Abstract
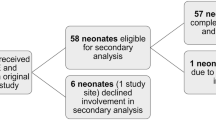
Germinal matrix intraventricular hemorrhage (IVH) is the most common type of intracranial hemorrhage observed in preterm neonates. It is a precursor of poor neurocognitive development, cerebral palsy, and death. The pathophysiology is not well defined, but damage to the fragile germinal matrix vasculature may be due to free radicals generated during inflammation and as a consequence of ischemia followed by reperfusion. Assessment of the oxidative stress status in these infants is therefore important. Urinary allantoin concentration was measured in preterm neonates as a marker of oxidative stress associated with IVH. Urine was collected from 44 preterm neonates at four time points between 24 and 72 hours of life (HOL), and the allantoin content was determined by gas chromatography mass spectrometry (GCMS). Records were retrospectively reviewed, and the incidence and severity of IVH was categorized as follows: no IVH (n = 24), mild (grade 1–2) IVH (n = 13), and severe (grade 3–4) IVH (n = 7). Neonates with severe IVH showed significantly elevated allantoin levels vs subjects with no IVH from 36 HOL (0.098 ± 0.013 μmol and 0.043 ± 0.007 μmol, respectively, p = 0.002). The allantoin concentration remained elevated even at 72 HOL (0.079 ± 0.014 μmol and 0.033 ± 0.008 μmol, respectively, p = 0.021). There were no significant differences in allantoin levels in the no IVH and mild IVH groups. IVH was diagnosed by head imaging on average at about 11th postnatal day. Urinary allantoin levels were significantly elevated during the first 3 days of life in the neonates subsequently diagnosed with severe IVH, suggesting that oxidative stress might be a crucial factor in IVH pathogenesis. Further studies are needed to assess the usefulness of urinary allantoin in early identification of preterm infants at risk for or with severe IVH and monitoring of the response to interventions designed to prevent or treat it.

Similar content being viewed by others
References
Ballabh P. Intraventricular hemorrhage in premature infants: mechanism of disease. Pediatr Res. 2010;67(1):1–8. doi:10.1203/PDR.0b013e3181c1b176.
Choi IR, Lee JH, Park MS, Kim JY, Park KH, Kim G, et al. Early neurodevelopment in very low birth weight infants with mild intraventricular hemorrhage or those without intraventricular hemorrhage. Kor J Pediatr. 2012;55(11):414–9. doi:10.3345/kjp.2012.55.11.414.
McCrea HJ, Ment LR. The diagnosis, management and postnatal prevention of intraventricular hemorrhage in the preterm neonate. Clin Perinatol. 2008;35(4):1–17. doi:10.1016/j.clp.2008.07.014.
Huang BY, Castillo M. Hypoxic-ischemic brain injury: imaging findings from birth to adulthood. Radiographics. 2008;28(2):417–39. doi:10.1148/rg.282075066.
Poralla C, Hertfelder HJ, Oldenburg J, Mueller A, Bartmann P, Heep A. Elevated interleukin-6 concentration and alterations of the coagulation system are associated with the development of intraventricular hemorrhage in extremely preterm infants. Neonatology. 2012;102(4):270–5. doi:10.1159/000341266.
Bhandari V, Bizzarro MJ, Shetty A, Zhong X, Page GP, Zhang H, et al. Familial and genetic susceptibility to major neonatal morbidities in preterm twins. Pediatrics. 2006;117(6):1901–6. doi:10.1542/peds.2005-1414.
Ballabh P. Pathogenesis and prevention of intraventricular hemorrhage. Clin Perinatol. 2014;41(1):47. doi:10.1016/j.c1p.2013.09.007.
Graham EM, Holcroft CI, Rai KK, Donohue PK, Allen MC. Neonatal cerebral white matter injury in preterm infants is associated with culture positive infections and only rarely with metabolic acidosis. Am J Obstet Gynecol. 2004;191(4):1305–10. doi:10.1016/j.ajog.2004.06.058.
Ment LR, Aden U, Lin AP, Kwon SH, Choi M, Hallman M, et al. Gene-environment interactions in severe intraventricular hemorrhage of preterm neonates. Pediatr Res. 2014;75(1):241–50. doi:10.1038/pr.2013.195.
Strahle J, Garton HJ, Maher CO, Muraszko KM, Keep RF, Xi G. Mechanisms of hydrocephalus after neonatal and adult intraventricular hemorrhage. Transl Stroke Res. 2012;3 Suppl 1:25–38. doi:10.1007/s12975-012-0182-9.
Perrone S, Tataranno ML, Negro S, Longini M, Marzocchi B, Proietti F, et al. Early identification of the risk for free radical-related diseases in preterm newborns. Early Hum Dev. 2010;86(4):241–4. doi:10.1016/j.earlhumdev.2010.03.008.
Weinberger B, Nisar S, Anwar M, Ostfeld B, Hegyi T. Lipid peroxidation in cord blood and neonatal outcome. Pediatr Int. 2006;48(5):479–83. doi:10.1111/j.1442-200X.2006.02257.x.
Plank MS, Calderon TC, Asmerom Y, Boskovic DS, Angeles DM. Biochemical measurement of neonatal hypoxia. J Vis Exp : JoVE. 2011;(54). doi:10.3791/2948.
Il'yasova D, Scarbrough P, Spasojevic I. Urinary biomarkers of oxidative status. Clin Chim Acta; Int J Clin Chem. 2012;413(19–20):1446–53. doi:10.1016/j.cca.2012.06.012.
Gruber J, Tang SY, Jenner AM, Mudway I, Blomberg A, Behndig A, et al. Allantoin in human plasma, serum, and nasal-lining fluids as a biomarker of oxidative stress: avoiding artifacts and establishing real in vivo concentrations. Antioxid Redox Signal. 2009;11(8):1767–76. doi:10.1089/ars.2008.2364.
Kaur H, Halliwell B. Action of biologically-relevant oxidizing species upon uric acid. Identification of uric acid oxidation products. Chem Biol Interact. 1990;73(2–3):235–47.
Becker BF, Kastenbauer S, Kodel U, Kiesl D, Pfister HW. Urate oxidation in CSF and blood of patients with inflammatory disorders of the nervous system. Nucleosides Nucleotides Nucleic Acids. 2004;23(8–9):1201–4. doi:10.1081/ncn-200027469.
Causse E, Pradelles A, Dirat B, Negre-Salvayre A, Salvayre R, Couderc F. Simultaneous determination of allantoin, hypoxanthine, xanthine, and uric acid in serum/plasma by CE. Electrophoresis. 2007;28(3):381–7. doi:10.1002/elps.200600205.
Kand'ar R, Zakova P. Allantoin as a marker of oxidative stress in human erythrocytes. Clin Chem Lab Med. 2008;46(9):1270–4. doi:10.1515/cclm.2008.244.
Skalicky J, Muzakova V, Kandar R, Meloun M, Rousar T. Oxidative stress and metabolic syndrome in obese adults with and without controlled diet restriction. Bratisl Lek Listy. 2009;110(3):152–7.
Ogihara T, Okamoto R, Kim HS, Nagai A, Morinobu T, Moji H, et al. New evidence for the involvement of oxygen radicals in triggering neonatal chronic lung disease. Pediatr Res. 1996;39(1):117–9. doi:10.1203/00006450-199601000-00017.
Seet RC, Lee CY, Chan BP, Sharma VK, Teoh HL, Venketasubramanian N, et al. Oxidative damage in ischemic stroke revealed using multiple biomarkers. Stroke; J Cerebr Circ. 2011;42(8):2326–9. doi:10.1161/strokeaha.111.618835.
Zinellu A, Sotgia S, Loriga G, Deiana L, Satta AE, Carru C. Oxidative stress improvement is associated with increased levels of taurine in CKD patients undergoing lipid-lowering therapy. Amino Acids. 2012;43(4):1499–507. doi:10.1007/s00726-012-1223-0.
Deulofeut R, Critz A, Adams-Chapman I, Sola A. Avoiding hyperoxia in infants <= 1250 g is associated with improved short- and long-term outcomes. J Perinatol. 2006;26(11):700–5. doi:10.1038/sj.jp.7211608.
Il'yasova D, Spasojevic I, Wang F, Tolun AA, Base K, Young SP, et al. Urinary biomarkers of oxidative status in a clinical model of oxidative assault. Cancer Epidemiol Biomark Prev. 2010;19(6):1506–10. doi:10.1158/1055-9965.epi-10-0211.
Hellsten Y, Tullson PC, Richter EA, Bangsbo J. Oxidation of urate in human skeletal muscle during exercise. Free Radic Biol Med. 1997;22(1–2):169–74. doi:10.1016/s0891-5849(96)00286-9.
Asmerom Y, Slater L, Boskovic DS, Bahjri K, Holden MS, Phillips R et al. Oral sucrose for heel lance increases adenosine triphosphate use and oxidative stress in preterm neonates. J Pediatr. 2013;163(1):29–35 e1. doi:10.1016/j.jpeds.2012.12.088.
Plank M, Slater L, Asmerom Y, Angeles KR, Mannoia K, Boskovic D et al. Altered urinary excretion of allantoin and purine catabolites in neonates with necrotizing enterocolitis. FASEB J. 2012;26(Meeting Abstracts):898.4.
Tolun AA, Zhang HY, Il'yasova D, Sztaray J, Young SP, Millington DS. Allantoin in human urine quantified by ultra-performance liquid chromatography-tandem mass spectrometry. Anal Biochem. 2010;402(2):191–3. doi:10.1016/j.ab.2010.03.033.
Berthemy A, Newton J, Wu DL, Buhrman D. Quantitative determination of an extremely polar compound allantoin in human urine by LC-MS/MS based on the separation on a polymeric amino column. J Pharm Biomed Anal. 1999;19(3–4):429–34. doi:10.1016/s0731-7085(98)00200-3.
Chung WY, Benzie IFF. Plasma allantoin measurement by isocratic liquid chromatography with tandem mass spectrometry: Method evaluation and application in oxidative stress biomonitoring. Clin Chim Acta. 2013;424:237–44. doi:10.1016/j.cca.2013.06.015.
Papile L, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 1978;92(4):529–34. doi:10.1016/S0022-3476(78)80282-0.
Saugstad OD. Update on oxygen radical disease in neonatology. Curr Opin Obstet Gynecol. 2001;13(2):147–53. doi:10.1097/00001703-200104000-00009.
Grootveld M, Halliwell B. Measurement of allantoin and uric acid in human body fluids. A potential index of free-radical reactions in vivo? Biochem J. 1987;243(3):803–8.
Marklund N, Ostman B, Nalmo L, Persson L, Hillered L. Hypoxanthine, uric acid and allantoin as indicators of in vivo free radical reactions. Description of a HPLC method and human brain microdialysis data. Acta Neurochir. 2000;142(10):1135–42. doi:10.1007/s007010070042.
Il'yasova D, Kennedy K, Spasojevic I, Wang F, Tolun AA, Base K, et al. Individual responses to chemotherapy-induced oxidative stress. Breast Cancer Res Treat. 2011;125(2):583–9. doi:10.1007/s10549-010-1158-7.
Lie KK, Groholt EK, Eskild A. Association of cerebral palsy with Apgar score in low and normal birthweight infants: population based cohort study. BMJ (Clin Res ed). 2010;341:c4990. doi:10.1136/bmj.c4990.
Shankaran S, Lin AP, Maller-Kesselman J, Zhang HP, O'Shea TM, Bada HS, et al. Maternal race, demography, and health care disparities impact risk for intraventricular hemorrhage in preterm neonates. J Pediatr. 2014;164(5):1005. doi:10.1016/j.jpeds.2014.01.036.
Gaudier FL, Goldenberg RL, Nelson KG, PeraltaCarcelen M, DuBard MB, Hauth JC. Influence of acid–base status at birth and Apgar scores on survival in 500-1000-g infants. Obstet Gynecol. 1996;87(2):175–80. doi:10.1016/0029-7844(95)00407-6.
Alvarez-Diaz A, Hilario E, de Cerio FG, Valls-i-Soler A, Alvarez-Diaz FJ. Hypoxic-ischemic injury in the immature brain—key vascular and cellular players. Neonatology. 2007;92(4):227–35. doi:10.1159/000103741.
Basu S, Barman S, Shukla R, Kumar A. Effect of oxygen inhalation on cerebral blood flow velocity in premature neonates. Pediatr Res. 2014;75(2):328–35. doi:10.1038/pr.2013.219.
Saugstad OD. Hypoxanthine as an indicator of hypoxia—its role in health and disease through free-radical production. Pediatr Res. 1988;23(2):143–50. doi:10.1203/00006450-198802000-00001.
Rogers S, Witz G, Anwar M, Hiatt M, Hegyi T. Antioxidant capacity and oxygen radical diseases in the preterm newborn. Arch Pediatr Adolesc Med. 2000;154(6):544–8. doi:10.1001/archpedi.154.6.544.
Gitto E, Pellegrino S, D'Arrigo S, Barberi I, Reiter RJ. Oxidative stress in resuscitation and in ventilation of newborns. Eur Respir J. 2009;34(6):1461–9. doi:10.1183/09031936.00032809.
Saugstad OD. Bronchopulmonary dysplasia and oxidative stress: are we closer to an understanding of the pathogenesis of BPD? Acta Paediatr. 1997;86(12):1277–82. doi:10.1111/j.1651-2227.1997.tb14897.x.
Dani C, Cecci A, Bertini G. Role of oxidative stress as physiopathologic factor in the preterm infant. Minerva Pediatr. 2004;56(4):381–94.
Takashima S, Itoh M, Oka A. A history of our understanding of cerebral vascular development and pathogenesis of perinatal brain damage over the past 30 years. Semin Pediatr Neurol. 2009;16(4):226–36. doi:10.1016/j.spen.2009.09.004.
Compliance with Ethics Requirements
Conflict of Interest
Ijeoma Esiaba, Danilyn M. Angeles, Megan S. Holden, John B.C. Tan, Yayesh Asmerom, Gerald Gollin, and Danilo S. Boskovic declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5). Informed consent was obtained from the parents of all patients for their inclusion in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported in part by Department of Basic Sciences, Department of Earth and Biological Sciences, and by NIH Grant NR011209 (DMA)
Rights and permissions
About this article
Cite this article
Esiaba, I., Angeles, D.M., Holden, M.S. et al. Urinary Allantoin Is Elevated in Severe Intraventricular Hemorrhage in the Preterm Newborn. Transl. Stroke Res. 7, 97–102 (2016). https://doi.org/10.1007/s12975-015-0405-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-015-0405-y