Abstract
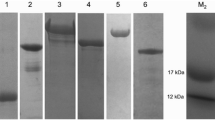
Ticks cause anemia, toxicosis, growth delay, and transmit infectious diseases in animals and humans. The current study aimed to evaluate the immunoprophylactic properties of two vaccine candidates to develop vaccine against tick infestations. These two vaccine candidates were specific fraction from the adults of the soft tick Ornithodoros savignyi and cross-reactive fraction from the adults of the hard tick Hyalomma dromedarii. Both specific and cross-reactive fractions were isolated by Cyanogen Bromide-activated Sepharose-4B affinity column chromatography. Both candidates proved their cross-reactivity by enzyme linked immunosorbent assay and Western blot. Characterization of the two vaccines by SDS-PAGE showed that the O. savignyi specific fraction consists of four bands; 97, 85, 66 and 11.5 kDa compared with nine bands associated with its crude antigen (196–11.5 kDa). The H. dromedarii cross-reactive vaccine candidate consists of three bands; 97, 66 and 45 kDa compared to eight bands of its crude antigen (196–21 kDa). Two common bands of 97 and 66 kDa between two candidates showed immunogenic cross-reactivity with the developed antisera of both infestations by Western blot. Immunization of rabbits intramuscularly with two doses of the fractions separately (40 µg/kg) led to immunological and parasitological changes. Immunologically; the level of immunoglobulins in vaccinated rabbits increased significantly compared with control infested non-vaccinated rabbits. These immunoglobulins are probably responsible for the protective effect of both candidates. Parasitologically, immunized rabbits showed protection against infestation by adult ticks as proved by significant feeding rejection percentage and significant reduction in egg and engorgement weights of H. dromedarii. While insignificant protection was observed against O. savignyi ticks infestation in feeding rejection and reduction in engorgement weight. In conclusion, this study suggests promising immunoprophylactic potentials of the purified fractions against tick infestations in rabbits through induction of IgG responses. The protective effect of both vaccine candidates deserves further evaluation in other hosts and against other tick infestations.







Similar content being viewed by others
References
Abdel-Shafy S, Allam NAT, Mediannikov O, Parola P, Raoult D (2012) Molecular detection of spotted fever group Rickettsiae associated with ixodid ticks in Egypt. Vector Borne Zoonotic Dis 12:346–359
Abdullah HHAM, El-Shanawany EE, Abdel-Shafy S, Abou-Zeina HAA, Abdel-Rahman EH (2018) Molecular and immunological characterization of Hyalomma dromedarii and Hyalomma excavatum (Acari: Ixodidae) vectors of Q fever in camels. Vet World 11(8):1109–1119
Antunes S, Merino O, Lerias J, Domingues N, Mosqueda J, de la Fuente J, Domingos A (2015) Artificial feeding of Rhipicephalus microplus female ticks with anti calreticulin. Ticks Tick Borne Dis 6:47–55
Carpio Y, Basabe L, Acosta J, Rodríguez A, Mendoza A, Lisperger A, Zamorano E, González M, Rivas M, Contreras S, Haussmann D, Figueroa J, Osorio VN, Asencio G, Mancilla J, Ritchie G, Borroto C, Estrada MP (2011) Novel gene isolated from Caligus rogercresseyi: a promising target for vaccine development against sea lice. Vaccine 29:2810–2820
Contrerasa M, de la Fuentea J (2016) Control of Ixodes ricinus and Dermacentor reticulatus tick infestations in rabbits vaccinated with the Q38 Subolesin/Akirin chimera. Vaccine 34:3010–3013
De la Fuente J, Almazán C, Canales M, Pérez de la Lastra JM, Kocan KM, Willadsen P (2007a) A ten-year review of commercial vaccine performance for control of tick infestations on cattle. Anim Health Res Rev 8(1):23–28
De la Fuente J, Kocan KM, Blouin EF (2007b) Tick vaccines and the transmission of tick-borne pathogens. Vet Res Commun 31(Suppl. 1):85–90
De la Fuente J, Estrada-Peña A, Venzal JM, Kocan KM, Sonenshine DE (2008) Overview: ticks as vectors of pathogens that cause disease in humans and animals. Front Biosci 13:6938–6946
De la Fuente J, Rodriguez M, Garcia-Garcia JC (2009) Immunological control of ticks through vaccination with Boophilus microplus gut antigens. Ann N Y Acad Sci 916:617–621
De la Fuente J, Moreno-Cid JA, Canales M, Villar M, Pérez de la Lastra JM, Kocan KM, Galindo RC, Almazán C, Blouin EF (2011) Targeting arthropod subolesin/akirin for the development of a universal vaccine for control of vector infestations and pathogen transmission. Vet Parasitol 181:17–22
Engvall E, Perlmann P (1971) Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry 8:871–874
Fragoso H, Rad PH, Ortiz M, Rodríguez M, Redondo M, Herrera L, de la Fuente J (1998) Protection against Boophilus annulatus infestations in cattle vaccinated with the B. microplus Bm86-containing vaccine Gavac. Vaccine 16:1990–1992
Garcîa-Varas S, Manzano-Român R, Fernândez-Soto P, Encinas-Grandes A, Oleaga A, Pérez-Sánchez R (2010) Purification and characterisation of a P-selectin-binding molecule from the salivary glands of Ornithodoros moubata that induces protective anti-tick immune responses in pigs. Int J Parasitol 40:313–326
Ghosh S, Khan MH (1999) Immunization of cattle against Hyalomma anatolicum anatolicum using larval antigens. Indian J Exp Biol 37:203–205
Guerrero FD, Miller RJ, Pérez de León AA (2012) Cattle tick vaccines: many can-didate antigens, but will a commercially viable product emerge? Int J Parasitol 42:421–427
Gunes T (2006) Crimean–Congo hemorrhagic fever. Mikrobiyol Bul 40:276–287
Hassan MI, Gabr HSM, Abdel-Shafy S, Hammad KM, Mokhtar MM (2017) Prevalence of tick-vectors of Theileria annulata infesting the one-humped camels in Giza, Egypt. J Egypt Soc Parasitol 47:425–432
Jongejan F, Uilenberg G (2004) The global importance of ticks. Parasitol 129:S3–S14
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Latif AA, Putterill JF, de Klerk DG, Pienaar R, Mans BJ (2012) Nuttalliellannamaquan(Ixodoidea: Nuttalliellidae): first description of the male, immature stages and re-description of the female. PLoS ONE 7:e41651
Loftis AD, Reeves WK, Szumlas DE, Abbassy MM, Helmy IM, Moriarity JR, Dasch GA (2006) Rickettsial agent in Egyptian ticks collected from domestic animals. Exp Appl Acarol 40:67–81
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Manzano-Román R, García-Varas S, Encinas-Grandes A, Pérez-Sánchez R (2007) Purification and characterization of a 45-kDa concealed antigen from the midgut membranes of Ornithodoros erraticus that induces lethal anti-tick immune responses in pigs. Vet Parasitol 145:314–325
Manzano-Román R, Díaz-Martín V, de la Fuente J, Ricardo Pérez-Sánchez R (2012) Soft ticks as pathogen vectors: distribution, surveillance and control. In: Manjur S (ed) Parasitology. Intech, Rijeka, pp 125–162
Merino O, Alberdi P, Pérez de la Lastra JM, de la Fuente J (2013) Tick vaccines and the control of tick-borne pathogens. Front Cell Infect Microbiol 3:30
Montasser AA (2005) Gram- negative bacteria from the camel tick Hyalomma dromedarii (Ixodidae) and the chicken tick Argas persicus (Argasidae) and their antibiotic sensitivities. J Egypt Soc Parasitol 35:95–106
Moreno-Cid JA, de la Lastra JMP, Villar M, Jiménez M, Pinal R, Estrada-Peña A, Molina R, Lucientes J, Gortázar C, de la Fuente J (2013) Control of multiple arthropod vector infestations with subolesin/akirin vaccines. Vaccine 31:1187–1196
Parizi LF, Githaka NW, Logullo C, Konnai S, Masuda A, Ohashi K, da Silva Vaz I Jr (2012) The quest FOs a universal vaccine against ticks: cross-immunity insights. Vet J 194:158–165
Peter RJ, Van den Bossche P, Penzhorn BL, Sharp B (2005) Tick, fly, and mosquito control-lessons from the past, solutions for the future. Vet Parasitol 132:205–215
Rajput ZI, Hu SH, Chen WJ, Arijo AG, Xiao CW (2006) Importance of ticks and their chemical and immunological control in livestock. J Zhejiang Univ Sci B 7:912–921
Rodríguez-Mallon A, Fernández E, Encinosa PE, Bello Y, Méndez-Pérez L, Ruiz LC, Pérez D, González M, Garay H, Reyes O, Méndez L, Estrada MP (2012) A novel tick antigen shows high vaccine efficacy against the dog tick, Rhipicephalus sanguineus. Vaccine 30:1782–1789
Rodriguez-Vivas RI, Trees AJ, Rosado-Aguilar JA, Villegas-Perez SL, Hodgkinson JE (2011) Evolution of acaricide resistance: Phenotypic and genotypic changes in field populations of Rhipicephalus (Boophilus) microplus in response to pyrethroid selection pressure. Int J Parasitol 41:895–903
Shapiro SZ, Voigt WP, Ellis JA (1989) Acquired resistance to ixodid ticks induced by tick cement antigen. Exp Appl Acarol 7:33–41
Szabó MPJ, Bechara GH (1997) Immunisation of dogs and guinea pigs against Rhipicephalus sanguineus ticks using gut extract. Vet Parasitol 68:283–294
Toaleb NI, Habeeb SM, Abdel-Rahman EH (2013) Purification, characterization and evaluation of larval immunogens of cattle tick Boophilus annulatus by infected cattle sera. Glob Vet 10(3):303–309
Towbin H, Stachelin T, Gordon J (1979) Electrophoretic transfer of proteins from polacrylamide gels to nitrocellulose sheets: procedure and some application. Proc Nat Acad Sci USA 179:4350–4354
Trimnell AR, Hails RS, Nuttall PA (2002) Dual action ectoparasite vaccine targeting ‘exposed’ and concealed’ antigens. Vaccine 20:3560–3568
Trimnell AR, Davies GM, Lissina O, Hails RS, Nuttall PAA (2005) Coss-reactive tick cement antigen is a candidate broad-spectrum tick vaccine. Vaccine 23:4329–4341
Vial L (2009) Biological and ecological characteristics of soft ticks (Ixodida: Argasidae) and their impact for predicting tick and associated disease distribution. Parasite 16:191–202
Vidarsson G, Dekkers G, Rispens T (2014) IgG subclasses and allotypes: from structure to effector functions. Front Immunol 5:520
Walker AR, Bouattour A, Camicas JL, Estrada-Peña A, Horak IG, Latif AA, Pegram RG, Preston PM (2003) Ticks of domestic animals in the Mediterranean region. A guide to identification of species, 1st edn. University of Zaragoza, Zaragoza, p 131
Willadsen P (2004) Anti-tick vaccines. Parasitol 129:S367–S387
Willadsen P (2008) Anti-tick vaccines. In: Bowman AS, Nuttall P (eds) Ticks: biology, disease and control. Cambridge University Press, Cambridge, pp 424–446
Wray W, Boulikas T, Wray VP, Hancock R (1981) Silver staining of proteins in polyacrylamide gels. Anal Biochem 118:197–203
Author information
Authors and Affiliations
Contributions
All authors participated in the study design. NIT and SA collected ticks and blood samples. NIT and EHA performed affinity chromatography, ELIZA, SDS-PAGE and western blot assays. NIT, HGS and SA shared in the rabbit-vaccine experiment. NIT and SA analyzed, figured and tabulated the data. NIT, SA and EHA participated in writing the manuscript. All authors revised and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was approved to the ethical standards of the relevant national and institutional guides on the care and use of laboratory animals by Medical Research Ethics Committee (No, 17132) at National Research Centre in Egypt.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Toaleb, N.I., Gabr, H.S.M., Abd El-Shafy, S. et al. Evaluation of vaccine candidates purified from the adult ticks of Ornithodoros savignyi (Acari: Argasidae) and Hyalomma dromedarii (Acari: Ixodidae) against tick infestations. J Parasit Dis 43, 246–255 (2019). https://doi.org/10.1007/s12639-018-01082-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-018-01082-3