Abstract
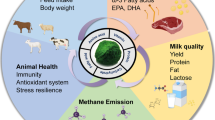
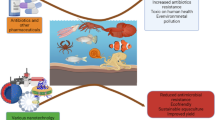
The use of novel and effective probiotic-based immunostimulating preparations, prebiotics, metabiotics, and phytobiotics is considered as a promising direction for the creation of new complex feed additives for aquaculture enterprises to increase the health and productivity of the cultivated hydrobionts. The information on the products of anaerobic solid-phase fermentation of plant substrates is presented as the basis for new probiotic-based additives for aquacultures. Biologically active feed additives ProStor and GerbaStor containing probiotics and medicinal herbs and their effects on aquaculture are discussed. Specific features of their application in aquatic environments with targeted action on hydrobionts are analyzed.





Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Castillo-Lypez RI, Gutiérrez-Grijalva EP, Leyva-López N, López-Martínez LX, Heredia JB (2017) Natural alternatives to growth-promoting antibiotics (GPA) in animal production. J Anim Plant Sci 27(2):349–359
Qi ZZ, Zhang XH, Boon N, Bossier P (2009) Probiotics in aquaculture of China. Current state, problems and prospect. Aquaculture 290(1–2):15–21. https://doi.org/10.1016/j.aquaculture.2009.02.012
Golovin PP, Golovina NA, Romanova NN (2005) Inventory of therapeutic drugs used and tested in aquaculture in Russia and abroad. FGNU “Rosinformagroteh”, Moscow, pp 5–9
Anderson JW, Gilliland SE (1999) Effect of fermented milk (yogurt) containing Lactobacillus acidophilus L1 on serum cholesterol in hypercholesterolemic humans. J Am Coll Nutr 18:43–50. https://doi.org/10.1080/07315724.1999.10718826
Balcazar JL, Blas I, Ruiz-Zarzuela I, Cunningham D, Vendrell D, Muzquiz J (2006) The role of probiotics in aquaculture. Vet Microbiol 114:173–186. 10.1016 / j. fsi.2015.05.038 https://doi.org/10.1016/j.vetmic.2006.01.009
Bruno MEC, Montville TJ (1993) Common mechanistic action of bacteriocins from lactic acid bacteria. Appl Environ Microbiol 59:3003–3010. https://doi.org/10.4172/1948-5948.1000062
Wegener HC (2003) Antibiotics in animal feed and their role in resistance development. Curr Opin Microbiol 6(5):439–445. https://doi.org/10.1016/j.mib.2003.09.009
Cabello FC (2006) Heavy use of prophylactic antibiotics in aquaculture: a growing problem for human and animal health and for the environment. Environ Microbiol 8(7):1137–1144. https://doi.org/10.1111/j.1462-2920.2006
Einar R (2020) Probiotics in shellfish aquaculture. Aquaculture and Fisheries 5:1–27. https://doi.org/10.1016/j.aaf.2019.12.001
CAC/MRL 2. (2012) Codex Alimentarius Commission. Maximum residue limits for veterinary drugs in foods. Updated as at the 35th Session of the Codex Alimentarius Commission. http://www.codexalimentarius.net/vetdrugs/data/vetdrugs/index.html
Balcázar JL, de Blas I, Ruiz-Zarzuela I, Cunningham D, Vendrell D, Múzquiz JL (2006) The role of probiotics in aquaculture. Vet Microbiol 114(3–4):173–186. https://doi.org/10.1016/j.vetmic.2006.01.009
Yukhimenko LN, Bychkova LI, Koidan GS (2001) Compound feed with probiotic as a means of preventing fish diseases. Collection of scientific works IFF: Feed Physiol Fish 77:91–95
Hill C, Guarner F, Reid G et al (2014) The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol 11:506–514. https://doi.org/10.1038/nrgastro.2014.66
Hoseinifar SH, Sun YZ, Wang A, Zhou Z (2018) Probiotics as means of diseases control in aquaculture. A review of current knowledge and future perspectives. Front Microbiol 9:2429. https://doi.org/10.3389/fmicb.2018.02429
Zorriehzahra MJ, Delshad ST, Adel M, Tiwari R, Karthik K, Dhama K, Lazado CC (2016) Probiotics as beneficial microbes in aquaculture: an update on their multiple modes of action: a review. J Vet Q 36(4):228–241. https://doi.org/10.1080/01652176.2016.1172132
Wang A, Ran C, Wang Y, Zhang Z, Ding Q, Yang Y, Olsen RE, Ring E, Bindelle J, Zhou Z (2019) Use of probiotics in aquaculture of China - a review of the past decade. Fish Shellfish Immunol 86:734–755. https://doi.org/10.1016/j.fsi.2018.12.026
Kuebutorny FKA, Abarike ED, Lu Y (2019) A review on the application of Bacillus as probiotics in aquaculture. Fish Shellfish Immunol 87:820–828. https://doi.org/10.1016/j.fsi.2019.02.010
Ravikant DT, Singh R, Singh S (2020) Applications and possible modes of action of probiotics in aquaculture. Int J Agric Sci 12(8):9753–9755
Lamsal BP (2012) Production, health aspects and potential food uses of dairy prebiotic galactooligosaccharides. J Sci Food Agric 92(10):2020–2028. https://doi.org/10.1002/jsfa.5712
Gibson G, Hutkins R, Sanders M et al (2017) Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol 14:491–502. https://doi.org/10.1038/nrgastro.2017.75
Slavin J (2013) Fiber and prebiotics: mechanisms and health benefits. Nutrients 5(4):1417–1435. https://doi.org/10.3390/nu5041417
Miller MB, Bassler BL (2001) Quorum sensing in bacteria. Annu Rev Microbiol 55:165–199. https://doi.org/10.1146/annurev.micro.55.1.165
Costi DS (2008) Quorum sensing: bacteria talk sence. Clin Infect Dis 47(8):1070–1076. https://doi.org/10.1086/592072
Navarrete P, Toledo I, Mardones P, Opazo R, Espejo R, Romero J (2010) Effect of Thymus vulgaris essential oil on intestinal bacterial microbiota of rainbow trout, Oncorhynchus mykiss (Walbaum) and bacterial isolates. Aqua Res 41:e667–e678. https://doi.org/10.1111/J.1365-2109.2010.02590.X
Yang C, Chowdhury MA, Huo Y, Gong J (2015) Phytogenic compounds as alternatives to in-feed antibiotics: potentials and challenges in application. Pathogens 4(1):137–156. https://doi.org/10.3390/pathogens4010137
Ardatskaya MD (2015) Probiotics, prebiotics and metabiotics in the correction of intestinal microecological disorders. Med Counc 13:94–99. https://doi.org/10.21518/2079-701X-2015-13-94-99
Windisch W, Schedle K, Plitzner C, Kroismayr A (2008) Use of phytogenic products as feed additives for swine and poultry. J Anim Sci 86(14):140–148. https://doi.org/10.2527/jas.2007-0459
Windisch W, Kroismayr A (2007) The effect of phytobiotics on performance and gut function in monogastrics. Biomin World Nutrition Forum. In: https://en.engormix.com/feedmachinery/articles/phytobiotics-on-performance-gut-function-in-monogastrics-t33528.htm
Salminen S, Collado MS, Endo A et al (2021) International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol. https://doi.org/10.1038/s41575-021-00440-6
Shenderov B.A. (2013) Metabiotics: a new idea or natural development of the probiotic concept. Microb Ecol Health Dis 24. 10.3402 / mehd.v24i0.20399
Gheisar MM, Kim IH (2018) Phytobiotics in poultry and swine nutrition - a review. Ital J Anim Sci 17(1):92–99. https://doi.org/10.1080/1828051X.2017.1350120
Bagno OA, Prokhorov ON, Shevchenko SA, Shevchenko AI, Dyadichkina TV (2018) Phytobiotics in feeding farm animals (review). Agric Biol 53(4):687–697. https://doi.org/10.15389/agrobiology.2018.4.687rus
DJ Newman 2008 Natural products as leads to potential drugs: an old process or the new hope for drug discovery? J Med Chem 2589–2599. https://doi.org/10.1021/jm0704090
Newman DJ, Gordon MC (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79(3):629–661. https://doi.org/10.1021/acs.jnatprod.5b01055
Manzanilla EG, Perez JF, Martin M, Kamel C, Baucells F, Gasa J (2004) Effect of plant extracts and formic acid on the intestinal equilibrium of early-weaned pigs. J Anim Sci 82:3210–3218. https://doi.org/10.4236/eng.2011.34046
Namkung H, Li M, Gong J, Yu H, Cottrill M, De Lange CFM (2004) Impact of feeding blends of organic acids and herbal extracts on growth performance, gut microbiota and digestive function in newly weaned pigs. Can J Anim Sci 84:697–704. https://doi.org/10.4141/A04-005
Adonizio AL, Downum K, Bennett BC, Mathee K (2006) Anti-quorum sensing activity of medicinal plants in southern Florida. J Ethnopharmacol 105(30):427–435. https://doi.org/10.1016/j.jep.2005.11.025
Husain FM, Ahmad I, Khan FI, Al-Shabib NA, Baig MH, Hussain A, Lobb KA (2018) Seed extract of Psoralea corylifolia and its constituent bakuchiol impairs AHL-based quorum sensing and biofilm formation in food and human-related pathogens. Front Cell Infect Microbiol 8:351. https://doi:https://doi.org/10.3389/fcimb.2018.00351
Chenia HY (2013) Anti-quorum sensing potential of crude Kigelia africana fruit extracts. J Sens 13(3):2802–2817. https://doi:https://doi.org/10.3390/s130302802
Hosoi T, Ametani A, Kiuchi K, Kaminogawa S (1999) Changes in fecal microflora induced by intubation of mice with Bacillus subtilis (natto) spores are dependent upon dietary components. Can J Microbiol 45:59–66. https://doi.org/10.1139/w98-206
Hosoi T, Kiuchi K. (2003) Natto A food made by fermenting cooked soybeans with Bacillus subtilis (natto). In: Farnworth ER (ed) Handbook of fermented functional foods, Boca Raton, Fla CRC Press, pp 227–405. https://doi.org/10.1201/9780203009727
Ushakova NA, Voznessenskaya VV, Kozlova AA, Pavlov DS (2010) Release of a somatostatin-like peptide by cells of Bacillus subtilis V-8130, an intestinal symbiont of the wild bird Tetrao urogallus: the influence of the bacillus on the animal. Dokl Biol Sci 434(1):328–331. https://doi.org/10.1134/S0012496610050108
Spencer GS, Hallett KG (1985) Somatostatin antagonist analogue stimulates growth in rats. Life Sci 37(1):27–30. https://doi.org/10.1016/0024-3205(85)90621-6
Dai J, Liu S, Hao L-l, Zhang Y, Zhang Q, Ren X-h, Jiang Q (2008) Stimultaneous expression of growth hormone releasing hormone (GHRH) and hepatitis B surface antigen/somatostatin (HBsAg/SS) fusion genes in a construct in the skeletal muscle enhances rabbit weight gain. Anim Biotec 19:104–116. https://doi.org/10.1080/10495390801888328
Melenteva GA, Antonova LA (1985) Pharmaceutical chemistry. Medicine, Moscow
Ushakova NA, Brodskiĭ ES, Kozlova AA, Nifatov AV (2009) [Anaerobic solid-phase fermentation of plant substrates by Bacillus subtilis]. Prikl Biokhim Mikrobiol. 45(1):70–7. Russian. PMID: 19235512
Ushakova NA, Nekrasov RV, Brodskiy ES, Voznesenskaya VV, Kozlova AA, Samoylenko VA, Chabaev MG, Tomgorova EK, Zelenchenkova AA (2017) Some properties of Bacillus subtilis probiotic supplements from byproducts of agricultural complex. Ukr J Ecol 7(4):597–603. https://doi.org/10.15421/2017_166
Bennet JW, Bentley R (1989) What is the name? Microbial secondary metabolism. Adv Appl Microbiol 34:1–28. https://doi.org/10.1016/S0065-2164(08)70316-2
Kaiser D, Losick R (1993) How and why bacteria talk to each other. Cell 73:873–885. https://doi.org/10.1016/0092-8674(93)90268-u
Juhas M, Eberl L, Tümmler B (2005) Quorum sensing: the power of cooperation in the world of Pseudomonas. Environ Microbiol 7(4):459–471. https://doi.org/10.1111/j.1462-2920.2005.00769.x
Pravdin VG, Kravtsova LZ, Ushakova NA (2011) Method of obtaining a complex biologically active feed additive for animals, poultry and fish with probiotics and medicinal herbs. Patent of the Russian Federation №2477614 from 11.06.2011
Кohli V, Rehman S, Rathlavath S, Aggarwal D (2018) Quorum sensing: survival strategy of microbes. Int J Curr Microbiol App Sci 7(5):165–172. https://doi.org/10.20546/ijcmas.2018.705.021
Ushakova NA, Ponomarev SV, Pravdin VG, Kravtsova LZ, Limon SA, Pavlov DS (2013) Evaluation of the effectiveness of the synbiotic drug ProStor in the diet of young sturgeon. Basic Res 6(5):1174–1177
Pavlov DS, Ushakova NA, Pravdin VG, Кravtsova LZ, Liman CA, Ponomarev SV (2014) The ProStor and Ferm KM complex probiotic additives: biotechnological innovation for enhancing the quality of domestic fish mixed feed. News in chemistry, biochemistry and biotechnology: state of the art and prospects of development. Nova Science Publishers, New York 20:239–244. 2F10.12012Fb17717-2449
Doan HV, Hoseinifar SH, Ringø E, Esteban MÁ, Dadar M, Dawood MAO, Faggio C (2020) Host-associated probiotics: a key factor in sustainable aquaculture. Rev Fish Sci Aquac 28(1):16–42. https://doi.org/10.1080/23308249.2019.1643288
Abdel-Daim MM, Dawood MA, Aleya L, Alkahtani S (2020) Effects of fucoidan on the hematic indicators and antioxidative responses of Nile tilapia (Oreochromis niloticus) fed diets contaminated with aflatoxin B1. Environ Sci Pollut Res 27:12579–12586. https://doi.org/10.1007/s11356-020-07854-w
Funding
The authors acknowledge the support of the Ministry of Science and Higher Education of the Russian Federation (contract no. 075–15-2019–1880 and state task no. 01201354245).
Author information
Authors and Affiliations
Contributions
NAU, VGP, LZK: introduction, general information on the use of probiotics in aquaculture, developers of probiotics and phytobiotics for animals, poultry, and fish. SVP, TSG, ENP: results of trials of medications, probiotics, and phytobiotics in aquaculture. DVR: content on the use of methods of phytobiotics production, preparation of the article. MLC: generalization of materials, preparation of the article.
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to Participate
For this type of study, formal consent is not required.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ushakova, N.A., Pravdin, V.G., Kravtsova, L.Z. et al. Complex Bioactive Supplements for Aquaculture—Evolutionary Development of Probiotic Concepts. Probiotics & Antimicro. Prot. 13, 1696–1708 (2021). https://doi.org/10.1007/s12602-021-09835-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-021-09835-y