Abstract
Purpose
Climate change is a global threat, and inhalational anesthetics contribute to global warming by altering the photophysical properties of the atmosphere. On a global perspective, there is a fundamental need to reduce perioperative morbidity and mortality and to provide safe anesthesia. Thus, inhalational anesthetics will remain a significant source of emissions in the foreseeable future. It is, therefore, necessary to develop and implement strategies to minimize the consumption of inhalational anesthetics to reduce the ecological footprint of inhalational anesthesia.
Source
We have integrated recent findings concerning climate change, characteristics of established inhalational anesthetics, complex simulative calculations, and clinical expertise to propose a practical and safe strategy to practice ecologically responsible anesthesia using inhalational anesthetics.
Principal findings
Comparing the global warming potential of inhalational anesthetics, desflurane is about 20 times more potent than sevoflurane and five times more potent than isoflurane. Balanced anesthesia using low or minimal fresh gas flow (≤ 1 L·min-1) during the wash-in period and metabolic fresh gas flow (0.35 L·min-1) during steady-state maintenance reduces CO2 emissions and costs by approximately 50%. Total intravenous anesthesia and locoregional anesthesia represent further options for lowering greenhouse gas emissions.
Conclusion
Responsible anesthetic management choices should prioritize patient safety and consider all available options. If inhalational anesthesia is chosen, the use of minimal or metabolic fresh gas flow reduces the consumption of inhalational anesthetics significantly. Nitrous oxide should be avoided entirely as it contributes to depletion of the ozone layer, and desflurane should only be used in justified exceptional cases.
Résumé
Objectif
Les changements climatiques constituent une menace mondiale et les anesthésiques volatils contribuent au réchauffement climatique en modifiant les propriétés photophysiques de l’atmosphère. Dans une perspective mondiale, il est fondamentalement nécessaire de réduire la morbidité et la mortalité périopératoires et de procurer une anesthésie sécuritaire. Par conséquent, les agents volatils demeureront une source importante d’émissions dans un avenir proche. Il est donc nécessaire d’élaborer et de mettre en œuvre des stratégies pour minimiser la consommation d’anesthésiques volatils afin de réduire l’empreinte écologique de l’anesthésie par inhalation.
Sources
Nous avons intégré les découvertes récentes concernant les changements climatiques, les caractéristiques des anesthésiques volatils connus, des calculs de simulation complexes et l’expertise clinique pour proposer une stratégie pratique et sécuritaire pour exercer une anesthésie écologiquement responsable en utilisant des anesthésiques volatils.
Constatations principales
En comparant le potentiel de réchauffement planétaire des anesthésiques volatils, le desflurane est environ 20 fois plus puissant que le sévoflurane et cinq fois plus puissant que l’isoflurane. Une anesthésie équilibrée avec un débit de gaz frais faible ou minimal (≤ 1 L·min-1) pendant la période de mise en route (‘wash-in’) et le débit métabolique de gaz frais (0,35 L·min-1) pendant le maintien à l’état d’équilibre réduit le CO2 et les coûts d’environ 50 %. L’anesthésie intraveineuse totale et l’anesthésie locorégionale représentent d’autres options pour réduire les émissions de gaz à effet de serre.
Conclusion
Les choix responsables en matière de prise en charge anesthésique devraient accorder la priorité à la sécurité des patients et à l’évaluation de toutes les options disponibles. Si l’anesthésie par inhalation est choisie, l’utilisation d’un débit minimal ou métabolique de gaz frais réduit considérablement la consommation d’anesthésiques volatils. Le protoxyde d’azote doit être complètement évité car il contribue à l’appauvrissement de la couche d’ozone, et le desflurane ne doit être utilisé que dans les cas exceptionnels et justifiés.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Climate change is considered to be the greatest challenge to our society and is a global threat.1 Global environmental changes such as climate change, extreme weather events, loss of biodiversity, and air pollution compromise the health and wellbeing of humans.2 The implications on global health are already apparent.3 According to the latest prediction by the World Health Organization, there will be an additional 25,000 deaths per year between 2030 and 2050.4
Consequently, there have been worldwide initiatives by governments, nongovernmental organizations, and individuals to promote or implement specific measures to limit the predicted increase in global temperature to 1.5°C. An imperative for the above should also apply to the health sector whose primary duty is to promote and safeguard health. Paradoxically, this sector is one of the largest global emitters of greenhouse gases, with a share of 4.4%.5 Anesthesia and critical care medicine significantly contribute to the enormous release of greenhouse gases due to their highly resource-intensive and high-tech working processes.6,7,8
As a significant source of emissions in the area of perioperative medicine, inhalational anesthetics contribute to the climate crisis by altering the photophysical properties of the atmosphere.9,10 Thus, it is necessary to develop and implement strategies to minimize the ecological footprint of anesthesia. Consequently, numerous anesthesiology societies and bodies have published recommendations in the past few years on how anesthesiologists can make their working area more sustainable and greener.7,11,12,13 A Letter to the Editor in this issue of the Journal illustrates one example of such initiatives: therein, He et al. report on a Greener Gases Starter Pack that the authors developed as part of the Greener Gases project at McMaster University (Hamilton, ON, Canada).14
In the present article, we focus on inhalational anesthetics and their impact on global warming and climate change. In this context, we review how the use of minimal and metabolic flow during general anesthesia (GA), subject of a Brief Review in the Journal a decade ago, reduces overall consumption of inhalational anesthetics, and contributes to lower emissions of greenhouse gases (GHGs).15 In addition, we discuss alternative anesthesia techniques along with their life cycles and environmental impacts, such as total intravenous anesthesia (TIVA) and locoregional anesthesia (LRA).16,17
Greenhouse gases and the global warming process
The term “atmosphere” refers to the totality of all layers that surround our Earth's surface as a gaseous envelope. In total, the atmosphere is divided into five different layers. For the purpose of the present discussion on the processes of global warming, we will focus on the following three layers: 1) the troposphere, up to 10,000 km; 2) the stratosphere, between 10,000 and 50,000 km; and 3) the mesosphere, beyond 50,000 km.
The GHGs defined and regulated by the Kyoto Protocol 1997 are carbon dioxide (CO2); methane (CH4); nitrous oxide (N2O); and fluorinated gases such as hydroxyfluorocarbon (HFC), perfluorocarbon (PFC), sulfur hexafluoride (SF6), and nitrogen trifluoride (NF3). Among the fluorinated gases are also the commonly used inhalational anesthetics desflurane, isoflurane, and sevoflurane. The global warming potential (GWP) of GHGs represents a common unit to measure absorption of energy in a defined period of time. Thus, it enables a comparison of the greenhouse potential of different gases. In general, the time period is set to 100 years. By definition, CO2 has a GWP of 1. In addition to the radiative effectiveness, the atmospheric lifetime is a second important determinant that influences GWP.18,19
In the atmosphere, natural concentrations of GHGs, clouds, water vapor, and other compounds adsorb and reflect incoming solar radiation back to space as well as outgoing infrared (IR) radiation back to the Earth. The equilibrium of incoming solar radiation and reflection as well as outgoing long wave IR radiation and IR reradiation creates and maintains appropriate living conditions on our planet.
A range of wavelengths through which electromagnetic radiation can pass the atmosphere back into space is called the atmospheric window. Infrared emission through the atmospheric window ranging from 8 to 14 µm plays an important role in regulating the Earth’s temperature. It is relatively transparent for IR as only little absorption by naturally occurring GHG occurs.20 Beyond this spectrum, IR radiation is mainly absorbed and reflected by these GHGs. Unfortunately, inhalational anesthetics absorb mainly within this atmospheric window. Hence, they act as GHGs and influence the Earth’s radiative balance by altering the IR radiation back to space. This favors the increase in global temperature.10
Stratospheric ozone depletion
In the stratosphere, there is a cyclic and constant formation and decomposition of ozone, which creates the ozone layer. The ozone layer absorbs ultraviolet (UV) radiation and protects plants, animals, and humans from increased and deleterious UV-B-radiation. This ozone can chemically be destroyed by reactive gases containing halogens such as chlorine and bromine. Halogen source gases are emitted at the surface and convert to reactive halogen gases by strong UV radiation in the stratosphere. In a catalytic cycle reaction, they initiate the breakdown of ozone and are reformed each time. Due to its extreme stability, one chlorine atom can thereby destroy thousands of ozone molecules before it is inactivated.21
Studies have shown that the depletion of stratospheric ozone changes the temperature in the southern troposphere. The resulting stronger winds heighten the ventilation of deep ocean water, increasing the surface water concentration of CO2. This mechanism limits the oceans’ capacity to take up and store further CO2 emissions and, in addition, increases the acidification of oceans, which has a negative impact on marine biodiversity.22
Tropospheric nitrous oxide cycle
In the troposphere, there is a cycle of decomposition and formation of ozone catalyzed by nitrous oxides. The cycle is influenced towards increased formation of ozone by increasing emission of carbon monoxide (CO), N2O, and other volatile organic compounds. Increasing the concentration of ozone in the troposphere contributes to the greenhouse effect. Thus, from anesthetic HFCs to N2O, anesthetic procedures can and do contribute to anthropogenic climate change on multiple levels.
“Green” inhalational anesthesia
The health and wellbeing of patients does not depend solely on the quality of health services but also on the condition of the Earth’s ecosystem. Nowadays, anesthesiologists are also challenged to consider ecological and sustainable aspects while promoting the health of their patients such as when deciding on the best way to administer GA.
General anesthesia and the choice of inhalational anesthetics
General anesthesia can either be provided as inhalational anesthesia, balanced anesthesia, or TIVA. While TIVA typically is based on a continuous intravenous application of propofol, inhalational anesthesia is delivered using an inhalational anesthetic for induction as well as for maintenance. Balanced GA is delivered using an intravenous hypnotic for induction, and maintained with a volatile inhalational agent such as sevoflurane, desflurane, isoflurane, or halothane, with or without N2O as a carrier gas.
In a 2012 French study, 72% of anesthesiologists performed GA as a combination of intravenous induction and either desflurane (48%) or sevoflurane (24%). Total intravenous anesthesia was only performed in 17% of the cases. In 8% of cases, the induction technique was not specified, and in 2% of cases, an inhalational induction was performed.23 In contrast, in a 2013 Scandinavian study, only 48.9% of anesthesiologists performed GA maintained with inhalational anesthetics, whereas 51.1% performed TIVA. In 11.9% of these GAs, N2O was used as carrier gas or coanalgesic.24
A 2019 environmental survey by the American Society of Anesthesiologists’ Committee on Equipment and Facilities found that sevoflurane was chosen by 66.4% of participating anesthesiologists (1,215/1,829; 95% confidence interval [CI], 64.2 to 68.6), whereas desflurane was chosen by 22.3% (408/1,829; 95% CI, 20.4 to 24.3).25
Inhalational anesthetics and their characteristics
Isoflurane, sevoflurane, and desflurane are halogenated ethers containing an oxygen atom that connects two fluorinated alkyl groups. Containing chlorine, isoflurane belongs to the chlorofluorocarbons (CFCs).26,27,28,29 These volatile inhalational anesthetics are liquid at room temperature and act through modulation of various membrane-associated proteins. For example, they amplify inhibitory postsynaptic potentials of γ-gamma-aminobutyric acid type A (GABAA) and glycine receptors, and decrease N-methyl-D-aspartate (NMDA) and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)-related excitatory postsynaptic potentials.30,31 Furthermore, they bind to nicotinic acetylcholine receptors, blocking the channel pore. By perturbating lipid rafts of cell membranes, they activate phospholipase D2. Consecutive activation of two-pore domain K+ channels causes reversible loss of consciousness.32 Nitrous oxide, in turn, is a colorless nonflammable gas that stimulates cerebral GABA receptors and inhibits spinal NMDA receptors.33
Inhalational anesthetics and their environmental impact
In addition to the clinical and pharmacological properties of inhalational anesthetics, it has become increasingly evident that their environmental toxicity and sustainability characteristics have to be considered as well. In 2014, inhalational anesthetics accounted for worldwide emission of approximately 3 Mt CO2e. Regarding N2O, anthropogenic sources contributed 43% of the total N2O emission of about 7.3 Tg per year, 1–4% of which originated from medical sources.20,34 Currently, inhalational anesthetics are emitted directly and unchanged from anesthesia machine-scavenging systems into the atmosphere. They influence the radiation balance of the Earth’s atmosphere negatively by intensifying IR absorption in the IR atmospheric window, affecting terrestrial regulation of temperature. In fact, they impede heat release back into space not only within but also outside the atmospheric window.6 The extent of this detrimental effect largely depends on molecular weight, specific type of halogen, and its atmospheric lifetime (Table 1).35
As stated above, isoflurane is a CFC. Chlorofluorocarbons as well as N2O (cf. above) contribute to ozone depletion in the stratosphere.10 In contrast, desflurane and sevoflurane have no ozone-depleting effect because of their complete fluorination.36 Nevertheless, comparing the GWP of inhalational anesthetics, desflurane is about 20 times more potent than sevoflurane and five times more potent than isoflurane.
The GWP of inhalational gases is determined by their atmospheric lifetime and their effectiveness at absorbing and emitting IR radiation, called “radiative efficiency.” The high GWP of desflurane is mainly due to its longer atmospheric lifetime of 14 years compared with 3.2 years for isoflurane and 1.1 years for sevoflurane. Additionally, desflurane has the highest radiative efficiency.19 It is important to notice that desflurane has a higher minimum alveolar concentration at 1 atm required to prevent 50% of subjects from moving in response to a noxious stimulus (MAC50) than sevoflurane and isoflurane (6.6 vol% vs 1.8 vol% and 1.2 vol%, respectively).37 Thus, higher concentrations are required to deliver the same clinical effect.38 In 2014, desflurane accounted for 80% of the CO2e emission related to inhalational anesthetics6.
To assess the life-cycle GHG emissions of inhalational anesthetics, it is important to consider the waste anesthetic gas emissions as well as the upstream environmental impact including manufacturing, transportation, packing, and drug delivery.16,39 Desflurane has not only the highest waste anesthetics gas emissions but also the largest nonwaste anesthetics gas emissions. The comparatively high emissions are mainly caused by manufacturing processes and electricity for volatilization during drug delivery.39 As shown by Richter et al., reduced use of desflurane in clinical practice significantly decreases the emission of CO2e due to waste anesthetics gas emissions.40 In contrast, a compensatory increase of isoflurane consumption should be avoided as it contributes to ozone depletion in the stratosphere and, therefore, counterbalances the positive effect.10
Making responsible choices with regard to the use of inhalational anesthetics during GA represents a cost-effective and quickly implementable strategy for reducing the CO2e footprint of anesthesia practice.18 |
Low-, minimal-, and metabolic flow anesthesia
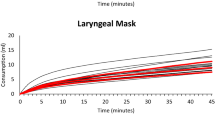
According to Baum,45 low-, minimal-, and metabolic flow anesthesia is defined by the part of rebreathing and not the absolute level of fresh gas flow (FGF) (Table 241,42,43,44). With decreasing FGF, rebreathing fraction increases (Fig. 1).45,46 The part of rebreathing depends on the oxygen consumption (\(\dot{\mathrm{V}}{\mathrm{O}}_{2})\) of the patient, FGF, and the volume of the anesthesia workstation’s circle system. Oxygen consumption can either be calculated using Fick’s formula if cardiac output (CO, in L·min-1) and arterial and venous oxygen content are known, or it can be estimated using Brody’s formula.47,48 Accordingly, a healthy 40-yr-old, 80-kg male patient with normal CO consumes about 260 mL oxygen per minute. Contemporary anesthesia machines calculate and display patients’ real-time \(\dot{\mathrm{V}}{\mathrm{O}}_{2}\) based on the partial pressure differences of inspiratory and expiratory oxygen. Therefore, FGF can be decreased to \(\dot{\mathrm{V}}{\mathrm{O}}_{2}\) to reduce waste anesthetic gas to a minimum and to save inhalational anesthetics as well as medical gases.
Contemporary anesthesia workstations with target-controlled concentrations of inspired oxygen and expiratory inhalational anesthetic allow for quantitative anesthesia in a closed-circuit system.43 Switching from conventional fresh gas mode to an electronically controlled mode such as “auto control” (Zeus IE®, Drägerwerk AG & Co. KGaA, Lübeck, Germany), “end-tidal control” (Aisys CS2®, General Electric Company, Boston, MA, USA), or “automated gas control” (FLOW-i®, Maquet, Solna, Sweden) is associated with lower consumption of inhalational anesthetics, costs, and GHG emissions.49
In general, anesthesia systems and their components can be safely used with FGFs ≤ 1 L·min-1 under the following conditions: 1) little systemic leakage; 2) mandatory measurement of inspiratory and expiratory concentration of oxygen, carbon dioxide, and inhalational anesthetics; and 3) presence of an integrated carbon dioxide absorber. It is important to prevent accidental dry-out of the carbon dioxide absorber and to use it according to the manufacturers’ instructions and recommendations.50
Intraoperative consumption of inhalational anesthetics
The overall consumption of inhalational anesthetics depends on several factors such as patient age, individual agant MAC50, duration of wash-in and wash-out periods, and time from incision to suture.
Wash-in
After intravenous induction and airway establishment, the inhalational anesthetic is washed in. Kim et al. showed that after application of an iv induction dose of 2 mg·kg-1 of propofol, depth of anesthesia is maintained for approximately nine to 11 minutes for stand-alone administration of propofol.51 Nevertheless, as GA is typically induced combining propofol with an intravenous analgesic drug (e.g., an opioid), the additive effects must be taken into account. The validated noxious stimulation response index (NSRI) can be used to predict the probability of responding to a noxious stimulus considering interactions between hypnotic and opioid.52,53
Prediction tools and previews such as SmartPilot View® (Drägerwerk AG & Co. KGaA, Lübeck, Germany) rely on validated concepts such as MAC or NSRI. They can facilitate anesthesiologic management as they visualize synergistic effects of inhalational anesthetics, intravenous hypnotics, and analgesics.
Clinical experience suggests that for desflurane, an FGF of approximately 0.7 L·min-1 with the vaporizer set to 18%, and for sevoflurane, a FGF of 0.5 L·min-1 with the vaporizer set to 8% appear to be sufficient to maintain adequate depth of anesthesia during the wash-in period. Decreasing FGF during the wash-in period helps prevent unnecessary deep anesthesia and reduces the consumption of inhalational anesthetics. For both inhalational anesthetics, a decrease of FGF from 4 to 1 L·min-1 decreases the consumption of inhalational anesthetic by 45.3% (desflurane) and 51.8% (sevoflurane), respectively. Consecutively, there is also a significant reduction by approximately 45–50% of CO2e emission and costs per minute GA (Table 3).54
Steady state
After initial wash-in of inhalational anesthetic, at a MAC50 of 0.7 to 1.0, a steady state is reached.
Anesthesia with an FGF ≤ 1 L·min-1 does not only effectively maintain intraoperative conditioning (i.e., heating and moisturizing) of inspired gases55 but also saves inhalational anesthetics. The following calculation shows the FGF-dependent consumption of sevoflurane and desflurane during steady state (Table 4). For both inhalational anesthetics, a decrease of FGF from 2 to 0.35 L·min-1 reduces consumption of sevoflurane by 65% and desflurane by 71.4%. This saves approximately 20 EURFootnote 1 and 1.1 kg of CO2e for a two-hour GA.
Routinely reducing fresh gas flow saves inhalational anesthetics, reduces CO2e emission, and is cost-effective. |
Wash-out
After completion of surgery, the administration of inhalational anesthetics can be terminated. At this point, there is no significant partial pressure difference between alveolar, blood, and cerebral compartments. To enable effective wash-out of inhalational anesthetics, the inspiratory gases must be cleared from inhalational anesthetics by closing the vapor and increasing the FGF to 4–6 L·min-1. During this period, adequate alveolar ventilation must be ensured, and spontaneous breathing efforts of the patient can be supported until the patient fully recovers.
Nitrous oxide as additional anesthetic
Nitrous oxide has been widely used as a carrier gas during balanced anesthesia, and additionally for analgesia during painful procedures (e.g., in children) or during labor, to address treatment-resistant depression, and as a strategy for preventing chronic postsurgical pain in particular populations.39,56,57 Nevertheless, the overall clinical use of N2O has declined.58 Nitrous oxide is emetogenic,59 inhibits the methionine synthase irreversibly by inactivating the enzyme’s vitamin B12 component, and can cause “diffusion hypoxia.”60,61 The ENIGMA-II trial supported the safety of N2O use in major noncardiac surgery,62 but the data remain conflicted.
When N2O is used as carrier gas instead of oxygen, the end-tidal concentration of halogenated inhalational anesthetics can be decreased, reducing the consumption of inhalational anesthetics.39,63 Nevertheless, Baum et al. showed that expenses for the use of N2O—such as technical maintenance for the central gas piping system, antiemetic prophylaxis, and the inhalational anesthetic itself—exceed the additional costs for inhalational anesthetics and intravenous analgesics.60,64 Therefore, the reduced consumption of inhalational anesthetics does not result in net savings. Comparing the total CO2e emission when N2O is used as a carrier gas, there is a net increase for sevoflurane and isoflurane and a decrease for desflurane (Table 5). Nevertheless, this calculation lacks the negative impact of additional N2O to the tropospheric nitrous oxide cycle and the resulting ozone-depleting effect.39,58
The renouncement of N2O as a carrier gas allows for safely performing low-, minimal-, and metabolic flow anesthesia. This enables significant savings of inhalational anesthetics and better conditioning of inspired gases compared with high-flow anesthesia, which is required during the wash-in and wash-out periods if N2O is used as a carrier gas. Additionally, the negative impact of N2O on CO2e emissions and ozone depletion can be prevented. |
New technologies to absorb inhalational anesthetics
The use of technologies for absorbing inhalational anesthetics from the exhausted air represents a future solution to achieve sustainability and to reduce the ecological footprint of GA.65 So far, several different purification systems received a patent grant such as adsorption by charcoal granules or by molecular sieve as well as destruction using gas-phase photochemistry.66 For example, Doyle et al. showed an effective removal of 1% isoflurane from the exhaled air over a period of eight hours using a silica zeolite hydrophobic molecular sieve adsorbent.67 Alternatively, inhalational anesthetics can be destroyed directly by UV light. The advantage of this system is the lack of additional treatment. Since these technologies are currently only used experimentally, further studies and technological development under consideration of ecological aspects are highly needed.66 It must be stressed that the efficiency of all these technologies is decreased by the use of high FGFs. Thus, minimal or metabolic flow anesthesia is essential to use these technologies beneficially in the future.68
Alternatives to inhalational anesthetics
In addition to the use of technical solutions, the transitions from inhalational anesthesia to alternative techniques such as LRA and TIVA represent another approach to reduce GHG emission.11 Locoregional anesthesia and TIVA can be used equally effectively as inhalational anesthesia if surgery and patient’s condition allows it.
Compared with inhalational anesthesia, TIVA reduces the risk of postoperative nausea and vomiting, pain scores after extubation, and duration of in postoperative anesthesia care unit stay; on the other hand, it prolongs the time to respiratory recovery and tracheal extubation.69 Nevertheless, performing TIVA instead of inhalational anesthesia has environmental costs of its own because of the increased disposable plastic waste, higher propofol consumption, and consecutively increased wastage rate.70,71 Other environmental effects of TIVA are still uncertain.71 Based on the detection of propofol metabolites in the waste water of hospitals, it is important to stress the aquatic toxicity of propofol.70,71,72,73 Propofol is excreted almost entirely in its metabolized, inactive form.74,75 Thus, it can be assumed that the detected propofol metabolites are primarily caused by improper disposal. Therefore, it is important to implement efficient waste disposal systems before converting to TIVA.
Another alternative to TIVA is LRA. A recent Australian study compared the life-cycle GHG emissions for GA with sevoflurane, spinal anesthesia, and the combination of both during total knee arthroplasty. Interestingly, the carbon footprint was similar for all three anesthetic modalities, albeit determined by serval choices. For spinal anesthesia, the emissions were mainly dominated by the electricity consumption for oxygen use and cleaning the reusable equipment. Switching from the Australian grid (mix mainly dominated by fossil energies) to the European grid (with a larger proportion of renewable energies) decreases the carbon footprint of spinal anesthesia by 40%.76 These continental differences stress the importance of defining full life-cycle assessments for different anesthetic approaches in different regions. Future analysis would benefit from datasets for medication synthesis provided by manufacturers.17,39
Strategies for “green” anesthesia
The daily practice of health care professionals is governed by the principle “First, do not harm,” and our first responsibility is to provide safe and effective anesthesia to our patients. Paradoxically, as one of the biggest GHG emitters, health care professionals harm public health.5
In the Montreal protocol, the medical use of inhalational anesthetics was declared as “essential” without restrictions. In view of global efforts to reduce the emission of greenhouse and ozone-depleting gases, it can be assumed that inhalational anesthetics will remain a significant source of emissions in the future.7,37
It is important to notice that the health care systems of low- and middle-income countries are confronted with a lack of health care resources and infrastructure, which challenges the provision of safe anesthesia. This lack of resources and infrastructure includes qualified anesthesiologic staff, equipment, and medication as well as basic needs such as continuous medical oxygen, power supply, and clean water.77 Moreover, donated equipment cannot be readily maintained and used as expertise and material are missing.78 Often, intramuscular application of ketamine or LRA are the only options to perform surgery.79 Thus, the perioperative morbidity and mortality rate (PMOR) remains high.80
These findings clearly show the global dilemma: paradoxically, high-income countries are challenged to define strategies to reduce anesthesia-related CO2 emissions, whereas low- and middle-income countries are confronted with the fundamental need to reduce PMOR and simultaneously have to face the consequences of climate change. Nevertheless, on a global perspective, we need to address this gap and it is imperative to develop solutions ensuring safe and sustainable anesthesia in the future.
Responsible choice of inhalational anesthetics
Among the inhalational anesthetics, desflurane has the largest negative ecological impact because of its substantial contribution to radiative forcing. Additionally, high volumes are necessary to maintain GA because of its high MAC50 of 6.6%. Therefore, desflurane should be avoided whenever feasible. The responsible choice of inhalational anesthetics during GA, thus, represents a cost-effective and quickly implementable solution to reduce the CO2e footprint of anesthesia practice.18
Renouncement of nitrous oxide
Nitrous oxide is a GHG with a long atmospheric lifetime. Accordingly, its use as a carrier gas does not reduce the consumption of inhalational anesthetics except for desflurane. In addition, N2O contributes a significant amount to ozone depletion. Therefore, N2O should be avoided entirely as a carrier gas.81 The use of N2O as a procedural analgesic should be considered cautiously because of its unfavorable adverse effects, taking into account alternative management including LRA.
Use of minimal or metabolic flow anesthesia
To significantly reduce the consumption of inhalational anesthetics, the lowest FGF applicable should be set. During the wash-in period, we recommend starting with an FGF of 1 L·min-1 until 0.7–1.0 MAC is reached. With increasing clinical experience, the FGF can be reduced further to 0.7–0.5 L·min-1 during the wash-in period. After that, FGF should be reduced to 0.35 L·min-1 until completion of the surgical procedure (Fig. 2). If available, we recommend using target control anesthesia in an automatically controlled mode to reduce FGF and inhalational consumption as much as possible. During GA, disconnections of breathing systems should be avoided to minimize waste of inhalational anesthetics. Prior to the wash-out period, volatile anesthetic administration must be discontinued and FGF can be increased to 8 L·min-1. We recommend using available prediction tools and previews such as SmartPilot View® (Drägerwerk AG & Co. KgaA, Lübeck, Germany), Navigator Suite (GE Healthcare, Wauwatosa, WI, USA), or Automatic Gas Control (ACG, Maquet GmbH, Getinge Group, Getinge Sweden) to facilitate anesthesiologic management.
Consider alternative anesthesia approaches
The transition to TIVA or LRA represents another alternative to reducing the environmental impact during GA if patient’s condition and surgery allow for or favors an alternative anesthesiologic management. The use of TIVA causes additional environmental costs due to propofol and plastic wastage. Nevertheless, the life-cycle carbon emission is lower than in inhalation anesthesia. Life-cycle GHG emissions of LRA mainly depend on the grid mix due to high electricity consumption. In Europe, spinal anesthesia has a smaller ecological footprint than inhalational anesthesia as the European grid mix consists of a larger part of renewable energies. Therefore, it is important to assess the life cycle of anesthesiologic management for different regions to enable a reasonable decision-making process.
Conclusion
Inhalational anesthetics remain essential for safe anesthesia. On a global perspective, it is important to reduce PMOR and anesthesia-related CO2 emissions at the same time. Responsible anesthetic management choices should prioritize patient safety and consider all available options. If inhalational anesthesia is chosen, the routine use of minimal or metabolic FGF improves conditioning of inspired gases and reduces the consumption of inhalational anesthetics significantly. In the future, new technologies to absorb and recycle inhalational anesthetics should be implemented to allow for sustainable inhalational anesthesia. Considering the ecological footprint of anesthesia, nitrous oxide should be avoided entirely as it contributes to the depletion of the ozone layer, and desflurane should only be used in justified exceptional cases.
Notes
Based on mean purchase prices of three different and independent German hospitals (cf. Table 4).
References
Costello A, Abbas M, Allen A, et al. Managing the health effects of climate change. Lancet 2009; 373 :1693–733. https://doi.org/10.1016/s0140-6736(09)60935-1
Watts N, Amann M, Arnell N, et al. The 2018 report of the Lancet Countdown on health and climate change: shaping the health of nations for centuries to come. Lancet 2018; 392: 2479–514. https://doi.org/10.1016/s0140-6736(18)32594-7
Prüss-Ustün A, Wolf J, Corvalán C, Neville T, Bos R, Neira M. Diseases due to unhealthy environments: an updated estimate of the global burden of disease attributable to environmental determinants of health. J Public Health (Oxf) 2017; 39: 464–75. https://doi.org/10.1093/pubmed/fdw085
World Health Organization. Quantitative risk assessment of the effects of climate change on selected causes of death, 2030s and 2050s, 2014. Available from URL: http://www.who.int/iris/handle/10665/134014 (accessed November 2022).
Health Care Without Harm. Health care’s climate footprint: how the health sector contributes to the global climate crisis and opportunities for action, 2019. Available from URL: https://noharm-global.org/sites/default/files/documents-files/5961/HealthCaresClimateFootprint_092319.pdf (accessed November 2022).
Kagoma YK, Stall N, Rubinstein E, Naudie D. People, planet and profits: the case for greening operating rooms. CMAJ 2012; 184: 1905–11. https://doi.org/10.1503/cmaj.112139
Vollmer MK, Rhee TS, Rigby M, et al. Modern inhalation anesthetics: potent greenhouse gases in the global atmosphere. Geophys Res Lett 2015; 42: 1606–11. https://doi.org/10.1002/2014GL062785
Chung JW, Meltzer DO. Estimate of the carbon footprint of the US health care sector. JAMA 2009; 302: 1970–2. https://doi.org/10.1001/jama.2009.1610
MacNeill AJ, Lillywhite R, Brown CJ. The impact of surgery on global climate: a carbon footprinting study of operating theatres in three health systems. Lancet Planet Health 2017; 1: e381–8. https://doi.org/10.1016/s2542-5196(17)30162-6
Sulbaek Andersen MP, Nielsen OJ, Wallington TJ, Karpichev B, Sander SP. Medical intelligence article: assessing the impact on global climate from general anesthetic gases. Anesth Analg 2012; 114: 1081–5. https://doi.org/10.1213/ane.0b013e31824d6150
Schuster M, Richter H, Pecher S, Koch S, Coburn M. Positionspapier mit konkreten Handlungsempfehlungen* der DGAI und des BDA: Ökologische Nachhaltigkeit in der Anästhesiologie und Intensivmedizin. Anästhes Intensivmed 2020; 61: 329–39. https://doi.org/10.19224/ai2020.329
McGain F, Ma SC, Burrell RH, et al. Why be sustainable? The Australian and New Zealand College of Anaesthetists Professional Document PS64: statement on environmental sustainability in anaesthesia and pain medicine practice and its accompanying background paper. Anaesth Intensive Care 2019; 47: 413–22. https://doi.org/10.1177/0310057x19884075
American Society of Anesthesiologists’. Greening the operating room and perioperative arena: environmental sustainability for anesthesia practice. Available from URL: https://www.asahq.org/about-asa/governance-and-committees/asa-committees/environmental-sustainability/greening-the-operating-room (accessed November 2022).
He H, Zhou F, Ly O, Paul J. Greener Gases Starter Pack: a tool for transitioning to more sustainable anesthetic volatile agents. Can J Anesth 2023; https://doi.org/10.1007/s12630-022-02394-y.
Brattwall M, Warren-Stomberg M, Hesselvik F, Jakobsson J. Brief review: theory and practice of minimal fresh gas flow anesthesia. Can J Anesth 2012; 59: 785–97. https://doi.org/10.1007/s12630-012-9736-2
White SM, Shelton CL. Abandoning inhalational anaesthesia. Anaesthesia 2020; 75: 451–4. https://doi.org/10.1111/anae.14853
Allen C, Baxter I. Comparing the environmental impact of inhalational anaesthesia and propofol-based intravenous anaesthesia. Anaesthesia 2021; 76: 862–3. https://doi.org/10.1111/anae.15356
Koch S, Pecher S. New challenges for anesthesia due to the climate change [German]. Anaesthesist 2020; 69: 453–62. https://doi.org/10.1007/s00101-020-00770-1
Sulbaek Andersen MP, Nielsen OJ, Karpichev B, Wallington TJ, Sander SP. Atmospheric chemistry of isoflurane, desflurane, and sevoflurane: Kinetics and mechanisms of reactions with chlorine atoms and OH radicals and global warming potentials. J Phys Chem A 2012; 116: 5806–20. https://doi.org/10.1021/jp2077598
Campbell M, Pierce JM. Atmospheric science, anaesthesia, and the environment. BJA Educ 2015; 15: 173–9. https://doi.org/10.1093/bjaceaccp/mku033
World Meteorological Organization. Scientific assessment of ozone depletion: 2018. Available from URL: https://csl.noaa.gov/assessments/ozone/2018/downloads/2018OzoneAssessment.pdf (accessed November 2022).
Lenton A, Codron F, Bopp L, et al. Stratospheric ozone depletion reduces ocean carbon uptake and enhances ocean acidification. Geophys Res Lett 2009; 36: 2896. https://doi.org/10.1029/2009GL038227
Benhamou D, Constant I, Longrois D, Molliex S, Nouette-Gaulain K, Bull A. Use of volatile anaesthetic agents in anaesthesia: a survey of practice in France in 2012. Anaesth Crit Care Pain Med 2015; 34: 205–9. https://doi.org/10.1016/j.accpm.2015.01.006
Husum B, Stenqvist O, Alahuhta S, Sigurdsson GH, Dale O. Current use of nitrous oxide in public hospitals in Scandinavian countries. Acta Anaesthesiologica Scand 2013; 57: 1131–7. https://doi.org/10.1111/aas.12165
Ard JL Jr, Tobin K, Huncke T, Kline R, Ryan SM, Bell C. A Survey of the American Society of Anesthesiologists regarding environmental attitudes, knowledge, and organization. A A Case Rep 2016; 6: 208–16. https://doi.org/10.1213/xaa.0000000000000184
National Center for Biotechnology Information. PubChem substance record for SID 319062518, 1-Chloro-2,2,2-trifluoroethyl difluoromethyl ether. Available from URL: https://pubchem.ncbi.nlm.nih.gov/substance/319062518 (accessed November 2022).
National Center for Biotechnology Information. PubChem compound summary for CID 948, Nitrous oxide. Available from URL: https://pubchem.ncbi.nlm.nih.gov/compound/Nitrous-oxide (accessed November 2022).
National Center for Biotechnology Information. PubChem compound summary for CID 5206, Sevoflurane. Available from URL: https://pubchem.ncbi.nlm.nih.gov/compound/Sevoflurane (accessed November 2022).
National Center for Biotechnology Information. PubChem compound summary for CID 42113, Desflurane. Available from URL: https://pubchem.ncbi.nlm.nih.gov/compound/Desflurane (accessed November 2022).
Garcia PS, Kolesky SE, Jenkins A. General anesthetic actions on GABA(A) receptors. Curr Neuropharmacol 2010; 8: 2–9. https://doi.org/10.2174/157015910790909502
Petrenko AB, Yamakura T, Sakimura K, Baba H. Defining the role of NMDA receptors in anesthesia: are we there yet? Eur J Pharmacol 2014; 723:29–37. https://doi.org/10.1016/j.ejphar.2013.11.039
Pavel MA, Petersen EN, Wang H, Lerner RA, Hansen SB. Studies on the mechanism of general anesthesia. Proc Natl Acad Sci U S A 2020; 117: 13757–66. https://doi.org/10.1073/pnas.2004259117
Emmanouil DE, Quock RM. Advances in understanding the actions of nitrous oxide. Anesth Prog 2007; 54: 9–18. https://doi.org/10.2344/0003-3006(2007)54[9:aiutao]2.0.co;2
Tian H, Xu R, Canadell JG, et al. A comprehensive quantification of global nitrous oxide sources and sinks. Nature 2020; 586: 248–56. https://doi.org/10.1038/s41586-020-2780-0
Hammitt JK, Camm F, Connell PS, et al. Future emission scenarios for chemicals that may deplete stratospheric ozone. Nature 1987; 330: 711–6. https://doi.org/10.1038/330711a0
Langbein T, Sonntag H, Trapp D, et al. Volatile anaesthetics and the atmosphere: atmospheric lifetimes and atmospheric effects of halothane, enflurane, isoflurane, desflurane and sevoflurane. Br J Anaesth 1999; 82: 66–73. https://doi.org/10.1093/bja/82.1.66
Eger EI. Age, minimum alveolar anesthetic concentration, and minimum alveolar anesthetic concentration-awake. Anesth Analg 2001; 93: 947–53. https://doi.org/10.1097/00000539-200110000-00029
Ryan SM, Nielsen CJ. Global warming potential of inhaled anesthetics: application to clinical use. Anesth Analg 2010; 111: 92–8. https://doi.org/10.1213/ane.0b013e3181e058d7
Sherman J, Le C, Lamers V, Eckelman M. Life cycle greenhouse gas emissions of anesthetic drugs. Anesth Analg 2012; 114: 1086–90. https://doi.org/10.1213/ane.0b013e31824f6940
Richter H, Weixler S, Schuster M. Der CO2-Fußabdruck der Anästhesie. Wie die Wahl volatiler Anästhetika die CO2-Emissionen einer anästhesiologischen Klinik beeinflusst. Anästh Intensivmed 2020; 61: 154–61. https://doi.org/10.19224/ai2020.154
Foldes FF, Ceravolo AJ, Carpenter SL. The administration of nitrous oxide-oxygen anesthesia in closed systems. Ann Surg 1952;1 36: 978–81. https://doi.org/10.1097/00000658-195212000-00009
Virtue RW. Minimal-flow nitrous oxide anesthesia. Anesthesiology 1974; 40: 196–8. https://doi.org/10.1097/00000542-197402000-00021
Lowe HJ, Mostert JW. Quantitative closed circuit anaesthesia. Anaesthesia. 1974; 29: 110–1.
Barton F, Nunn JF. Totally closed circuit nitrous oxide/oxygen anaesthesia. Br J Anaesth 1975; 47: 350–7. https://doi.org/10.1093/bja/47.3.350
Baum J. Die Inhalationsnarkose mit niedrigem Frischgasfluss: Praxis der Low-Flow- und der Minimal-Flow-Anästhesie sowie der Narkose mit geschlossenem System, 3rd ed. Stuttgart: Thieme; 1998.
Hoenemann C, Mierke B. Low-Flow-, Minimal-Flow- und Metabolic-Flow-Anästhesien: Klinische Techniken zur Nutzung von Rückatemsystemen. Lübeck: Drägerwerk AG & Co. KgaA; 2015.
Fick A. Ueber die Messung dea Blutquantums in den Herzventrikela. Sb Phys Med Ges Worzburg 1870: 36: 16–7.
Brody S. Bioenergetics and Growth: With Special Reference to the Efficiency Complex in Domestic Animals. New York: Reinhold Publishing Corporation; 1945.
Tay S, Weinberg L, Peyton P, Story D, Briedis J. Financial and environmental costs of manual versus automated control of end-tidal gas concentrations. Anaesth Intensive Care 2013; 41: 95–101. https://doi.org/10.1177/0310057x1304100116
Coppens MJ, Versichelen LF, Rolly G, Mortier EP, Struys MM. The mechanisms of carbon monoxide production by inhalational agents. Anaesthesia 2006; 61: 462–8. https://doi.org/10.1111/j.1365-2044.2006.04536.x
Kim KM, Choi BM, Park SW, et al. Pharmacokinetics and pharmacodynamics of propofol microemulsion and lipid emulsion after an intravenous bolus and variable rate infusion. Anesthesiology 2007; 106: 924–34. https://doi.org/10.1097/01.anes.0000265151.78943.af
Bouillon TW, Bruhn J, Radulescu L, et al. Pharmacodynamic interaction between propofol and remifentanil regarding hypnosis, tolerance of laryngoscopy, bispectral index, and electroencephalographic approximate entropy. Anesthesiology 2004; 100: 1353–72. https://doi.org/10.1097/00000542-200406000-00006
Luginbühl M, Schumacher PM, Vuilleumier P, et al. Noxious stimulation response index: a novel anesthetic state index based on hypnotic-opioid interaction. Anesthesiology 2010; 112: 872–80. https://doi.org/10.1097/aln.0b013e3181d40368
Garfield JM, Paskin S, Philip JH. An evaluation of the effectiveness of a computer simulation of anaesthetic uptake and distribution as a teaching tool. Med Educ 1989; 23: 457–62. https://doi.org/10.1111/j.1365-2923.1989.tb00902.x
Bengtson JP, Bengtson A, Stenqvist O. The circle system as a humidifier. Br J Anaesth 1989; 63: 453–7. https://doi.org/10.1093/bja/63.4.453
Chan MT, Peyton PJ, Myles PS, et al. Chronic postsurgical pain in the evaluation of nitrous oxide in the gas mixture for anaesthesia (ENIGMA)-II trial. Br J Anaesth 2016; 117: 801–11. https://doi.org/10.1093/bja/aew338
Nagele P, Palanca BJ, Gott B, et al. A phase 2 trial of inhaled nitrous oxide for treatment-resistant major depression. Sci Transl Med 2021; 13: eabe1376. https://doi.org/10.1126/scitranslmed.abe1376
Brown SM, Sneyd JR. Nitrous oxide in modern anaesthetic practice. BJA Educ 2016; 16: 87–91. https://doi.org/10.1093/bjaceaccp/mkv019
Divatia JV, Vaidya JS, Badwe RA, Hawaldar RW. Omission of nitrous oxide during anesthesia reduces the incidence of postoperative nausea and vomiting. A meta-analysis. Anesthesiology 1996; 85: 1055–62. https://doi.org/10.1097/00000542-199611000-00014
Ko H, Kaye AD, Urman RD. Nitrous oxide and perioperative outcomes. J Anesth 2014; 28: 420–8. https://doi.org/10.1007/s00540-013-1729-y
Brodsky JB, Cohen EN. Adverse effects of nitrous oxide. Med Toxicol 1986; 1: 362–74. https://doi.org/10.1007/bf03259849
Myles PS, Leslie K, Chan MT, et al. The safety of addition of nitrous oxide to general anaesthesia in at-risk patients having major non-cardiac surgery (ENIGMA-II): a randomised, single-blind trial. Lancet 2014; 384: 1446–54. https://doi.org/10.1016/s0140-6736(14)60893-x
Stevens WC, Dolan WM, Gibbons RT, et al. Minimum alveolar concentrations (MAC) of isoflurande with and without nitrous oxide in patients of various ages. Anesthesiology 1975; 42: 197–200. https://doi.org/10.1097/00000542-197502000-00014
Baum J, Sievert B, Stanke HG, Brauer K, Sachs G. Nitrous oxide free low-flow anesthesia [German]. Anaesthesiol Reanim 2000; 25: 60–7.
Mehrata M, Moralejo C, Anderson WA. Adsorbent comparisons for anesthetic gas capture in hospital air emissions. J Environ Sci Health A Tox Hazard Subst Environ Eng 2016; 51: 805–9. https://doi.org/10.1080/10934529.2016.1181438
Rauchenwald V, Rollins MD, Ryan SM, et al. New method of destroying waste anesthetic gases using gas-phase photochemistry. Anesth Analg 2020; 131: 288–97. https://doi.org/10.1213/ane.0000000000004119
Doyle DJ, Byrick R, Filipovic D, Cashin F. Silica zeolite scavenging of exhaled isoflurane: a preliminary report. Can J Anesth 2002; 49: 799–804. https://doi.org/10.1007/bf03017411
Barwise JA, Lancaster LJ, Michaels D, Pope JE, Berry JM. Technical communication: an initial evaluation of a novel anesthetic scavenging interface. Anesth Analg 2011; 113: 1064–7. https://doi.org/10.1213/ane.0b013e31822c9a2c
Schraag S, Pradelli L, Alsaleh AJ, et al. Propofol vs. inhalational agents to maintain general anaesthesia in ambulatory and in-patient surgery: a systematic review and meta-analysis. BMC Anesthesiol 2018; 18: 162. https://doi.org/10.1186/s12871-018-0632-3
Mankes RF. Propofol wastage in anesthesia. Anesth Analg 2012; 114: 1091–2. https://doi.org/10.1213/ane.0b013e31824ea491
Gillerman RG, Browning RA. Drug use inefficiency: a hidden source of wasted health care dollars. Anesth Analg 2000; 91: 921–4. https://doi.org/10.1097/00000539-200010000-00028
McGain F, Muret J, Lawson C, Sherman JD. Environmental sustainability in anaesthesia and critical care. Br J Anaesth 2020; 125: 680–92. https://doi.org/10.1016/j.bja.2020.06.055
Mullot JU, Karolak S, Fontova A, Levi Y. Modeling of hospital wastewater pollution by pharmaceuticals: first results of Mediflux study carried out in three French hospitals. Water Sci Technol 2010; 62: 2912–9. https://doi.org/10.2166/wst.2010.986
Dinis-Oliveira RJ. Metabolic profiles of propofol and fospropofol: clinical and forensic interpretative aspects. Biomed Res Int 2018; 2018: 6852857. https://doi.org/10.1155/2018/6852857
Dawidowicz AL, Kalityński R, Trocewicz J, Nestorowicz A, Fijałkowska A, Trela-Stachurska K. Investigation of propofol renal elimination by HPLC using supported liquid membrane procedure for sample preparation. Biomed Chromatogr 2002; 16: 455–8. https://doi.org/10.1002/bmc.183
McGain F, Sheridan N, Wickramarachchi K, Yates S, Chan B, McAlister S. Carbon footprint of general, regional, and combined anesthesia for total knee replacements. Anesthesiology 2021; 135: 976–91. https://doi.org/10.1097/aln.0000000000003967
Hendel S, Coonan T, Thomas S, McQueen K. The rate-limiting step: the provision of safe anesthesia in low-income countries. World J Surg 2015; 39: 833–41. https://doi.org/10.1007/s00268-014-2775-9
Funk LM, Weiser TG, Berry WR, et al. Global operating theatre distribution and pulse oximetry supply: an estimation from reported data. Lancet 2010; 376: 1055–61. https://doi.org/10.1016/s0140-6736(10)60392-3
Joshi GP, Onajin-Obembe B. The role of ketamine in low- and middle-income countries: what would happen if ketamine becomes a scheduled drug? Anesth Analg 2016; 122: 908–10. https://doi.org/10.1213/ane.0000000000001104
Walker IA, Bashford T, Fitzgerald JE, Wilson IH. Improving anesthesia safety in low-income regions of the world. Curr Anesthesiol Rep 2014; 4: 90–9. https://doi.org/10.1007/s40140-014-0056-7
Hönemann C, Kim SC. Please stop using nitrous oxide in routine clinical practice (comment on: Use of nitrous oxide in contemporary anesthesia-an ongoing tug of war). Can J Anesth 2022; 69: 271–2. https://doi.org/10.1007/s12630-021-02136-6
Author contributions
Se-Chan Kim and Christian Hönemann designed this review and its main conceptual ideas, Yvonne Dietzler, Miriam Kropf, and Birgit Bette provided measurements and data, Philippe Kruse performed the ecological computations, Marie L. Ruebsam aided in interpreting the results and wrote the manuscript together with Philippe Kruse. All authors provided critical feedback and revised the final manuscript.
Acknowledgements
The authors would like to thank Steffen Suermann, Kolja Fritz, and Henning Mueller from Drägerwerk AG & Co. KgaA, Lübeck, Germany, for their kind support. We thank Oliver Hauss for his editorial assistance and for improving language and grammar.
Disclosures
Christian Hönemann has received honorariums for lectures and travel expenses from Sysmex Germany, Vifor Pharma Germany, Sedana Medical, Dräger Medical GmbH & Co. KGaA, DGAI e.V., WIVIM e.V., and Hartmannbund e.V.; and research grant funding from the Patient Blood Management Academy, Germany (2020 and 2022). Marie L. Ruebsam, Philippe Kruse, Miriam Kropf, Yvonne Dietzler, Se-Chan Kim, and Birgit Bette report no conflicts of interest.
Funding statement
Open Access funding enabled and organized by Projekt DEAL.
Editorial responsibility
This submission was handled by Dr. Stephan K. W. Schwarz, Editor-in-Chief, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Rübsam, ML., Kruse, P., Dietzler, Y. et al. A call for immediate climate action in anesthesiology: routine use of minimal or metabolic fresh gas flow reduces our ecological footprint. Can J Anesth/J Can Anesth 70, 301–312 (2023). https://doi.org/10.1007/s12630-022-02393-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-022-02393-z