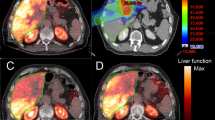
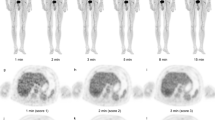
Abstract
Purpose
Establishment of the specifications and standards for successful radiotherapy treatments through identifying three objectives: administering the appropriate low-waste dose, developing dose-delivery skills and monitoring an earlier response to therapy.
Methods
The appropriate low-waste dose is administered via the work-energy principle, considering the interaction between the drug and the tumor as an isolated system. Then, chelated with any compound that could form a lipid-soluble complex with the radioactive metal ions, it is injected directly into the tumor via a multihole needle to improve the distribution of the injectate solution. This can be detected by monitoring the tumor response through newer imaging techniques that combine single photon emission computed tomography (SPECT) with computed tomography (CT), or positron emission tomography (PET) with CT, so that nonresponding tumors can be identified early to modify the administered dose.
Results
The accuracy of estimating the initial effective radioactive dose depends on the equivalence of the growth energy of the tumor estimated from the CT scan and the decay energy of the effective radioactive dose. Besides earlier or more accurate assessment of the tumor response by PET with the glucose analogue 18F-fluoro-2-deoxyglucose (18F-FDG), this contributes to the most safe and low-cost successful treatment. This approach assessed the therapeutic significance of lipid-soluble compounds with the radioactive metal ions in protecting system isolation, which plays a major role in targeted tumor therapy.
Conclusion
Treatment success shows that the three identified objectives are completely dependent objectives. It should also be taken into consideration that radionuclide decay-generated Auger electrons may be more effective in very small tumors to avoid a cross dose.

Similar content being viewed by others
References
Graham J, Muhsin M, Kirkpatrick P (2004) Cetuximab. Nat Rev Drug Discov 3:549–550
Czernin J, Weber WA, Herschman HR (2006) Molecular imaging in the development of cancer therapeutics. Annu Rev Med 57:99–118
Thakur ML, Ron Coss (2003) Role of lipid-soluble complexes in targeted tumor therapy. J Nucl Med 2003;44:1293–1300
Weber WA (2009) Assessing tumor response to therapy. J Nucl Med 50:1S–10S. doi:10.2967/jnumed.108.057174
Barendswaard EC, Humm JL et al (2001) Relative therapeutic efficacy of (125)I- and (131)I-labeled monoclonal antibody A33 in a human colon cancer xenograft. J Nucl Med 42:1251–1256
ICRU (1984) Stopping powers for electrons and positrons, report 37. International Commission on Radiation Units and Measurements, Bethesda
Serway RA, Jewett JW (2008) Physics for scientists and engineers, vol 1, 7th edn. Tomson-Brooks/Cole, Pacific Grove
Smithers DW (1958) The therapeutic use of radioactive isotopes. Ann R Coll Surg Engl 23(4):238–247
Minchinton A, Tannock I (2006) Drug penetration in solid tumors. Nat Rev Cancer 6:583–592
Gillies RJ, Gatenby RA (2007) Hypoxia and adaptive landscapes in the evolution of carcinogenesis. Cancer Metastasis Rev 26:1573
Sullivan R, Graham CH (2007) Hypoxia-driven selection of the metastatic phenotype. Cancer Metastasis Rev 26:319–331
Brown JM (1999) The hypoxic cell: a target for selective cancer therapy—eighteenth Bruce F. Cain Memorial Award lecture. Cancer Res 59:5863–5870
Castello PR, David PS (2006) Mitochondrial cytochrome oxidase produces nitric oxide under hypoxic conditions: implications for oxygen sensing and hypoxic signaling in eukaryotes. Cell Metab 3:277–287
Kuei-Meng Wu (2009) A new classification of prodrugs: regulatory perspectives. Pharmaceuticals 2(3):77–81
EnvironmentalChemistry.com (2010) Periodic table of elements: element iodine—I http://environmentalchemistry.com/yogi/periodic/I.html
Shleien B, Slaback LA Jr, Birky BK (1998) Health physics and radiological health handbook, 3rd edn. Williams and Wilkins, Baltimore, pp 6–11
Holden NE (2005) Table of the isotopes. In: Lide DR (ed) CRC Handbook of chemistry and physics, 85th edn, online version. CRC Press, Boca Raton
Shen DHY, Marsee DK (2004) Effects of dose, intervention time, and radionuclide on sodium iodide symporter (NIS)-targeted radionuclide therapy. Gene Ther 11:161–9. doi:10.1038/sj.gt.3302147
Roberson PL, Buchsbaum DJ (1995) Reconciliation of tumor dose response to external beam radiotherapy versus radioimmunotherapy with 131Iodine-labeled antibody for a colon cancer model. Cancer Res 55:5811S–5816S
Torlakovic G, Grover VK (2005) Easy method of assessing volume of prostate adenocarcinoma from estimated tumor area: using prostate tissue density to bridge gap between percentage involvement and tumor volume. Croat Med J 46(3):423–428
Shields AF, Mankoff DA (1998) Carbon-11-thymidine and FDG to measure therapy response. J Nucl Med 39:1757–1762
Weber WA, Czernin J, Phelps ME, Herschman HR (2008) Technology insight: novel imaging of molecular targets is an emerging area crucial to the development of targeted drugs. Nat Clin Pract Oncol 5:44–54
Wahl RL, Zasadny K, Helvie M, Hutchins GD, Weber B, Cody R (1993) Metabolic monitoring of breast cancer chemohormonotherapy using positron emission tomography: initial evaluation. J Clin Oncol 11:2101–2111
Hoekstra OS, van Lingen A, Ossenkoppele GJ, Golding R, Teule GJ (1993) Early response monitoring in malignant lymphoma using fluorine-18 fluorodeoxyglucose single-photon emission tomography. Eur J Nucl Med 20:1214–1217
Shankar LK, Hoffman JM, Bacharach S et al (2006) Consensus recommendations for the use of 18F-FDG PET as an indicator of therapeutic response in patients in National Cancer Institute Trials. J Nucl Med 47:1059–1066
Thakur ML, McAfee JG (1984) The significance of chromosomal aberrations in indium-111-labeled lymphocytes. J Nucl Med 25:922–927
Goddu SM, Howell RW, Rao DV (1995) A generalized approach to absorbed dose calculations for dynamic tumor and organ masses. J Nucl Med 36:1923–1927
Howell RW, Rao DV, Sastry KS (1989) Macroscopic dosimetry for radioimmunotherapy: nonuniform activity distributions in solid tumors. Med Phys 16:66–74
Howell RW (1992) Radiation spectra for Auger electron emitting radionuclides: report No. 2 of AAPM Nuclear Medicine Task Group No. 6. Med Phys 19:1371–1383
Howell RW, Narra VR, Rao DV (1992) Absorbed dose calculations for rapidly growing tumors. J Nucl Med 33:277–281
EnvironmentalChemistry.com (2010) Periodic table of elements: element indium—In http://environmentalchemistry.com/yogi/periodic/In.html
Leroy C. Principles of radiation interaction in matter and detection. 2004.
Meiring WJ, van Klinken J, Wichers VA (1991) Differences between electrons and positrons interacting with detector material. Phys Rev A 44:2960–2967
Hubbel JH (1982) Photon mass attenuation and energy absorption coefficients from 1keV to 20 MeV. Int J Appl Radiat Isot 33:1269–1290
Kassis AI, Howell RW, Sastry KSR, Adelstein SJ (1988) Positional effects of Auger decays in mammalian cells in culture. In: Baverstock KF, Charlton DE (eds) DNA damage of Auger emitters. Taylor and Francis, London, pp 1–4
Sastry KSR (1992) Biological effects of the Auger emitter iodine-125: a review. Report no. 1 of AAPM Nuclear Medicine Task Group No. 6. Med Phys 19:1361–1370
Hofer KG (1980) Radiation biology and potential therapeutic applications of radionuclides. Bull Cancer 67:343–353
Beyer T, Townsend DW, Brun T, Kinahan PE, Charron M, Roddy R et al (2000) A combined PET/CT scanner for clinical oncology. J Nucl Med 41:1369–1379
Yau JSY, Chu KS (2009) Usage of a fixed dose of radioactive iodine for the treatment of hyperthyroidism: one-year outcome in a regional hospital in Hong Kong. Hong Kong Med J 15:267–273
Hindorf C, Linden O, Stenberg L, Tennvall J, Strand SE (2003) Change in tumor-absorbed dose due to decrease in mass during fractionated radioimmunotherapy in lymphoma patients. Clin Cancer Res 9:4003S–4006S
Acknowledgements
The author is grateful to the journals which offered open access to the provided experimental treatments and would like to thank all authors, editors and persons contributing to them, as those experiments provided a great deal to help prove this new concept for calculation of the radiation dose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moawad, E. Isolated System Towards A Successful Radiotherapy Treatment. Nucl Med Mol Imaging 44, 123–136 (2010). https://doi.org/10.1007/s13139-010-0029-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-010-0029-9