Abstract
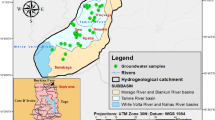
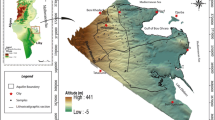
Spatial variation of hydrochemical groundwater parameters indicates that the chemistry is ruled by complex process interactions (mixing of natural and anthropogenic processes). For this approach, analysis of 32 groundwater wells was conducted. Anthropogenic-influenced surface recharge and geogenic processes such as silicate weathering and redox process are identified by multivariate statistics, principal component analysis (PCA) and cluster analysis (CA). Surface water recharge tops the list of factors affecting groundwater chemistry in the Sohag area. The main recharging sources are leaching of salts of overlain soils and irrigation canals associated with anthropogenic impacts such as domestic sewage and agricultural nutrients. The excess of SO42− and HCO3− relative to Ca2+ in the semiconfined shallow aquifer at the floodplain can be attributed to the anthropogenic influence, infiltrated irrigation water and wastewater. On the other hand, in deep wells at the desert fringes, the deficiency of SO42− relative to Ca2+ concentration (however the dissolution of gypsum modeled by MINTEQ) is evidence on sulfides precipitation under reducing conditions as confirmed by the PCA analysis. Ion exchange on clay is another possible reason responsible for the deficiency of Ca2+ + Mg2+/SO42− + HCO3− relative to Na+/Cl− in semiconfined aquifer. Different water types (Na–Cl, Ca–Cl, Mg-–Cl and Na–HCO3) indicate different ion exchange processes formed due to varying contacts between freshwater and saline water. Excessive Mn/Fe ratio is attributed to fertilization and redox processes. Results enable the spatial separation of wells under semiconfined hydraulic conditions recharged by infiltrated irrigation water, from those under unconfined conditions recharged by seepage water leaching salty soils. Accordingly, different water types and values of salinity are complex and could be identified and spatially separated using AqQa and statistical analysis.







Similar content being viewed by others
References
Abdel Latif A, El Kashouty M (2010) Groundwater investigation in Awlad Salameh, Southern Sohag, Upper Egypt. Earth Sci Res J 14:63–75
Abu El Ella EM (1992) Variations in the chemical characters of the Nile Basin Water in Sohag Area, Upper Egypt. In: Proceedings of first international conference on geology of the Arab World, Cairo Univ, Egypt, pp 449–521
Ahmed AA (2007) Using contamination and pesticide DRASTIC GIS-based models for vulnerability assessment of quaternary aquifer at Sohag, Egypt. In: The 5th intern. conf geol Africa, pp 29–47
Awad MA, Nada AA, Hamza MS, Froehlich K (1995) Chemical and isotopic investigation of groundwater in Tahta region, Sohag-Egypt. Environ Geochem Health 17:147–153
Boyacioglu H, Boyacioglu H (2008) Water pollution sources assessment by multivariate statistical methods in the Tahtali Basin, Turkey. Environ Geol 54:275–282
Chen T, Liu X, Zhu M et al (2008) Identification of trace element sources and associated risk assessment in vegetable soils of the urban–rural transitional area of Hangzhou, China. Environ Pollut 151:67–78
Deer WA, Howie RA, Zussman J et al (1992) An introduction to the rock-forming minerals. Longman Scientific and Technical, Hong Kong
Fritz SJ (1994) A survey of charge-balance errors on published analyses of potable ground and surface waters. Groundwater 32:539–546
GASG (General Authority of Sohag Governorate) (2004) Periodical Reports of Information Center. GASG, Sohag
Gomaa AA (2006) Hydrogeological and geophysical assessment of the reclaimed areas in Sohag, Nile Valley, Egypt. Ph.D. Thesis, Faculty of Science, Ain Shams University, Egypt
Gustafsson JP (2011) Visual MINTEQ version 3.1: a windows version of MINTEQA2
Issawi B, McCauley JF (1992) The Cenozoic rivers of Egypt: the Nile problem. Follow Horus Stud Dedic Michael Allen Hoffman Oxbow Monogr 20:121–138
Issawi B, Hassan MY, Osman R (1978) Geological studies in the area of Kom Ombo, Eastern Desert. Egypt Ann Geol Surv Egypt 8:187–235
Kelepertzis E (2014) Accumulation of heavy metals in agricultural soils of Mediterranean: insights from Argolida basin, Peloponnese, Greece. Geoderma 221:82–90
Ledesma-Ruiz R, Pastén-Zapata E, Parra R et al (2015) Investigation of the geochemical evolution of groundwater under agricultural land: a case study in northeastern Mexico. J Hydrol 521:410–423
Leventeli Y, Yalcin F, Kilic M (2019) An investigation about heavy metal pollution of Duden and Goksu Streams (Antalya, Turkey). Appl Ecol Environ Res 17(2):2423–2436
Matiatos I, Paraskevopoulou V, Lazogiannis K, Botsou F et al (2018) Surface–ground water interactions and hydrogeochemical evolution in a fluvio-deltaic setting: the case study of the Pinios River delta. J Hydrol 561:236–249
McGrath D, Zhang C, Carton OT (2004) Geostatistical analyses and hazard assessment on soil lead in Silvermines area, Ireland. Environ Pollut 127:239–248
Moghimi H (2017) The study of processes affecting groundwater hydrochemistry by multivariate statistical analysis (case study: coastal aquifer of Ghaemshahr, NE-Iran). Open J Geol 7:830
Omer AA (1996) Geological, mineralogical and geochemical studies on the Neogene and Quaternary Nile basin deposits, Qena-Assiut stretch, Egypt. Ph.D. thesis, Geology Dept, Faculty of Science, Sohag, South Valley University
Omer AAM (2003) Impact of the Pleistocene Nile basin sediments on the distribution of iron and manganese in the groundwater, Tema-Nag Hammadi area, Nile Valley, Egypt. In: The 3rd international conference on the geology of Africa, Assiut, Egypt, pp 21–23
Omer AAM, Abdel Moneim AA (2001) Geochemical characteristics of the Pliocene and Pleistocene Nile basin deposits and their influence on the groundwater chemistry in Sohag area. Ann Geol Surv Egypt 24:567–584
Rizk S (2010) Assessment of groundwater quality in Sohag governorate, Egypt. A study in the environmental geochemistry. South Valley University, Sohag
Said R (1990) The geology of Egypt: AA balkema. Brookfield, Rotterdam
Said I, Salman SA, Elnazer AA (2019a) Multivariate statistics and contamination factor to identify trace elements pollution in soil around Gerga city. Egypt Bull Natl Res Cent 43:43
Said I, Salman SA, Samy Y, Awad SA, Melegy A, Hursthouse AS (2019b) Environmental factors controlling potentially toxic element behaviour in urban soils, El Tebbin Egypt. Environ Monit Assess 191:267
Shamrukh M, Corapcioglu MY, Hassona FAA (2001) Modeling the effect of chemical fertilizers on ground water quality in the Nile Valley Aquifer. Egypt Groundw 39:59–67
Shukri NM, Azer N (1952) The mineralogy of Pliocene and more recent sediments in the Faiyum. Inst du Desert Bull 2:10–39
Tabachnick BG, Fidell LS (2007) Using multivariate statistics. Allyn and Bacon/Pearson Education
Yalcin MG, Coskun B, Nyamsari DG, Yalcin F (2019) Geomedical, ecological risk, and statistical assessment of hazardous elements in shore sediments of the Iskenderun Gulf, Eastern Mediterranean. Turk Environ Earth Sci 78:438. https://doi.org/10.1007/s12665-019-8435-5
Yidana SM, Ophori D, Yakubo BB, Samed AA (2012) A factor model to explain the hydrochemistry and causes of fluoride enrichment in groundwater from the middle Voltaian sedimentary aquifers in the northern region, Ghana. ARPN J Eng Appl Sci 7:50–68
Zhang Y, Xu M, Li X et al (2018) Hydrochemical characteristics and multivariate statistical analysis of natural water system: a case study in Kangding County, Southwestern China. Water 10:80
Acknowledgements
We are grateful to Science and Technology Development Fund (STDF), Egypt, and Freie Universität berlin (Germany) for providing support to carry out this study (The scientific project No. 12354).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Said, I., Merz, C., Salman, S.A.ER. et al. Identification of hydrochemical processes using multivariate statistics in a complex aquifer system of Sohag region, Egypt. Environ Earth Sci 79, 169 (2020). https://doi.org/10.1007/s12665-020-08913-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-020-08913-8