Abstract
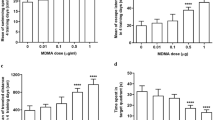
When burning crack cocaine, the pyrolysis of cocaine generates anhydroecgonine methyl ester (AEME). AEME has been shown to be highly neurotoxic but its effects on cognitive function and oxidative stress are still unknown. Thus, this study investigated the effects of AEME on spatial working memory and on parameters of oxidative stress in the prefrontal cortex, hippocampus, and striatum. First, 18 well-trained rats in 8-arm radial maze (8-RM) procedures received acute intracerebroventricular (icv) administration of AEME at doses of 10, 32, or 100 μg or saline (SAL) in a counterbalanced order and were tested 5 min later in 1-h delayed tasks in the 8-RM. Secondly, separated animals received acute icv administration of AEME at doses of 10 (n = 5), 32 (n = 5), or 100 μg (n = 5) or SAL (n = 5) for analysis of advanced oxidation protein products, thiobarbituric acid, catalase, glutathione peroxidase, and superoxide dismutase. A higher number of errors were seen in the 1-h post-delay performance after AEME 32 μg and AEME 100 μg when compared to SAL. In the striatum, animals receiving AEME 100 μg icv showed increased advanced oxidation protein products levels when compared to 10 μg, and also showed increased activity of glutathione peroxidase enzyme when compared to SAL but also comparing to AEME 32 μg and AEME 10 μg. These results showed that AEME impairs long-term spatial working memory and also induces greater protein oxidation and increased levels of antioxidant enzymes in the striatum.





Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Altunoglu E et al (2015) Ischemia-modified albumin and advanced oxidation protein products as potential biomarkers of protein oxidation in Alzheimer’s disease. Geriatr Gerontol Int 15:872–880. https://doi.org/10.1111/ggi.12361
Baddeley A, Della Sala S (1996) Working memory and executive control. Philos Trans R Soc Lond B Biol Sci 351:1397–1403; discussion 1403–1394. https://doi.org/10.1098/rstb.1996.0123
Baier B, Karnath HO, Dieterich M, Birklein F, Heinze C, Muller NG (2010) Keeping memory clear and stable--the contribution of human basal ganglia and prefrontal cortex to working memory. J Neurosci 30:9788–9792. https://doi.org/10.1523/JNEUROSCI.1513-10.2010
Bainbridge NK, Koselke LR, Jeon J, Bailey KR, Wess J, Crawley JN, Wrenn CC (2008) Learning and memory impairments in a congenic C57BL/6 strain of mice that lacks the M2 muscarinic acetylcholine receptor subtype. Behav Brain Res 190:50. https://doi.org/10.1016/j.bbr.2008.02.001
Bannister JV, Calabrese L (1987) Assays for superoxide dismutase. Methods Biochem Anal 32:279–312
Bashkatova V, Meunier J, Maurice T, Vanin A (2005) Memory impairments and oxidative stress in the hippocampus of in-utero cocaine-exposed rats. Neuroreport 16:1217–1221
Blot K et al (2015) Modulation of hippocampus-prefrontal cortex synaptic transmission and disruption of executive cognitive functions by MK-801. Cereb Cortex 25:1348–1361. https://doi.org/10.1093/cercor/bht329
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bunce JG, Sabolek HR, Chrobak JJ (2003) Intraseptal infusion of oxotremorine impairs memory in a delayed-non-match-to-sample radial maze task. Neuroscience 121:259–267
Buresova O, Bures J (1982) Radial maze as a tool for assessing the effect of drugs on the working memory of rats. Psychopharmacology (Berl) 77:268–271
Canli T, Cook RG, Miczek KA (1990) Opiate antagonists enhance the working memory of rats in the radial maze. Pharmacol Biochem Behav 36:521–525
Chan W et al (2017) Mice lacking M1 and M3 muscarinic acetylcholine receptors have impaired odor discrimination and learning. Front Synaptic Neurosci 9:4. https://doi.org/10.3389/fnsyn.2017.00004
de Souza Custodio JC, Martins CW, Lugon MD, Fregni F, Nakamura-Palacios EM (2013) Epidural direct current stimulation over the left medial prefrontal cortex facilitates spatial working memory performance in rats. Brain Stimul 6:261–269. https://doi.org/10.1016/j.brs.2012.07.004
Dietrich JB, Mangeol A, Revel MO, Burgun C, Aunis D, Zwiller J (2005) Acute or repeated cocaine administration generates reactive oxygen species and induces antioxidant enzyme activity in dopaminergic rat brain structures. Neuropharmacology 48:965–974. https://doi.org/10.1016/j.neuropharm.2005.01.018
Doeller CF, Opitz B, Krick CM, Mecklinger A, Reith W (2005) Prefrontal-hippocampal dynamics involved in learning regularities across episodes. Cereb Cortex 15:1123–1133. https://doi.org/10.1093/cercor/bhh211
Duailibi LB, Ribeiro M, Laranjeira R (2008) Profile of cocaine and crack users in Brazil. Cad Saude Publica 24(Suppl 4):s545–s557
Eckart C, Wozniak-Kwasniewska A, Herweg NA, Fuentemilla L, Bunzeck N (2016) Acetylcholine modulates human working memory and subsequent familiarity based recognition via alpha oscillations. Neuroimage 137:61–69. https://doi.org/10.1016/j.neuroimage.2016.05.049
Ellis KA, Nathan PJ (2001) The pharmacology of human working memory. Int J Neuropsychopharmacol 4:299–313. https://doi.org/10.1017/S1461145701002541
Erzouki HK, Allen AC, Newman AH, Goldberg SR, Schindler CW (1995) Effects of cocaine, cocaine metabolites and cocaine pyrolysis products on the hindbrain cardiac and respiratory centers of the rabbit. Life Sci 57:1861–1868
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186:407–421
Falck RS, Wang J, Carlson RG (2007) Crack cocaine trajectories among users in a midwestern American city. Addiction 102:1421–1431. https://doi.org/10.1111/j.1360-0443.2007.01915.x
Flood JF, Landry DW, Jarvik ME (1981) Cholinergic receptor interactions and their effects on long-term memory processing. Brain Res 215:177–185
Floresco SB, Seamans JK, Phillips AG (1997) Selective roles for hippocampal, prefrontal cortical, and ventral striatal circuits in radial-arm maze tasks with or without a delay. J Neurosci 17:1880–1890
Funahashi S (2006) Prefrontal cortex and working memory processes. Neuroscience 139:251–261. https://doi.org/10.1016/j.neuroscience.2005.07.003
Garcia RC et al (2012) Neurotoxicity of anhydroecgonine methyl ester, a crack cocaine pyrolysis product. Toxicol Sci 128:223–234. https://doi.org/10.1093/toxsci/kfs140
Garcia RC et al (2015) M1 and M3 muscarinic receptors may play a role in the neurotoxicity of anhydroecgonine methyl ester, a cocaine pyrolysis product. Sci Rep 5:17555. https://doi.org/10.1038/srep17555
Garcia RC et al (2017) Anhydroecgonine methyl ester, a cocaine pyrolysis product, may contribute to cocaine behavioral sensitization. Toxicology 376:44–50. https://doi.org/10.1016/j.tox.2016.04.009
Gaspar P, Bloch B, Le Moine C (1995) D1 and D2 receptor gene expression in the rat frontal cortex: cellular localization in different classes of efferent neurons. Eur J Neurosci 7:1050–1063
George O, Mandyam CD, Wee S, Koob GF (2008) Extended access to cocaine self-administration produces long-lasting prefrontal cortex-dependent working memory impairments. Neuropsychopharmacology 33:2474–2482. https://doi.org/10.1038/sj.npp.1301626
Guan ZZ (2008) Cross-talk between oxidative stress and modifications of cholinergic and glutaminergic receptors in the pathogenesis of Alzheimer's disease. Acta Pharmacol Sin 29:773–780. https://doi.org/10.1111/j.1745-7254.2008.00819.x
Haas C, Karila L, Lowenstein W (2009) Cocaine and crack addiction: a growing public health problem. Bull Acad Natl Med 193:947–962 discussion 962–943
Halliwell B (1992) Reactive oxygen species and the central nervous system. J Neurochem 59:1609–1623
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658. https://doi.org/10.1111/j.1471-4159.2006.03907.x
Hatsukami DK, Fischman MW (1996) Crack cocaine and cocaine hydrochloride. Are the differences myth or reality? JAMA 276:1580–1588
Hersch SM, Gutekunst CA, Rees HD, Heilman CJ, Levey AI (1994) Distribution of m1-m4 muscarinic receptor proteins in the rat striatum: light and electron microscopic immunocytochemistry using subtype-specific antibodies. J Neurosci 14:3351–3363
Huang X, Gu HH, Zhan CG (2009) Mechanism for cocaine blocking the transport of dopamine: insights from molecular modeling and dynamics simulations. J Phys Chem B 113:15057–15066. https://doi.org/10.1021/jp900963n
Inagawa K (1993) Cholinergic modulation of spatial working memory of mice in radial maze performance: retention curve analysis. Yakubutsu Seishin Kodo 13:233–238
Jacob P 3rd, Jones RT, Benowitz NL, Shulgin AT, Lewis ER, Elias-Baker BA (1990a) Cocaine smokers excrete a pyrolysis product, anhydroecgonine methyl ester. J Toxicol Clin Toxicol 28:121–125
Jacob P 3rd, Lewis ER, Elias-Baker BA, Jones RT (1990b) A pyrolysis product, anhydroecgonine methyl ester (methylecgonidine), is in the urine of cocaine smokers. J Anal Toxicol 14:353–357
Jones MW (2002) A comparative review of rodent prefrontal cortex and working memory. Curr Mol Med 2:639–647
Joseph JA, Fisher DR, Strain J (2002) Muscarinic receptor subtype determines vulnerability to oxidative stress in COS-7 cells. Free Radic Biol Med 32:153–161
Jucaite A (2002) Dopaminergic modulation of cerebral activity and cognitive functions. Medicina (Kaunas) 38:357–362
Kang S, Ling QL, Liu WT, Lu B, Liu Y, He L, Liu JG (2013) Down-regulation of dorsal striatal RhoA activity and impairment of working memory in middle-aged rats. Neurobiol Learn Mem 103:3–10. https://doi.org/10.1016/j.nlm.2013.03.005
Kesner RP, Hunt ME, Williams JM, Long JM (1996) Prefrontal cortex and working memory for spatial response, spatial location, and visual object information in the rat. Cereb Cortex 6:311–318
Kintz P, Cirimele V, Sengler C, Mangin P (1995) Testing human hair and urine for anhydroecgonine methyl ester, a pyrolysis product of cocaine. J Anal Toxicol 19:479–482
Korkmaz GG, Altinoglu E, Civelek S, Sozer V, Erdenen F, Tabak O, Uzun H (2013) The association of oxidative stress markers with conventional risk factors in the metabolic syndrome. Metabolism 62:828–835. https://doi.org/10.1016/j.metabol.2013.01.002
Lara AH, Wallis JD (2015) The role of prefrontal cortex in working memory: a mini review. Front Syst Neurosci 9:173. https://doi.org/10.3389/fnsys.2015.00173
Laroche S, Davis S, Jay TM (2000) Plasticity at hippocampal to prefrontal cortex synapses: dual roles in working memory and consolidation. Hippocampus 10:438–446. https://doi.org/10.1002/1098-1063(2000)10:4<438::AID-HIPO10>3.0.CO;2-3
Li HB, Matsumoto K, Yamamoto M, Watanabe H (1997) NMDA but not AMPA receptor antagonists impair the delay-interposed radial maze performance of rats. Pharmacol Biochem Behav 58:249–253
Lopez-Pedrajas R et al (2015) Cocaine promotes oxidative stress and microglial-macrophage activation in rat cerebellum. Front Cell Neurosci 9:279. https://doi.org/10.3389/fncel.2015.00279
Mansouri FA, Rosa MG, Atapour N (2015) Working memory in the service of executive control functions. Front Syst Neurosci 9:166. https://doi.org/10.3389/fnsys.2015.00166
Martin BR, Lue LP, Boni JP (1989) Pyrolysis and volatilization of cocaine. J Anal Toxicol 13:158–162
McElvain JS, Schenk JO (1992) Studies of the mechanism of inhibition of the dopamine uptake carrier by cocaine in vitro using rotating disk electrode voltammetry. Ann N Y Acad Sci 654:480–482
Medeiros MS et al (2016) Iron and Oxidative Stress in Parkinson’s Disease: An Observational Study of Injury Biomarkers. PLoS One 11:e0146129. https://doi.org/10.1371/journal.pone.0146129
Mizumori SJ, Rosenzweig MR, Bennett EL (1985) Long-term working memory in the rat: effects of hippocampally applied anisomycin. Behav Neurosci 99:220–232
Moura HF, Benzano D, Pechansky F, Kessler FH (2014) Crack/cocaine users show more family problems than other substance users. Clinics (Sao Paulo) 69:497–499
Muir JL (1997) Acetylcholine, aging, and Alzheimer’s disease. Pharmacol Biochem Behav 56:687–696
Nakamura EM, da Silva EA, Concilio GV, Wilkinson DA, Masur J (1991) Reversible effects of acute and long-term administration of delta-9-tetrahydrocannabinol (THC) on memory in the rat. Drug Alcohol Depend 28:167–175
Nakamura-Palacios EM (2010) Working memory and prefrontal cortex and their relation with the brain reward system and drug addiction. In: Levin ES (ed) Working memory: capacity, developments and improvement techniques. NOVA Publishers, New York, pp 109–140
Numa R, Kohen R, Poltyrev T, Yaka R (2008) Tempol diminishes cocaine-induced oxidative damage and attenuates the development and expression of behavioral sensitization. Neuroscience 155:649–658. https://doi.org/10.1016/j.neuroscience.2008.05.058
Ohno M, Yamamoto T, Watanabe S (1994) Blockade of hippocampal M1 muscarinic receptors impairs working memory performance of rats. Brain Res 650:260–266
Oliveira LG, Ponce Jde C, Nappo SA (2010) Crack cocaine use in Barcelona: a reason of worry. Subst Use Misuse 45:2291–2300. https://doi.org/10.3109/10826081003682883
Olton DS (1987) The radial arm maze as a tool in behavioral pharmacology. Physiol Behav 40:793–797
Olton DS, Samuelson RJ (1976) Relembrance of places passed: spatial memory in rats. J Exp Physiol: Anim Behav Process 2:97–116
Oztekin I, McElree B, Staresina BP, Davachi L (2009) Working memory retrieval: contributions of the left prefrontal cortex, the left posterior parietal cortex, and the hippocampus. J Cogn Neurosci 21:581–593. https://doi.org/10.1162/jocn.2008.21016
Paquette C, Roy E, Petit G, Boivin JF (2010) Predictors of crack cocaine initiation among Montreal street youth: a first look at the phenomenon. Drug Alcohol Depend 110:85–91. https://doi.org/10.1016/j.drugalcdep.2010.02.010
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates. Academic Press, USA
Pearson JN, Patel M (2016) The role of oxidative stress in organophosphate and nerve agent toxicity. Ann N Y Acad Sci 1378:17–24. https://doi.org/10.1111/nyas.13115
Picciotto MR, Higley MJ, Mineur YS (2012) Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron 76:116–129. https://doi.org/10.1016/j.neuron.2012.08.036
Pifl C, Drobny H, Reither H, Hornykiewicz O, Singer EA (1995) Mechanism of the dopamine-releasing actions of amphetamine and cocaine: plasmalemmal dopamine transporter versus vesicular monoamine transporter. Mol Pharmacol 47:368–373
Poch C, Campo P (2012) Neocortical-hippocampal dynamics of working memory in healthy and diseased brain states based on functional connectivity. Front Hum Neurosci 6:36. https://doi.org/10.3389/fnhum.2012.00036
Pomierny-Chamiolo L, Moniczewski A, Wydra K, Suder A, Filip M (2013) Oxidative stress biomarkers in some rat brain structures and peripheral organs underwent cocaine. Neurotox Res 23:92–102. https://doi.org/10.1007/s12640-012-9335-6
Porter MC, Mair RG (1997) The effects of frontal cortical lesions on remembering depend on the procedural demands of tasks performed in the radial arm maze. Behav Brain Res 87:115–125
Potvin S, Stavro K, Rizkallah E, Pelletier J (2014) Cocaine and cognition: a systematic quantitative review. J Addict Med 8:368–376. https://doi.org/10.1097/ADM.0000000000000066
Pratico D, Uryu K, Leight S, Trojanoswki JQ, Lee VM (2001) Increased lipid peroxidation precedes amyloid plaque formation in an animal model of Alzheimer amyloidosis. J Neurosci 21:4183–4187
Provost JS, Petrides M, Monchi O (2010) Dissociating the role of the caudate nucleus and dorsolateral prefrontal cortex in the monitoring of events within human working memory. Eur J Neurosci 32:873–880. https://doi.org/10.1111/j.1460-9568.2010.07333.x
Rauch SL, Raskin LA (1984) Cholinergic mediation of spatial memory in the preweanling rat: application of the radial arm maze paradigm. Behav Neurosci 98:35–43
Richter SH, Zeuch B, Lankisch K, Gass P, Durstewitz D, Vollmayr B (2013) Where have I been? Where should I go? Spatial working memory on a radial arm maze in a rat model of depression. PLoS One 8:e62458. https://doi.org/10.1371/journal.pone.0062458
Rios Valentim SJ Jr, Gontijo AV, Peres MD, Rodrigues LC, Nakamura-Palacios EM (2009) D1 dopamine and NMDA receptors interactions in the medial prefrontal cortex: modulation of spatial working memory in rats. Behav Brain Res 204:124–128. https://doi.org/10.1016/j.bbr.2009.05.026
Rodrigues LC, Conti CL, Nakamura-Palacios EM (2011) Clozapine and SCH 23390 prevent the spatial working memory disruption induced by Delta9-THC administration into the medial prefrontal cortex. Brain Res 1382:230–237. https://doi.org/10.1016/j.brainres.2011.01.069
Santibanez SS et al (2005) Prevalence and correlates of crack-cocaine injection among young injection drug users in the United States, 1997-1999. Drug Alcohol Depend 77:227–233. https://doi.org/10.1016/j.drugalcdep.2004.08.020
Scheidweiler KB, Plessinger MA, Shojaie J, Wood RW, Kwong TC (2003) Pharmacokinetics and pharmacodynamics of methylecgonidine, a crack cocaine pyrolyzate. J Pharmacol Exp Ther 307:1179–1187. https://doi.org/10.1124/jpet.103.055434
Schifano F, Corkery J (2008) Cocaine/crack cocaine consumption, treatment demand, seizures, related offences, prices, average purity levels and deaths in the UK (1990-2004). J Psychopharmacol 22:71–79. https://doi.org/10.1177/0269881107079170
Seamans JK, Yang CR (2004) The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74:1–58. https://doi.org/10.1016/j.pneurobio.2004.05.006
Seamans JK, Floresco SB, Phillips AG (1998) D1 receptor modulation of hippocampal-prefrontal cortical circuits integrating spatial memory with executive functions in the rat. J Neurosci 18:1613–1621
Seeger T et al (2004) M2 muscarinic acetylcholine receptor knock-out mice show deficits in behavioral flexibility, working memory, and hippocampal plasticity. J Neurosci 24:10117–10127. https://doi.org/10.1523/JNEUROSCI.3581-04.2004
Shih YT, Chen PS, Wu CH, Tseng YT, Wu YC, Lo YC (2010) Arecoline, a major alkaloid of the areca nut, causes neurotoxicity through enhancement of oxidative stress and suppression of the antioxidant protective system. Free Radic Biol Med 49:1471–1479. https://doi.org/10.1016/j.freeradbiomed.2010.07.017
Siegel RK (1979) Cocaine smoking. N Engl J Med 300:373. https://doi.org/10.1056/NEJM197902153000731
Siegel RK (1982) Cocaine smoking. J Psychoactive Drugs 14:271–359. https://doi.org/10.1080/02791072.1982.10524303
Silva de Melo LC, Cruz AP, Rios Valentim SJ Jr, Marinho AR, Mendonca JB, Nakamura-Palacios EM (2005) Delta(9)-THC administered into the medial prefrontal cortex disrupts the spatial working memory. Psychopharmacology (Berl) 183:54–64. https://doi.org/10.1007/s00213-005-0141-1
Smith EE, Jonides J (1999) Storage and executive processes in the frontal lobes. Science 283:1657–1661
Sofuoglu M, Mooney M (2009) Cholinergic functioning in stimulant addiction: implications for medications development. CNS Drugs 23:939–952. https://doi.org/10.2165/11310920-000000000-00000
Steketee JD (2003) Neurotransmitter systems of the medial prefrontal cortex: potential role in sensitization to psychostimulants. Brain Res Brain Res Rev 41:203–228
Sudai E et al (2011) High cocaine dosage decreases neurogenesis in the hippocampus and impairs working memory. Addict Biol 16:251–260. https://doi.org/10.1111/j.1369-1600.2010.00241.x
Taylor CL, Latimer MP, Winn P (2003) Impaired delayed spatial win-shift behaviour on the eight arm radial maze following excitotoxic lesions of the medial prefrontal cortex in the rat. Behav Brain Res 147:107–114
Toennes SW, Fandino AS, Kauert G (1999) Gas chromatographic-mass spectrometric detection of anhydroecgonine methyl ester (methylecgonidine) in human serum as evidence of recent smoking of crack. J Chromatogr B Biomed Sci Appl 735:127–132
Toennes SW, Fandino AS, Hesse FJ, Kauert GF (2003) Artifact production in the assay of anhydroecgonine methyl ester in serum using gas chromatography-mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 792:345–351
Vaughn MG, Fu Q, Perron BE, Bohnert AS, Howard MO (2010) Is crack cocaine use associated with greater violence than powdered cocaine use? Results from a national sample. Am J Drug Alcohol Abuse 36:181–186. https://doi.org/10.3109/00952990.2010.491877
Versace M, Zorzi M (2010) The role of dopamine in the maintenance of working memory in prefrontal cortex neurons: input-driven versus internally-driven networks. Int J Neural Syst 20:249–265. https://doi.org/10.1142/S0129065710002401
Vivancos R, Maskrey V, Rumball D, Harvey I, Holland R (2006) Crack/cocaine use in a rural county of England. J Public Health (Oxf) 28:96–103. https://doi.org/10.1093/pubmed/fdl010
Watanabe T et al (2009) Decreased acetylcholine release is correlated to memory impairment in the Tg2576 transgenic mouse model of Alzheimer’s disease. Brain Res 1249:222–228. https://doi.org/10.1016/j.brainres.2008.10.029
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–333
Williams MJ, Adinoff B (2008) The role of acetylcholine in cocaine addiction. Neuropsychopharmacology 33:1779–1797. https://doi.org/10.1038/sj.npp.1301585
Wirt RA, Hyman JM (2017) Integrating spatial working memory and remote memory: interactions between the medial prefrontal cortex and hippocampus Brain Sci 7 doi:https://doi.org/10.3390/brainsci7040043
Witko-Sarsat V, Nguyen Khoa T, Jungers P, Drueke T, Descamps-Latscha B (1998) Advanced oxidation protein products: oxidative stress markers and mediators of inflammation in uremia. Adv Nephrol Necker Hosp 28:321–341
Yang Y, Ke Q, Cai J, Xiao YF, Morgan JP (2001) Evidence for cocaine and methylecgonidine stimulation of M(2) muscarinic receptors in cultured human embryonic lung cells. Br J Pharmacol 132:451–460. https://doi.org/10.1038/sj.bjp.0703819
Yoon T, Okada J, Jung MW, Kim JJ (2008) Prefrontal cortex and hippocampus subserve different components of working memory in rats. Learn Mem 15:97–105. https://doi.org/10.1101/lm.850808
Yoshihara T, Ichitani Y (2004) Hippocampal N-methyl-D-aspartate receptor-mediated encoding and retrieval processes in spatial working memory: delay-interposed radial maze performance in rats. Neuroscience 129:1–10. https://doi.org/10.1016/j.neuroscience.2004.07.030
You ZB, Wang B, Zitzman D, Wise RA (2008) Acetylcholine release in the mesocorticolimbic dopamine system during cocaine seeking: conditioned and unconditioned contributions to reward and motivation. J Neurosci 28:9021–9029. https://doi.org/10.1523/JNEUROSCI.0694-08.2008
Zahrt J, Taylor JR, Mathew RG, Arnsten AF (1997) Supranormal stimulation of D1 dopamine receptors in the rodent prefrontal cortex impairs spatial working memory performance. J Neurosci 17:8528–8535
Zhang Y, Bailey KR, Toupin MM, Mair RG (2005) Involvement of ventral pallidum in prefrontal cortex-dependent aspects of spatial working memory. Behav Neurosci 119:399–409. https://doi.org/10.1037/0735-7044.119.2.399
Acknowledgements
We thank Professor Nyam Florencio da Silva from Federal University of Espírito Santo, the Laboratory of Molecular Histology and Immunohistochemistry (LHMI) from Federal University of Espírito Santo and the Civil Police Department of Espírito Santo State.
Funding
EMNP is a recipient of a researcher fellowship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (proc. 304374/2014-8) and is also funded by this agency (proc. 466650/2014-0; 475232/2013-5 and 443824/2014-2) investigating the effects of brain stimulation in drug addiction. LCRM is funded by CNPq (proc. 456041/2014-1). EFG and IFSL were recipients of a graduate student fellowship from Espírito Santo Research Foundation FAPES (69923329) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All procedures performed in studies involving animals were in accordance with the ethical principles in animal experimentation (Brazilian College in Animal Experimentation—COBEA, revised, 2000). This project was submitted to the Commission on Ethics and Animal experimentation (CEUA) of CCS/UFES, approved under the number 10/2015.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gomes, E.F., Lipaus, I.F.S., Martins, C.W. et al. Anhydroecgonine Methyl Ester (AEME), a Product of Cocaine Pyrolysis, Impairs Spatial Working Memory and Induces Striatal Oxidative Stress in Rats. Neurotox Res 34, 834–847 (2018). https://doi.org/10.1007/s12640-017-9813-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-017-9813-y