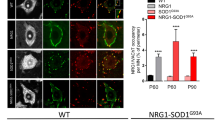
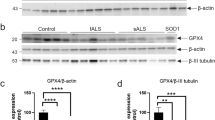
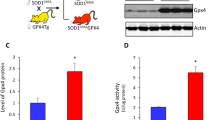
Abstract
The degeneration and death of motor neurons lead to motor neuron diseases such as amyotrophic lateral sclerosis (ALS). Although the exact mechanism by which motor neuron degeneration occurs is not well understood, emerging evidence implicates the involvement of ferroptosis, an iron-dependent oxidative mode of cell death. We reported previously that treating Gpx4NIKO mice with tamoxifen to ablate the ferroptosis regulator glutathione peroxidase 4 (GPX4) in neurons produces a severe paralytic model resembling an accelerated form of ALS that appears to be caused by ferroptotic cell death of spinal motor neurons. In this study, in support of the role of ferroptosis in this model, we found that the paralytic symptoms and spinal motor neuron death of Gpx4NIKO mice were attenuated by a chemical inhibitor of ferroptosis. In addition, we observed that the paralytic symptoms of Gpx4NIKO mice were malleable and could be tapered by lowering the dose of tamoxifen, allowing for the generation of a mild paralytic model without a rapid onset of death. We further used both models to evaluate mitochondrial reactive oxygen species (mtROS) in the ferroptosis of spinal motor neurons and showed that overexpression of peroxiredoxin 3, a mitochondrial antioxidant defense enzyme, ameliorated symptoms of the mild but not the severe model of the Gpx4NIKO mice. Our results thus indicate that the Gpx4NIKO mouse is a versatile model for testing interventions that target ferroptotic death of spinal motor neurons in vivo.





Similar content being viewed by others
Availability of Data and Material
Available upon request.
References
Abdalkader M, Lampinen R, Kanninen KM, Malm TM, Liddell JR (2018) Targeting Nrf2 to suppress ferroptosis and mitochondrial dysfunction in neurodegeneration. Front Neurosci 12:1–9. https://doi.org/10.3389/fnins.2018.00466
Angeli JPF, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch A, Eggenhofer E, Basavarajappa D, Radmark O, Kobayashi S, Seibt T, Beck H, Neff F, Esposito I, Wanke R, Forster H, Yefremova O, Heinrichmeyer M, Bornkamm GW, Geissler EK, Thomas SB, Stockwell BR, O’Donnell VB, Kagan VE, Schick JA, Conrad M (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16:1180–1191. https://doi.org/10.1038/ncb3064
Barber SC, Shaw PJ (2010) Oxidative stress in ALS: key role in motor neuron injury and therapeutic target. Free Radic Biol Med 48:629–641. https://doi.org/10.1016/j.freeradbiomed.2009.11.018
Battaglia AM, Chirillo R, Aversa I, Sacco A, Costanzo F, Biamonte F (2020) Ferroptosis and cancer: mitochondria meet the “iron maiden” cell death. Cells 9:1–26. https://doi.org/10.3390/cells9061505
Chen L, Hambright WS, Na R, Ran Q (2015) Ablation of the ferroptosis inhibitor glutathione peroxidase 4 in neurons results in rapid motor neuron degeneration and paralysis. J Biol Chem 290:28097–28106. https://doi.org/10.1074/jbc.M115.680090
Chen L, Na R, Gu M, Salmon AB, Liu Y, Liang H, Qi W, Van Remmen H, Richardson A, Ran Q (2008) Reduction of mitochondrial H2O2 by overexpressing peroxiredoxin 3 improves glucose tolerance in mice. Aging Cell 7:866–878. https://doi.org/10.1111/j.1474-9726.2008.00432.x
Chen L, Na R, McLane KD, Thompson CS, Gao J, Wang X, Ran Q (2021) Overexpression of ferroptosis defense enzyme Gpx4 retards motor neuron disease of SOD1G93A mice. Sci Rep 11:1–13. https://doi.org/10.1038/s41598-021-92369-8
Devos D, Moreau C, Kyheng M, Garçon G, Rolland AS, Blasco H, Gelé P, Lenglet TT, Veyrat-Durebex C, Corcia P, Dutheil M, Bede P, Jeromin A, Oeckl P, Otto M, Meininger V, Danel-Brunaud V, Devedjian J, Duce JA, Pradat PF (2019) A ferroptosis-based panel of prognostic biomarkers for Amyotrophic Lateral Sclerosis. Sci Rep 9:1–6. https://doi.org/10.1038/s41598-019-39739-5
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS, Morrison B III, B.R. Stockwell BR, (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149:1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Do VB, Gouel F, Jonneaux A, Timmerman K, Gele P, Petrault M, Bastide M, Laloux C, Moreau C, Bordet R, Devos D, Devedjian JC (2016) Ferroptosis, a newly characterized form of cell death in Parkinson’s disease that is regulated by PKC. Neurobiol Dis 94:169–178. https://doi.org/10.1016/j.nbd.2016.05.011
Drey M, Grosch C, Neuwirth C, Bauer JM, Sieber CC (2013) The Motor Unit Number Index (MUNIX) in sarcopenic patients. Exp Gerontol 48:381–384. https://doi.org/10.1016/j.exger.2013.01.011
Gordon PH (2013) Amyotrophic Lateral Sclerosis: An update for 2013 Clinical features, pathophysiology, management and therapeutic trials. Aging Dis 4:295–310. https://doi.org/10.14336/2FAD.2013.0400295
Gordon PH, Moore DH, Miller RG, Florence JM, Verheijde JL, Doorish C, Hilton JF, Spitalny GM, MacArthur RB, Mitsumoto H et al (2007) Efficacy of minocycline in patients with amyotrophic lateral sclerosis: a phase III randomised trial. Lancet Neurol 6:1045–1053. https://doi.org/10.1016/S1474-4422(07)70270-3
Gurney ME, Pu H, Chiu AY, Dal Canto MC, Polchow CY, Alexander DD et al (1994) Motor neuron degeneration in mice that express a human Cu, Zn superoxide dismutase mutation. Science 264:1772–1775. https://doi.org/10.1126/science.8209258
Hall ED, Andrus PK, Oostveen JA, Fleck TJ, Gurney ME (1998) Relationship of oxygen radical-induced lipid peroxidative damage to disease onset and progression in a transgenic model of familial ALS. J Neurosci Res 53:66–77. https://doi.org/10.1002/(SICI)1097-4547(19980701)53:1%3C66::AID-JNR7%3E3.0.CO;2-H
Heimer-McGinn V, Young P (2011) Efficient inducible Pan-neuronal cre-mediated recombination in SLICK-H transgenic mice. Genesis 49:942–949. https://doi.org/10.1002/dvg.20777
Kiernan MC, Vucic S, Talbot K, McDermott CJ, Hardiman O, Shefner JM, Al-Chalabi A, Huynh W, Cudkowicz M, Talman P, Van den Berg LH, Dharmadasa T, Wicks P, Reilly C, Turner MR (2021) Improving clinical trial outcomes in amyotrophic lateral sclerosis. Nat Rev Neurol 17:104–118. https://doi.org/10.1038/s41582-020-00434-z
Linkermann A, Skouta R, Himmerkus N, Mulay SR, Dewitz C, De ZF, Prokai A, Zuchtriegel G, Krombach F, Welz PS, Weinlich R, Vanden Berghe T, Vandenabeele P, Pasparakis M, Bleich M, Weinberg JM, Reichel CA, Brasen JH, Kunzendorf U, Anders HJ, Stockwell BR, Green DR, Krautwald S (2014) Synchronized renal tubular cell death involves ferroptosis. Proc Natl Acad Sci USA 111:16836–16841. https://doi.org/10.1073/pnas.1415518111
Miana-Mena FJ, Muñoz MJ, Yagüe G, Mendez M, Moreno M, Ciriza J, Zaragoza P, Osta R (2005) Optimal methods to characterize the G93A mouse model of ALS. Amyotroph Lateral Scler Frontotemporal Degener 6:55–62. https://doi.org/10.1080/14660820510026162
Pedersen WA, Fu W, Keller JN, Markesbery WR, Appel S, Smith RG, Kasarskis E, Mattson MP (1998) Protein modification by the lipid peroxidation product 4-hydroxynonenal in the spinal cords of amyotrophic lateral sclerosis patients. Ann Neurol 44:819–824. https://doi.org/10.1002/ana.410440518
Perluigi M, Poon HF, Hensley K, Pierce WM, Klein JB, Calabrese V, De Marco C, Butterfield DA (2005) Proteomic analysis of 4-hydroxy-2-nonenal-modified proteins in G93A-SOD1 transgenic mice–a model of familial amyotrophic lateral sclerosis. Free Radic Biol Med 38:960–968. https://doi.org/10.1016/j.freeradbiomed.2004.12.021
Reinert RB, Kantz J, Misfeldt AA, Poffenberger G, Gannon M, Brissova M, Powers AC (2012) Tamoxifen-induced Cre-loxP recombination is prolonged in pancreatic islets of adult mice. PLoS ONE 7:e33529. https://doi.org/10.1371/journal.pone.0033529
Simpson EP, Henry YK, Henkel JS, Smith RG, Appel SH (2004) Increased lipid peroxidation in sera of ALS patients: a potential biomarker of disease burden. Neurology 62:1758–1765. https://doi.org/10.1212/WNL.62.10.1758
Stephenson J, Amor S (2017) Modelling amyotrophic lateral sclerosis in mice. Drug Discov Today Dis Models 25–26:35–44. https://doi.org/10.3389/fnmol.2020.00093
Weydt P, Hong SY, Kliot M, Möller T (2003) Assessing disease onset and progression in the SOD1 mouse model of ALS. NeuroReport 14:1051–1054. https://doi.org/10.1097/01.wnr.0000073685.00308.89
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji AF, Clish CB, Brown LM, Girotti AW, Cornish VW, Schreiber SL, Stockwell BR (2014) Regulation of ferroptotic cancer cell death by GPX4. Cell 156:317–331. https://doi.org/10.1016/j.cell.2013.12.010
Yang WS, Stockwell BR (2016) Ferroptosis: death by lipid peroxidation. Trends Cell Biol 26:165–176. https://doi.org/10.1016/j.tcb.2015.10.014
Yoo SE, Chen L, Na R, Liu Y, Rios C, Van RH, Richardson A, Ran Q (2012) Gpx4 ablation in adult mice results in a lethal phenotype accompanied by neuronal loss in brain. Free Radic Biol Med 52:1820–1827. https://doi.org/10.1016/j.freeradbiomed.2012.02.043
Zhang H, Go YM, Jones DP (2007) Mitochondrial thioredoxin-2/peroxiredoxin-3 system functions in parallel with mitochondrial GSH system in protection against oxidative stress. Arch Biochem Biophys 465:119–126. https://doi.org/10.1016/j.abb.2007.05.001
Funding
This work was supported in part by Merit Review Award I01 BX003507 from the United States (US) Department of Veterans Affairs Biomedical Laboratory Research and Development Program and by an award from Owens Foundation San Antonio to Q.R. Q.R. is also supported by grant AG064078 from NIA, NIH.
Author information
Authors and Affiliations
Contributions
R.C.E., L.C., R.N., K.Y., performed experiments; R.C.E. and Q.R. wrote the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Procedures for handling mice in this study were reviewed and approved by the Institutional Animal Care and Use Committees of the University of Texas Health San Antonio and the Audie Murphy Memorial Veterans Hospital, South Texas Veterans Health Care System. All methods were performed in accordance with the relevant guidelines and regulations.
Consent for Publication
All authors have given their permission for publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Evans, R.C., Chen, L., Na, R. et al. The Gpx4NIKO Mouse Is a Versatile Model for Testing Interventions Targeting Ferroptotic Cell Death of Spinal Motor Neurons. Neurotox Res 40, 373–383 (2022). https://doi.org/10.1007/s12640-021-00469-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00469-0