694–704
McKee KL, Mendelssohn IA (1989) Response of a freshwater marsh plant community to increased salinity and increased water level. Aquatic Botany 34:301–316
Mitsch WJ, Gosselink JG (2007) Wetlands, 4th edn. Wiley, New York
Morris JT, Sundareshwar PV, Nietch CT, Kjerfve B, Cahoon DR (2002) Responses of coastal wetlands to rising sea level. Ecology 83:2869–2877
Munns R (2002) Comparative physiology of salt and water stress. Plant, Cell and Environment 25:239–250
NOAA: National Oceanic and Atmospheric Administration, Office of Ocean and Coastal Resource Management, National Estuarine Research Reserve System-wide Monitoring Program (2012) Centralized Data Management Office, Baruch Marine Field Lab, University of South Carolina (http://cdmo.baruch.sc.edu)
Neubauer SC (2008) Contributions of mineral and organic components to tidal freshwater marsh accretion. Estuarine, Coastal and Shelf Science 78:78–88
Neubauer SC, Anderson IC (2003) Transport of dissolved inorganic carbon from a tidal freshwater marsh to the York River estuary. Limnology and Oceanography 48:299–307
Neubauer SC, Craft CB (2009) Global change and tidal freshwater wetlands: Scenarios and impacts. In: Barendregt A, Whigham DF, Baldwin AH (eds) Tidal freshwater wetlands. Backhuys, Leiden
Neubauer SC, Miller WD, Anderson IC (2000) Carbon cycling in a tidal freshwater marsh ecosystem: A carbon gas flux study. Marine Ecology Progress Series 199:13–30
Neubauer SC, Anderson IC, Constantine JA, Kuehl SA (2002) Sediment deposition and accretion in a Mid-Atlantic (U.S.A.) tidal freshwater marsh. Estuarine. Coastal and Shelf Science 54:713–727
Odum WE (1984) Comparative ecology of tidal freshwater and salt marshes. Annual Review of Ecology and Systematics 19:147–176
Odum EP, Birch JB, Cooley JL (1983) Comparison of giant cutgrass productivity in tidal and impounded marshes with special reference to tidal subsidy and waste assimilation. Estuaries 6:88–94
Pearcy RW, Ustin SL (1984) Effects of salinity on growth and photosynthesis of three California tidal marsh species. Oecologia 62:68–73
Perry JE, Atkinson RB (1997) Plant diversity along a salinity gradient of four marshes on the York and Pamunkey Rivers in Virginia. Castanea 62:112–118
Perry JE, Hershner CH (1999) Temporal changes in the vegetation pattern in a tidal freshwater marsh. Wetlands 19:90–99
Perry JE, Bilkovic DM, Havens KJ, Hershner CH (2009) Tidal freshwater wetlands of the Mid-Atlantic and southeastern United States. In: Barendregt A, Whigham DF, Baldwin AH (eds) Tidal freshwater wetlands. Backhuys, Leiden
Peterson-Smith J, Baldwin AH (2006) Constraints on understory vegetation communities of tidal freshwater swamps: a reciprocal transplant experiment. Journal of the Torrey Botanical Society 133:566–576
Pinheiro J, Bates DM (2000) Mixed-effects models in S and S-Plus. Statistics and computing series. Springer-Verlag, New York
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Redondo-Gomez S, Mateos-Naranjo E, Davy AJ, Fernandez-Munoz F, Castellanos EM, Luque T, Figueroa ME (2007) Growth and photosynthetic responses to salinity of the salt-marsh shrub Atriplex portulacoides. Annals of Botany 100:555–563
Redondo-Gomez S, Naranjo EM, Garzon O, Castillo JM, Luque T, Figueroa ME (2008) Effects of salinity on germination and seedling establishment of endangered Limonium emarginatum (Willd.) O. Kuntze. Journal of Coastal Research 24(1A):201–205
Rozas LP, Odum WE (1987) Use of tidal freshwater marshes by fishes and macrofaunal crustaceans along a marsh stream-order gradient. Estuaries 10:36–43
Sharpe PJ, Baldwin AH (2012) Tidal marsh plant community response to sea-level rise: a mesocosm study. Aquatic Botany 101:34–40
Simpson RL, Good RE, Leck MA, Whigham DF (1983) The ecology of freshwater tidal wetlands. BioScience 33:255–259
Stevenson CJ, Kearney MS (2009) Impacts of global climate change and sea-level rise on tidal wetlands. In: Silliman BR, Grosholz ED, Bertness MD (eds) Human impacts on salt marshes: a global perspective. University of California Press, Berkeley
Stewart GR, Lee JA (1974) The role of proline accumulation in halophytes. Planta 120:279–289
Storey R, Wyn Jones RG (1977) Quarternary ammonium compounds in plants in relation to salt resistance. Phytochemistry 16:447–453
Stribling JM (1997) the relative importance of sulfate availability in the growth of Spartina alterniflora and Spartina cynosuroides. Aquatic Botany 56:131–143
Turner RE (1978) Community plankton respiration in a salt marsh estuary and the importance of leahcates. Limnology and Oceanography 23:442–451
USDA (2013) The PLANTS database. National Plants Data Center, Baton Rouge Louisiana, USA. http://plants.usda.gov
Zewdie S, Olsson M, Fetene M (2007) Growth, gas exchange, chlorophyll a fluorescence, biomass accumulation and partitioning in droughted and irrigated plants of two enset (Ensete ventricosum Welw. Cheesman) clones. Journal of Agronomy 6:499–508
Acknowledgments
Several undergraduates supported this project: L. Zoe Almeida and Kelsey Bryant braved hours at the “plant farm”, and Madeline Karp and Jeffrey Knox helped prepare many belowground tissues. We also thank Scott Neubauer for an early review and the anonymous reviewer whose comments improved the manuscript. Partial funding was provided by the Garden Club of America. This paper was developed under STAR Fellowship Assistance Agreement no. FP-91736901 awarded by the U.S. Environmental Protection Agency (EPA). It has not been formally reviewed by EPA. The views expressed in this paper are solely those of the authors, and EPA does not endorse any products or commercial services mentioned in this paper. This paper is contribution number 3312 of the Virginia Institute of Marine Science, College of William and Mary.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sutter, L.A., Perry, J.E. & Chambers, R.M. Tidal Freshwater Marsh Plant Responses to Low Level Salinity Increases. Wetlands 34, 167–175 (2014). https://doi.org/10.1007/s13157-013-0494-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-013-0494-x