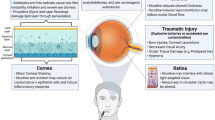
Abstract
Nitrous oxide (N2O), also known as “laughing gas,” is a colorless, nonirritating gas. Clinically, it is widely used as an inhaled anesthetic, analgesic, and anxiolytic. In recent years, recreational abuse of N2O has become increasingly common, especially among young adults and adolescents, but many of them lack awareness of the possible side effects associated with this drug. N2O abuse can damage multiple systems, especially the nervous system, but the exact mechanism of N2O toxicity remains controversial. At present, an increasing number of cases of nervous system damage caused by N2O abuse have been reported both at home and abroad. Discontinuation of N2O use and timely supplementation with vitamin B12 are essential for a good prognosis. Long-term abuse without timely treatment will eventually lead to irreversible neurological damage. In this article, we discuss the epidemiology of N2O abuse, neurotoxicity mechanisms, clinical manifestations, relevant auxiliary examinations, treatments, and prognosis to improve social awareness of N2O exposure risk, especially among users and clinicians.

Similar content being viewed by others
References
Ahn SC, Brown AW (2005) Cobalamin deficiency and subacute combined degeneration after nitrous oxide anesthesia: a case report. Arch Phys Med Rehabil 86:150–153. https://doi.org/10.1016/j.apmr.2004.01.019
Amiri F, Neghab M, Shouroki FK, Yousefinejad S, Hassanzadeh J (2018) Earlysubclinical hematological changes associated with occupational exposure to high levels of nitrous oxide. Toxics 6https://doi.org/10.3390/toxics6040070
Andres E, Fothergill H, Mecili M (2010) Efficacy of oral cobalamin (vitamin B12) therapy. Expert Opin Pharmacother 11:249–256. https://doi.org/10.1517/14656560903456053
Bahadir A, Reis PG, Erduran E (2014) Oral vitamin B12 treatment is effective for children with nutritional vitamin B12 deficiency. J Paediatr Child Health 50:721–725. https://doi.org/10.1111/jpc.12652
Belliveau JF et al (1991) Clinical pharmacokinetics of 3-day continuous infusion cisplatin and daily bolus 5-fluorouracil. Eur J Clin Pharmacol 40:115–117. https://doi.org/10.1007/bf00315150
Binkerd EF, Kolari OE (1975) The history and use of nitrate and nitrite in the curing of meat. Food Cosmet Toxicol 13:655–661. https://doi.org/10.1016/0015-6264(75)90157-1
Bolaman Z, Kadikoylu G, Yukselen V, Yavasoglu I, Barutca S, Senturk T (2003) Oral versus intramuscular cobalamin treatment in megaloblastic anemia: a single-center, prospective, randomized, open-label study. Clin Ther 25:3124–3134. https://doi.org/10.1016/s0149-2918(03)90096-8
Bor MV, Cetin M, Aytac S, Altay C, Ueland PM, Nexo E (2008) Long term biweekly 1 mg oral vitamin B12 ensures normal hematological parameters, but does not correct all other markers of vitamin B12 deficiency. A study in patients with inherited vitamin B12 deficiency. Haematologica 93:1755–1758. https://doi.org/10.3324/haematol.13122
Briani C, Dalla Torre C, Citton V, Manara R, Pompanin S, Binotto G, Adami F (2013) Cobalamin deficiency: clinical picture and radiological findings. Nutrients 5:4521–4539. https://doi.org/10.3390/nu5114521
Brodsky JB, Cohen EN (1986) Adverse effects of nitrous oxide. Med Toxicol 1:362–374. https://doi.org/10.1007/bf03259849
Butzkueven H, King JO (2000) Nitrous oxide myelopathy in an abuser of whipped cream bulbs. J Clin Neurosci 7:73–75. https://doi.org/10.1054/jocn.1998.0149
Cantrill RC, Oldfield M, van der Westhuyzen J, McLoughlin J (1983) Protein profile of the myelin membrane of the fruit bat Rousettus aegyptiacus. Comp Biochem Physiol B 76:881–884. https://doi.org/10.1016/0305-0491(83)90407-8
Castelli MC, Friedman K, Sherry J, Brazzillo K, Genoble L, Bhargava P, Riley MG (2011) Comparing the efficacy and tolerability of a new daily oral vitamin B12 formulation and intermittent intramuscular vitamin B12 in normalizing low cobalamin levels: a randomized, open-label, parallel-group study. Clin Ther 33:358-371 e352. https://doi.org/10.1016/j.clinthera.2011.03.003
Chen HJ, Huang CS (2016) Nitrous oxide-induced subacute combined degeneration presenting with dystonia and pseudoathetosis: a case report. Acta Neurol Taiwan 25:50–55
Chen T, Zhong N, Jiang H, Zhao M, Chen Z, Sun H (2018) Neuropsychiatric symptoms induced by large doses of nitrous oxide inhalation: a case report. Shanghai Arch Psychiatry 30:56–59. https://doi.org/10.11919/j.issn.1002-0829.217084
Chiang TT, Hung CT, Wang WM, Lee JT, Yang FC (2013) Recreational nitrous oxide abuse-induced vitamin B12 deficiency in a patient presenting with hyperpigmentation of the skin. Case Rep Dermatol 5:186–191. https://doi.org/10.1159/000353623
Chien WH, Huang MC, Chen LY (2020) Psychiatric and other medical manifestations of nitrous oxide abuse: implications from case series. J Clin Psychopharmacol 40:80–83. https://doi.org/10.1097/JCP.0000000000001151
Clarke R, Daly L, Robinson K, Naughten E, Cahalane S, Fowler B, Graham I (1991) Hyperhomocysteinemia: an independent risk factor for vascular disease. N Engl J Med 324:1149–1155. https://doi.org/10.1056/NEJM199104253241701
Deacon R, Purkiss P, Green R, Lumb M, Perry J, Chanarin I (1986) Vitamin B12 neuropathy is not due to failure to methylate myelin basic protein. J Neurol Sci 72:113–117. https://doi.org/10.1016/0022-510x(86)90040-7
Dehring DJ, Gupta B, Peruzzi WT (1991) Postoperative opisthotonus and torticollis after fentanyl, enflurane, and nitrous oxide. Can J Anaesth 38:919–925. https://doi.org/10.1007/bf03036975
den Uil SH, Vermeulen EGJ, Metz R, Rijbroek A, de Vries M (2018) Aortic arch thrombus caused by nitrous oxide abuse. J Vasc Surg Cases Innov Tech 4:80–82. https://doi.org/10.1016/j.jvscit.2018.01.001
Dong X, Ba F, Wang R, Zheng D (2019) Imaging appearance of myelopathy secondary to nitrous oxide abuse: a case report and review of the literature. Int J Neurosci 129:225–229. https://doi.org/10.1080/00207454.2018.1526801
Eaton DK et al (2012) Youth risk behavior surveillance - United States 2011. MMWR Surveill Summ 61:1–162
Ehirim EM, Naughton DP, Petroczi A (2017) No laughing matter: presence, consumption trends, drug awareness, and perceptions of “hippy crack” (nitrous oxide) among young adults in England. Front Psych 8:312. https://doi.org/10.3389/fpsyt.2017.00312
El Otmani H, El Moutawakil B, Moutaouakil F, Gam I, Rafai MA, Slassi I (2007) [Postoperative dementia: toxicity of nitrous oxide]. Encephale 33:95–97. https://doi.org/10.1016/s0013-7006(07)91563-8
Erving HW (1933) The discoverer of anaesthesia: Dr Horace Wells of Hartford Yale. J Biol Med 5:421–430
Fang X, Li W, Gao H, Ma Y, Dong X, Zheng D (2019) Skin hyperpigmentation: a rare presenting symptom of nitrous oxide abuse Clin Toxicol (Phila):1–6 https://doi.org/10.1080/15563650.2019.1665181
Faraci FM, Lentz SR (2004) Hyperhomocysteinemia, oxidative stress, and cerebral vascular dysfunction. Stroke 35:345–347. https://doi.org/10.1161/01.STR.0000115161.10646.67
Felmet K, Robins B, Tilford D, Hayflick SJ (2000) Acute neurologic decompensation in an infant with cobalamin deficiency exposed to nitrous oxide. J Pediatr 137:427–428. https://doi.org/10.1067/mpd.2000.107387
Flippo TS, Holder WD Jr (1993) Neurologic degeneration associated with nitrous oxide anesthesia in patients with vitamin B12 deficiency. Arch Surg 128:1391–1395. https://doi.org/10.1001/archsurg.1993.01420240099018
Garakani A, Jaffe RJ, Savla D, Welch AK, Protin CA, Bryson EO, McDowell DM (2016) Neurologic, psychiatric, and other medical manifestations of nitrous oxide abuse: a systematic review of the case literature. Am J Addict 25:358–369. https://doi.org/10.1111/ajad.12372
Garland EL, Howard MO, Perron BE (2009) Nitrous oxide inhalation among adolescents: prevalence, correlates, and co-occurrence with volatile solvent inhalation. J Psychoactive Drugs 41:337–347. https://doi.org/10.1080/02791072.2009.10399771
Gillman MA, Lichtigfeld FJ (1989) Analgesic nitrous oxide in neuropsychiatry: past, present and future. Int J Neurosci 49:75–81. https://doi.org/10.3109/00207458909087041
Gowing LR, Ali RL, Allsop S, Marsden J, Turf EE, West R, Witton J (2015) Global statistics on addictive behaviours: 2014 status report. Addiction 110:904–919. https://doi.org/10.1111/add.12899
Graber JJ, Sherman FT, Kaufmann H, Kolodny EH, Sathe S (2010) Vitamin B12-responsive severe leukoencephalopathy and autonomic dysfunction in a patient with “normal” serum B12 levels. J Neurol Neurosurg Psych 81:1369–1371. https://doi.org/10.1136/jnnp.2009.178657
Grattan-Smith PJ, Wilcken B, Procopis PG, Wise GA (1997) The neurological syndrome of infantile cobalamin deficiency: developmental regression and involuntary movements. Mov Disord 12:39–46. https://doi.org/10.1002/mds.870120108
Green R (2017) Vitamin B12 deficiency from the perspective of a practicing hematologist. Blood 129:2603–2611. https://doi.org/10.1182/blood-2016-10-569186
Green R et al (2017) Vitamin B12 deficiency. Nat Rev Dis Primers 3:17040. https://doi.org/10.1038/nrdp.2017.40
Green R, Kinsella LJ (1995) Current concepts in the diagnosis of cobalamin deficiency. Neurology 45:1435–1440. https://doi.org/10.1212/wnl.45.8.1435
Hankey GJ, Eikelboom JW, Ho WK, van Bockxmeer FM (2004) Clinical usefulness of plasma homocysteine in vascular disease. Med J Aust 181:314–318
Hannibal L et al (2016) Biomarkers and Algorithms for the Diagnosis of Vitamin B12 Deficiency. Front Mol Biosci 3:27. https://doi.org/10.3389/fmolb.2016.00027
Hathout L, El-Saden S (2011) Nitrous oxide-induced B(1)(2) deficiency myelopathy: Perspectives on the clinical biochemistry of vitamin B(1)(2). J Neurol Sci 301:1–8. https://doi.org/10.1016/j.jns.2010.10.033
Healton EB, Savage DG, Brust JC, Garrett TJ, Lindenbaum J (1991) Neurologic aspects of cobalamin deficiency. Medicine (Baltimore) 70:229–245. https://doi.org/10.1097/00005792-199107000-00001
Hirvioja J, Joutsa J, Wahlsten P, Korpela J (2016) Recurrent paraparesis and death of a patient with ‘whippet’ abuse. Oxf Med Case Reports 2016:41–43. https://doi.org/10.1093/omcr/omw012
Hsu CK, Chen YQ, Lung VZ, His SC, Lo HC, Shyu HY (2012) Myelopathy and polyneuropathy caused by nitrous oxide toxicity: a case report. Am J Emerg Med 30(1016):e1013-1016. https://doi.org/10.1016/j.ajem.2011.05.001
Hutto BR (1997) Folate and cobalamin in psychiatric illness. Compr Psychiatry 38:305–314. https://doi.org/10.1016/s0010-440x(97)90925-1
Hvas AM, Nexo E (2006) Diagnosis and treatment of vitamin B12 deficiency–an update. Haematologica 91:1506–1512
Irwin MG, Trinh T, Yao CL (2009) Occupational exposure to anaesthetic gases: a role for TIVA. Expert Opin Drug Saf 8:473–483. https://doi.org/10.1517/14740330903003778
Jakovljevic M, Vidmar G, Mekjavic IB (2012) Psychomotor function during mild narcosis induced by subanesthetic level of nitrous oxide: individual susceptibility beyond gender effect. Undersea Hyperb Med 39:1067–1074
Jevtovic-Todorovic V, Beals J, Benshoff N, Olney JW (2003) Prolonged exposure to inhalational anesthetic nitrous oxide kills neurons in adult rat brain. Neuroscience 122:609–616. https://doi.org/10.1016/j.neuroscience.2003.07.012
Jevtovic-Todorovic V et al (1998) Nitrous oxide (laughing gas) is an NMDA antagonist, neuroprotectant and neurotoxin. Nat Med 4:460–463. https://doi.org/10.1038/nm0498-460
Ji R, Xie Z, Wang K, Zheng X (2018) Glucocorticoid treatment of myeloneuropathy induced by nitrous oxide toxicity. Neurol India 66:1167–1169. https://doi.org/10.4103/0028-3886.237029
Johnson EO, Schutz CG, Anthony JC, Ensminger ME (1995) Inhalants to heroin: a prospective analysis from adolescence to adulthood. Drug Alcohol Depend 40:159–164. https://doi.org/10.1016/0376-8716(95)01201-x
Johnson K, Mikhail P, Kim MG, Bosco A, Huynh W (2018) Recreational nitrous oxide-associated neurotoxicity. J Neurol Neurosurg Psych 89:897–898. https://doi.org/10.1136/jnnp-2017-317768
Kaar SJ, Ferris J, Waldron J, Devaney M, Ramsey J, Winstock AR (2016) Up: The rise of nitrous oxide abuse An international survey of contemporary nitrous oxide use. J Psychopharmacol 30:395–401. https://doi.org/10.1177/0269881116632375
Keddie S et al (2018) No laughing matter: subacute degeneration of the spinal cord due to nitrous oxide inhalation. J Neurol 265:1089–1095. https://doi.org/10.1007/s00415-018-8801-3
Kondo H et al (1981) Nitrous oxide has multiple deleterious effects on cobalamin metabolism and causes decreases in activities of both mammalian cobalamin-dependent enzymes in rats. J Clin Invest 67:1270–1283. https://doi.org/10.1172/jci110155
Kumar A, Singh AK (2009) Teaching NeuroImage: Inverted V sign in subacute combined degeneration of spinal cord. Neurology 72:e4. https://doi.org/10.1212/01.wnl.0000338663.59433.9c
Kuzminski AM, Del Giacco EJ, Allen RH, Stabler SP, Lindenbaum J (1998) Effective treatment of cobalamin deficiency with oral cobalamin. Blood 92:1191–1198
Lan SY et al (2019) Recreational nitrous oxide abuse related subacute combined degeneration of the spinal cord in adolescents - A case series and literature review. Brain Dev 41:428–435. https://doi.org/10.1016/j.braindev.2018.12.003
Li HT, Chu CC, Chang KH, Liao MF, Chang HS, Kuo HC, Lyu RK (2016) Clinical and electrodiagnostic characteristics of nitrous oxide-induced neuropathy in Taiwan. Clin Neurophysiol 127:3288–3293. https://doi.org/10.1016/j.clinph.2016.08.005
Lichtigfeld FJ, Gillman MA (2003) Another marker for different types of depression. Int J Neuropsychopharmacol 6:91–92. https://doi.org/10.1017/S1461145703003286
Lim CC (2011) Neuroimaging in postinfectious demyelination and nutritional disorders of the central nervous system. Neuroimaging Clin N Am 21:843–858. https://doi.org/10.1016/j.nic.2011.08.001
Lin RJ, Chen HF, Chang YC, Su JJ (2011) Subacute combined degeneration caused by nitrous oxide intoxication: case reports. Acta Neurol Taiwan 20:129–137
Lindenbaum J et al (1988) Neuropsychiatric disorders caused by cobalamin deficiency in the absence of anemia or macrocytosis. N Engl J Med 318:1720–1728. https://doi.org/10.1056/NEJM198806303182604
Loscalzo J (2002) Homocysteine and dementias. N Engl J Med 346:466–468. https://doi.org/10.1056/NEJM200202143460702
Louis-Ferdinand RT (1994) Myelotoxic, neurotoxic and reproductive adverse effects of nitrous oxide. Adverse Drug React Toxicol Rev 13:193–206
Mancke F, Kaklauskaite G, Kollmer J, Weiler M (2016) Psychiatric comorbidities in a young man with subacute myelopathy induced by abusive nitrous oxide consumption: a case report. Subst Abuse Rehabil 7:155–159. https://doi.org/10.2147/SAR.S114404
Marie RM, Le Biez E, Busson P, Schaeffer S, Boiteau L, Dupuy B, Viader F (2000) Nitrous oxide anesthesia-associated myelopathy. Arch Neurol 57:380–382. https://doi.org/10.1001/archneur.57.3.380
Marsolek MR, White NC, Litovitz TL (2010) Inhalant abuse: monitoring trends by using poison control data, 1993–2008. Pediatrics 125:906–913. https://doi.org/10.1542/peds.2009-2080
Massey TH, Pickersgill TT, K JP (2016) Nitrous oxide misuse and vitamin B12 deficiency. BMJ Case Rep 2016 https://doi.org/10.1136/bcr-2016-215728
Mayer EL, Jacobsen DW, Robinson K (1996) Homocysteine and coronary atherosclerosis. J Am Coll Cardiol 27:517–527. https://doi.org/10.1016/0735-1097(95)00508-0
Metz J (1992) Cobalamin deficiency and the pathogenesis of nervous system disease. Annu Rev Nutr 12:59–79. https://doi.org/10.1146/annurev.nu.12.070192.000423
Morris N, Lynch K, Greenberg SA (2015) Severe motor neuropathy or neuronopathy due to nitrous oxide toxicity after correction of vitamin B12 deficiency. Muscle Nerve 51:614–616. https://doi.org/10.1002/mus.24482
Myles PS, Chan MT, Leslie K, Peyton P, Paech M, Forbes A (2008) Effect of nitrous oxide on plasma homocysteine and folate in patients undergoing major surgery. Br J Anaesth 100:780–786. https://doi.org/10.1093/bja/aen085
Nagele P et al (2015) Nitrous oxide for treatment-resistant major depression: a proof-of-concept trial. Biol Psychiatry 78:10–18. https://doi.org/10.1016/j.biopsych.2014.11.016
Nasr SZ, Nasrallah AI, Abdulghani M, Sweet SC (2018) The impact of conventional and nonconventional inhalants on children and adolescents. Pediatr Pulmonol 53:391–399. https://doi.org/10.1002/ppul.23836
Ng J, O’Grady G, Pettit T, Frith R (2003) Nitrous oxide use in first-year students at Auckland University. Lancet 361:1349–1350. https://doi.org/10.1016/S0140-6736(03)13045-0
Nouri A, Patel K, Montejo J, Nasser R, Gimbel DA, Sciubba DM, Cheng JS (2019) The Role of Vitamin B12 in the Management and Optimization of Treatment in Patients With Degenerative Cervical Myelopathy. Global Spine J 9:331–337. https://doi.org/10.1177/2192568218758633
Oussalah A et al. (2019) Global burden related to nitrous oxide exposure in medical and recreational settings: a systematic review and individual patient data meta-analysis. J Clin Med 8 https://doi.org/10.3390/jcm8040551
Ozer EA, Turker M, Bakiler AR, Yaprak I, Ozturk C (2001) Involuntary movements in infantile cobalamin deficiency appearing after treatment. Pediatr Neurol 25:81–83. https://doi.org/10.1016/s0887-8994(01)00289-2
Pandey S, Kalita J, Misra UK (2004) A sequential study of visual evoked potential in patients with vitamin B12 deficiency neurological syndrome. Clin Neurophysiol 115:914–918. https://doi.org/10.1016/j.clinph.2003.11.013
Peng TI, Jou MJ (2010) Oxidative stress caused by mitochondrial calcium overload. Ann N Y Acad Sci 1201:183–188. https://doi.org/10.1111/j.1749-6632.2010.05634.x
Pratt DN, Patterson KC, Quin K (2019) Venous thrombosis after nitrous oxide abuse, a case report. J Thromb Thrombolysis https://doi.org/10.1007/s11239-019-02010-9
Pugliese RS, Slagle EJ, Oettinger GR, Neuburger KJ, Ambrose TM (2015) Subacute combined degeneration of the spinal cord in a patient abusing nitrous oxide and self-medicating with cyanocobalamin. Am J Health Syst Pharm 72:952–957. https://doi.org/10.2146/ajhp140583
Quere I et al (2002) Red blood cell methylfolate and plasma homocysteine as risk factors for venous thromboembolism: a matched case-control study. Lancet 359:747–752. https://doi.org/10.1016/S0140-6736(02)07876-5
Reggio E, Lanzafame S, Giliberto C, Nastasi L, Nicoletti A, Zappia M (2013) Subacute combined degeneration of the spinal cord presenting with pseudoathetosis of the upper limbs. Eur J Neurol 20:e26-27. https://doi.org/10.1111/ene.12008
Reynolds E (2006) Vitamin B12, folic acid, and the nervous system. Lancet Neurol 5:949–960. https://doi.org/10.1016/S1474-4422(06)70598-1
Reynolds IJ, Hastings TG (1995) Glutamate induces the production of reactive oxygen species in cultured forebrain neurons following NMDA receptor activation. J Neurosci 15:3318–3327
Richardson PG (2010) Peripheral neuropathy following nitrous oxide abuse. Emerg Med Australas 22:88–90. https://doi.org/10.1111/j.1742-6723.2009.01262.x
Sahenk Z, Mendell JR, Couri D, Nachtman J (1978) Polyneuropathy from inhalation of N2O cartridges through a whipped-cream dispenser. Neurology 28:485–487. https://doi.org/10.1212/wnl.28.5.485
Sanders RD, Weimann J, Maze M (2008) Biologic effects of nitrous oxide: a mechanistic and toxicologic review. Anesthesiology 109:707–722. https://doi.org/10.1097/ALN.0b013e3181870a17
Savage S, Ma D (2014) The neurotoxicity of nitrous oxide: the facts and “putative” mechanisms. Brain Sci 4:73–90. https://doi.org/10.3390/brainsci4010073
Scalabrino G (2005) Cobalamin (vitamin B(12)) in subacute combined degeneration and beyond: traditional interpretations and novel theories. Exp Neurol 192:463–479. https://doi.org/10.1016/j.expneurol.2004.12.020
Scalabrino G (2009) The multi-faceted basis of vitamin B12 (cobalamin) neurotrophism in adult central nervous system: Lessons learned from its deficiency. Prog Neurobiol 88:203–220. https://doi.org/10.1016/j.pneurobio.2009.04.004
Scalabrino G et al (2004) High tumor necrosis factor-alpha [corrected] levels in cerebrospinal fluid of cobalamin-deficient patients. Ann Neurol 56:886–890. https://doi.org/10.1002/ana.20325
Scalabrino G, Corsi MM, Veber D, Buccellato FR, Pravettoni G, Manfridi A, Magni P (2002) Cobalamin (vitamin B(12)) positively regulates interleukin-6 levels in rat cerebrospinal fluid. J Neuroimmunol 127:37–43. https://doi.org/10.1016/s0165-5728(02)00095-4
Scalabrino G, Mutti E, Veber D, Aloe L, Corsi MM, Galbiati S, Tredici G (2006) Increased spinal cord NGF levels in rats with cobalamin (vitamin B12) deficiency. Neurosci Lett 396:153–158. https://doi.org/10.1016/j.neulet.2005.11.029
Scalabrino G, Tredici G, Buccellato FR, Manfridi A (2000) Further evidence for the involvement of epidermal growth factor in the signaling pathway of vitamin B12 (cobalamin) in the rat central nervous system. J Neuropathol Exp Neurol 59:808–814. https://doi.org/10.1093/jnen/59.9.808
Scalabrino G, Veber D, Mutti E (2008) Experimental and clinical evidence of the role of cytokines and growth factors in the pathogenesis of acquired cobalamin-deficient leukoneuropathy. Brain Res Rev 59:42–54. https://doi.org/10.1016/j.brainresrev.2008.05.001
Shen Q, Lu H, Wang H, Xu Y (2019) Acute cognitive disorder as the initial manifestation of nitrous oxide abusing: a case report. Neurol Sci. https://doi.org/10.1007/s10072-019-04183-w
Singer MA, Lazaridis C, Nations SP, Wolfe GI (2008) Reversible nitrous oxide-induced myeloneuropathy with pernicious anemia: case report and literature review. Muscle Nerve 37:125–129. https://doi.org/10.1002/mus.20840
Song Z, Uriarte S, Sahoo R, Chen T, Barve S, Hill D, McClain C (2005) S-adenosylmethionine (SAMe) modulates interleukin-10 and interleukin-6, but not TNF, production via the adenosine (A2) receptor. Biochim Biophys Acta 1743:205–213. https://doi.org/10.1016/j.bbamcr.2004.12.001
Stabler SP (2013) Clinical practice. Vitamin B12 deficiency. N Engl J Med 368:149–160. https://doi.org/10.1056/NEJMcp1113996
Stacy CB, Di Rocco A, Gould RJ (1992) Methionine in the treatment of nitrous-oxide-induced neuropathy and myeloneuropathy. J Neurol 239:401–403. https://doi.org/10.1007/bf00812159
Sun W, Liao JP, Hu Y, Zhang W, Ma J, Wang GF (2019) Pulmonary embolism and deep vein thrombosis caused by nitrous oxide abuse: A case report. World J Clin Cases 7:4057–4062. https://doi.org/10.12998/wjcc.v7.i23.4057
Tani J, Weng HY, Chen HJ, Chang TS, Sung JY, Lin CS (2019) Elucidating unique axonal dysfunction between nitrous oxide abuse and vitamin B12 deficiency. Front Neurol 10:704. https://doi.org/10.3389/fneur.2019.00704
Thompson AG, Leite MI, Lunn MP, Bennett DL (2015) Whippits, nitrous oxide and the dangers of legal highs. Pract Neurol 15:207–209. https://doi.org/10.1136/practneurol-2014-001071
Tuan TA et al (2020) The clinical and subclinical features of spinal cord injury on magnetic resonance imaging of patients with N2O intoxication. Neurol Int 12:8652. https://doi.org/10.4081/ni.2020.8652
van Amsterdam J, Nabben T, van den Brink W (2015) Recreational nitrous oxide use: prevalence and risks. Regul Toxicol Pharmacol 73:790–796. https://doi.org/10.1016/j.yrtph.2015.10.017
Vasconcelos OM, Poehm EH, McCarter RJ, Campbell WW, Quezado ZM (2006) Potential outcome factors in subacute combined degeneration: review of observational studies. J Gen Intern Med 21:1063–1068. https://doi.org/10.1111/j.1525-1497.2006.00525.x
Veber D et al (2006) Increased levels of the CD40:CD40 ligand dyad in the cerebrospinal fluid of rats with vitamin B12(cobalamin)-deficient central neuropathy. J Neuroimmunol 176:24–33. https://doi.org/10.1016/j.jneuroim.2006.04.002
Vidal-Alaball J et al (2005) Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency Cochrane Database. Syst Rev:CD004655 https://doi.org/10.1002/14651858.CD004655.pub2
Waclawik AJ, Luzzio CC, Juhasz-Pocsine K, Hamilton V (2003) Myeloneuropathy from nitrous oxide abuse: unusually high methylmalonic acid and homocysteine levels. WMJ 102:43–45
Wagner SA, Clark MA, Wesche DL, Doedens DJ, Lloyd AW (1992) Asphyxial deaths from the recreational use of nitrous oxide. J Forensic Sci 37:1008–1015
Wald DS, Law M, Morris JK (2002) Homocysteine and cardiovascular disease: evidence on causality from a meta-analysis. BMJ 325:1202. https://doi.org/10.1136/bmj.325.7374.1202
Wang H, Li L, Qin LL, Song Y, Vidal-Alaball J, Liu TH (2018) Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency. Cochrane Database Syst Rev 3:CD004655. https://doi.org/10.1002/14651858.CD004655.pub3
Watson WH, Zhao Y, Chawla RK (1999) S-adenosylmethionine attenuates the lipopolysaccharide-induced expression of the gene for tumour necrosis factor alpha. Biochem J 342(Pt 1):21–25
Weimann J (2003) Toxicity of nitrous oxide. Best Pract Res Clin Anaesthesiol 17:47–61. https://doi.org/10.1053/bean.2002.0264
West JB (2014) Humphry Davy, nitrous oxide, the Pneumatic Institution, and the Royal Institution. Am J Physiol Lung Cell Mol Physiol 307:L661-667. https://doi.org/10.1152/ajplung.00206.2014
Wijesekera NT, Davagnanam I, Miszkiel K (2009) Subacute combined cord degeneration: a rare complication of nitrous oxide misuse A case report. Neuroradiol J 22:194–197. https://doi.org/10.1177/197140090902200210
Winstock AR, Ferris JA (2019) Nitrous oxide causes peripheral neuropathy in a dose dependent manner among recreational users. J Psychopharmacol:269881119882532 https://doi.org/10.1177/0269881119882532
Wu LT, Pilowsky DJ, Schlenger WE (2004) Inhalant abuse and dependence among adolescents in the United States. J Am Acad Child Adolesc Psych 43:1206–1214. https://doi.org/10.1097/01.chi.0000134491.42807.a3
Wu LT, Ringwalt CL (2006) Inhalant use and disorders among adults in the United States. Drug Alcohol Depend 85:1–11. https://doi.org/10.1016/j.drugalcdep.2006.01.017
Wu MS, Hsu YD, Lin JC, Chen SC, Lee JT (2007) Spinal myoclonus in subacute combined degeneration caused by nitrous oxide intoxication. Acta Neurol Taiwan 16:102–105
Yagiela JA (1991) Health hazards and nitrous oxide: a time for reappraisal. Anesth Prog 38:1–11
Yuan JL, Wang SK, Jiang T, Hu WL (2017) Nitrous oxide induced subacute combined degeneration with longitudinally extensive myelopathy with inverted V-sign on spinal MRI: a case report and literature review. BMC Neurol 17:222. https://doi.org/10.1186/s12883-017-0990-3
Zheng D, Ba F, Bi G, Guo Y, Gao Y, Li W (2019) The sharp rise of neurological disorders associated with recreational nitrous oxide use in China: a single-center experience and a brief review of Chinese literature. J Neurol. https://doi.org/10.1007/s00415-019-09600-w
Zheng D, Ba F, Bi G, Guo Y, Gao Y, Li W (2020) The sharp rise of neurological disorders associated with recreational nitrous oxide use in China: a single-center experience and a brief review of Chinese literature. J Neurol 267:422–429. https://doi.org/10.1007/s00415-019-09600-w
Funding
This work was supported by Key Technology Research and Development Program of Shandong (No. 2015 GGH318020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xiang, Y., Li, L., Ma, X. et al. Recreational Nitrous Oxide Abuse: Prevalence, Neurotoxicity, and Treatment. Neurotox Res 39, 975–985 (2021). https://doi.org/10.1007/s12640-021-00352-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00352-y