Abstract
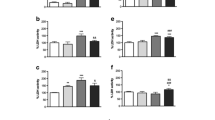
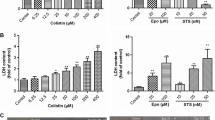
Doxorubicin (DOX) is neurotoxic to serum-free cultures of rat cortical neurons in a biphasic concentration manner. For concentrations up to 0.5 μM, cell death follows an apoptotic pattern, while for higher concentrations apoptosis is inhibited and necrosis becomes dominant. Considering the potential toxic effects of DOX resulting from its redox-cycling, in this study we investigated the generation of reactive species and subsequent oxidative stress effects, formation of quinoproteins, activation of NF-kB, depletion of energy levels and consequent cell death in cultures of primary rat cortical cells challenged with this antitumour drug. The influence of neuronal nitric oxide synthase (nNOS) on DOX-induced neuronal cell damage was subsequently evaluated. The exposure of rat cortical primary cell cultures to DOX resulted in a significant generation of ROS/RNS, activation of NF-kB, depletion of GSH levels, depletion of ATP, and cell death, in a concentration biphasic manner. Doxorubicin also significantly increased protein-bound quinone products in neurons in a concentration-dependent manner. Inhibition of nNOS decreased neuronal cell death induced by DOX in a significant way, at the first phase of the biphasic curve. In conclusion, this study shows, for the first time, a clear involvement of nNOS and subsequent ROS/RNS generation as crucial signalling mediators of DOX-induced neurotoxicity on isolated cortical neurons. Inhibition of ROS/RNS formation, modulation of NOS isoforms and modulation of NF-kB activation could be of beneficial in preventing damage in the CNS caused by DOX.






Similar content being viewed by others
References
Abd El-Gawad HM, El-Sawalhi MM (2004) Nitric oxide and oxidative stress in brain and heart of normal rats treated with doxorubicin: role of aminoguanidine. J Biochem Mol Toxicol 18:69–77
Aleyasin H, Cregan SP, Iyirhiaro G, O’Hare MJ, Callaghan SM, Slack RS, Park DS (2004) Nuclear factor-(kappa)B modulates the p53 response in neurons exposed to DNA damage. J Neurosci 24:2963–2973
Aoki M, Nata T, Morishita R, Matsushita H, Nakagami H, Yamamoto K, Yamazaki K, Nakabayashi M, Ogihara T, Kaneda Y (2001) Endothelial apoptosis induced by oxidative stress through activation of NF-kappaB: antiapoptotic effect of antioxidant agents on endothelial cells. Hypertension 38:48–55
Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA (1990) Apparent hydroxyl radical production by peroxynitrite: implications for endothelial injury from nitric oxide and superoxide. Proc Natl Acad Sci USA 87:1620–1624
Bigotte L, Olsson Y (1982) Cytofluorescence localization of adriamycin in the nervous system. III. Distribution of the drug in the brain of normal adult mice after intraventricular and arachnoidal injections. Acta Neuropathol 58:193–202
Boland MP, Foster SJ, O’Neill LA (1997) Daunorubicin activates NFkappaB and induces kappaB-dependent gene expression in HL-60 promyelocytic and Jurkat T lymphoma cells. J Biol Chem 272:12952–12960
Brewer GJ (1995) Serum-free B27/neurobasal medium supports differentiated growth of neurons from the striatum, substantia nigra, septum, cerebral cortex, cerebellum, and dentate gyrus. J Neurosci Res 42:674–683
Burdo J, Schubert D, Maher P (2008) Glutathione production is regulated via distinct pathways in stressed and non-stressed cortical neurons. Brain Res 1189:12–22
Carvalho M, Remiao F, Milhazes N, Borges F, Fernandes E, Monteiro Mdo C, Goncalves MJ, Seabra V, Amado F, Carvalho F, Bastos ML (2004) Metabolism is required for the expression of ecstasy-induced cardiotoxicity in vitro. Chem Res Toxicol 17:623–632
Dohi K, Ohtaki H, Inn R, Ikeda Y, Shioda HS, Aruga T (2003) Peroxynitrite and caspase-3 expression after ischemia/reperfusion in mouse cardiac arrest model. Acta Neurochir Suppl 86:87–91
Eisenhauer EA, Vermorken JB (1998) The taxoids. Comparative clinical pharmacology and therapeutic potential. Drugs 55:5–30
Fu J, Yamamoto K, Guan ZW, Kimura S, Iyanagi T (2004) Human neuronal nitric oxide synthase can catalyze one-electron reduction of adriamycin: role of flavin domain. Arch Biochem Biophys 427:180–187
Gomes A, Fernandes E, Lima JL (2005) Fluorescence probes used for detection of reactive oxygen species. J Biochem Biophys Methods 65:45–80
Gutierrez PL (2000) The role of NAD(P)H oxidoreductase (DT-Diaphorase) in the bioactivation of quinone-containing antitumor agents: a review. Free Radic Biol Med 29:263–275
Hill IE, Murray C, Richard J, Rasquinha I, MacManus JP (2000) Despite the internucleosomal cleavage of DNA, reactive oxygen species do not produce other markers of apoptosis in cultured neurons. Exp Neurol 162:73–88
Joshi G, Sultana R, Tangpong J, Cole MP, St Clair DK, Vore M, Estus S, Butterfield DA (2005) Free radical mediated oxidative stress and toxic side effects in brain induced by the anti cancer drug adriamycin: insight into chemobrain. Free Radic Res 39:1147–1154
Kappus H (1987) Oxidative stress in chemical toxicity. Arch Toxicol 60:144–149
Kim R, Yoshida K, Toge T (2002) Current status and future perspectives of post-operative adjuvant therapy for gastric carcinoma. Anticancer Res 22:283–289
Kleiner HE, Rivera MI, Pumford NR, Monks TJ, Lau SS (1998) Immunochemical detection of quinol–thioether-derived protein adducts. Chem Res Toxicol 11:1283–1290
Koh JY, Choi DW (1987) Quantitative determination of glutamate mediated cortical neuronal injury in cell culture by lactate dehydrogenase efflux assay. J Neurosci Methods 20:83–90
Laurent G, Jaffrezou JP (2001) Signaling pathways activated by daunorubicin. Blood 98:913–924
Lopes MA, Meisel A, Dirnagl U, Carvalho FD, Bastos Mde L (2008) Doxorubicin induces biphasic neurotoxicity to rat cortical neurons. Neurotoxicology 29:286–293
Luo D, Vincent SR (1994) Inhibition of nitric oxide synthase by antineoplastic anthracyclines. Biochem Pharmacol 47:2111–2112
Martin D, Salinas M, Fujita N, Tsuruo T, Cuadrado A (2002) Ceramide and reactive oxygen species generated by H2O2 induce caspase-3-independent degradation of Akt/protein kinase B. J Biol Chem 277:42943–42952
Minotti G, Cairo G, Monti E (1999) Role of iron in anthracycline cardiotoxicity: new tunes for an old song? FASEB J 13:199–212
Minotti G, Menna P, Salvatorelli E, Cairo G, Gianni L (2004) Anthracyclines: molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol Rev 56:185–229
Miyazaki I, Asanuma M, Diaz-Corrales FJ, Fukuda M, Kitaichi K, Miyoshi K, Ogawa N (2006) Methamphetamine-induced dopaminergic neurotoxicity is regulated by quinone-formation-related molecules. FASEB J 20:571–573
Ohnishi T, Tamai I, Sakanaka K, Sakata A, Yamashima T, Yamashita J, Tsuji A (1995) In vivo and in vitro evidence for ATP-dependency of P-glycoprotein-mediated efflux of doxorubicin at the blood-brain barrier. Biochem Pharmacol 49:1541–1544
Ruscher K, Reuter M, Kupper D, Trendelenburg G, Dirnagl U, Meisel A (2000) A fluorescence based non-radioactive electrophoretic mobility shift assay. J Biotechnol 78:163–170
Solenski NJ, Kostecki VK, Dovey S, Periasamy A (2003) Nitric-oxide-induced depolarization of neuronal mitochondria: implications for neuronal cell death. Mol Cell Neurosci 24:1151–1169
Tangpong J, Cole MP, Sultana R, Joshi G, Estus S, Vore M, St Clair W, Ratanachaiyavong S, St Clair DK, Butterfield DA (2006) Adriamycin-induced, TNF-alpha-mediated central nervous system toxicity. Neurobiol Dis 23:127–139
Tangpong J, Cole MP, Sultana R, Estus S, Vore M, St Clair W, Ratanachaiyavong S, St Clair DK, Butterfield DA (2007) Adriamycin-mediated nitration of manganese superoxide dismutase in the central nervous system: insight into the mechanism of chemobrain. J Neurochem 100(1):191–201
Vail DM, Amantea MA, Colbern GT, Martin FJ, Hilger RA, Working PK (2004) Pegylated liposomal doxorubicin: proof of principle using preclinical animal models and pharmacokinetic studies. Semin Oncol 31:16–35
Vasquez-Vivar J, Martasek P, Hogg N, Masters BS, Pritchard KA Jr, Kalyanaraman B (1997) Endothelial nitric oxide synthase-dependent superoxide generation from adriamycin. Biochemistry 36:11293–11297
Wagner S, Peters O, Fels C, Janssen G, Liebeskind AK, Sauerbrey A, Suttorp M, Hau P, Wolff JE (2008) Pegylated-liposomal doxorubicin and oral topotecan in eight children with relapsed high-grade malignant brain tumors. J Neurooncol 86:175–181
Wang S, Kotamraju S, Konorev E, Kalivendi S, Joseph J, Kalyanaraman B (2002) Activation of nuclear factor-kappaB during doxorubicin-induced apoptosis in endothelial cells and myocytes is pro-apoptotic: the role of hydrogen peroxide. Biochem J 367:729–740
Wang JY, Shum AY, Ho YJ, Wang JY (2003) Oxidative neurotoxicity in rat cerebral cortex neurons: synergistic effects of H2O2 and NO on apoptosis involving activation of p38 mitogen-activated protein kinase and caspase-3. J Neurosci Res 72:508–519
Weiss RB (1992) The anthracyclines: will we ever find a better doxorubicin? Semin Oncol 19:670–686
Acknowledgements
The authors would like to thank Professor Ulrich Dirnagl for his help in the integration and discussion of the presented data. Miguel Ângelo Lopes was the recipient of a PhD grant from “Fundação para a Ciência e Tecnologia” (Ref. SFRD/BD/17899/2004).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lopes, M.Â., Meisel, A., Carvalho, F.D. et al. Neuronal Nitric Oxide Synthase is a Key Factor in Doxorubicin-Induced Toxicity to Rat-Isolated Cortical Neurons. Neurotox Res 19, 14–22 (2011). https://doi.org/10.1007/s12640-009-9135-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-009-9135-9