Abstract
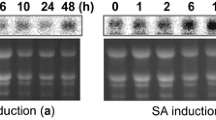
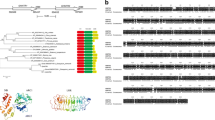
The new CaSn gene belonging to the snakin family in pepper (Capsicum annuum) encodes a novel antimicrobial peptide and responds to root-knot nematode (Meloidogyne spp.) infection. CaSn was isolated and cloned using suppression subtractive hybridization (SSH), and the gene was characterized and expressed in Escherichia coli. The CaSnakin protein encoded by CaSn is an antimicrobial peptide consisting of a signal peptide of 23 amino acid residues, an acidic peptide of 14 amino acid residues (pI = 4.18), and a mature protein of 66 amino acid residues that corresponds to a molecular mass of 7.03 kDa. The peptide sequence has 12 conserved cysteines forming six disulfide bridges. CaSnakin is highly homologous to the peptide snakin-2 (StSN2) of potato (Solanum tuberosum); CaSnakin also shows 88.5% identity to StSN2. Phylogenetic tree analysis indicated that the CaSn gene belongs to subfamily II of the snakin family. Real-time quantitative reverse transcriptase polymerase chain reaction (RT-qPCR) results showed that the CaSn gene was induced and expressed evidently by root-knot nematode infection; CaSn is also expressed in buds, stems, roots and leaves. The CaSnakin protein expressed in E. coli showed strong antimicrobial activity against free-living nematodes (Caenorhabditis elegans) and root-knot nematodes in vitro. In addition, the virus-induced gene silencing (VIGS) results revealed that the CaSn gene participates in the defense of plants against nematodes. In conclusion, the CaSn gene can be activated by nematode infections, and it plays an important role in host defense. As far as we know, this is the first investigation reporting the role of a snakin gene in the defense of plants against nematodes. In addition, the CaSn gene is the first gene of the snakin family isolated from pepper.













Similar content being viewed by others
References
Abad, P., Gouzy, J., Aury, J. M., Castagnone-Sereno, P., Danchin, E. G., Deleury, E., et al. (2008). Genome sequence of the metazoan plant-parasitic nematode Meloidogyne incognita. Nature Biotechnology, 26, 909–915.
Almasia, N. I., Bazzini, A. A., Hopp, H. E., & Vazquez-Rovere, C. (2008). Overexpression of snakin-1 gene enhances resistance to Rhizoctonia solani and Erwinia carotovora in transgenic potato plants. Molecular Plant Pathology, 9, 329–338.
Berrocal-Lobo, M., Segura, A., Moreno, M., Lopez, G., Garcia-Olmedo, F., & Molina, A. (2002). Snakin-2, an antimicrobial peptide from potato whose gene is locally induced by wounding and responds to pathogen infection. Plant Physiology, 128, 951–961.
Chan, Y. L., Yang, A. H., Chen, J. T., Yeh, K. W., & Chan, M. T. (2010). Heterologous expression of taro cystatin protects transgenic tomato against Meloidogyne incognita infection by means of interfering sex determination and suppressing gall formation. Plant Cell Reports, 29, 231–238.
Chen, K. C., Lin, C. Y., Kuan, C. C., Sung, H. Y., & Chen, C. S. (2002). A novel defensin encoded by a mungbean cDNA exhibits insecticidal activity against bruchid. Journal of Agriculture and Food Chemistry, 50, 7258–7263.
Chen, Y. Y., Lin, Y. M., Chao, T. C., Wang, J. F., Liu, A. C., Ho, F. I., et al. (2009). Virus-induced gene silencing reveals the involvement of ethylene, salicylic acid and mitogen-activated protein kinase-related defense pathways in the resistance of tomato to bacterial wilt. Physiologia Plantarum, 136, 324–335.
Dellaporta, S., Wood, J., & Hicks, J. (1983). A plant DNA minipreparation: version II. Plant Molecular Biology Reporter, 1, 19–21.
Fery, R. L., & Thies, J. A. (1997). Evaluation of Capsicum chinense Jacq. cultigens for resistance to the southern root-knot nematode. HortScience, 32, 923–926.
Garcia-Olmedo, F., Molina, A., Alamillo, J. M., & Rodriguez-Palenzuela, P. (1998). Plant defense peptides. Biopolymers, 47, 479–491.
Garcia-Olmedo, F., Rodriguez-Palenzuela, P., Molina, A., Alamillo, J. M., Lopez-Solanilla, E., Berrocal-Lobo, M., et al. (2001). Antibiotic activities of peptides, hydrogen peroxide and peroxynitrite in plant defence. FEBS Letters, 498, 219–222.
Kim, J. Y., Park, S. C., Hwang, I., Cheong, H., Nah, J. W., Hahm, K. S., et al. (2009). Protease inhibitors from plants with antimicrobial activity. International Journal of Molecular Science, 10, 2860–2872.
Kovalskaya, N., & Hammond, R. W. (2009). Expression and functional characterization of the plant antimicrobial snakin-1 and defensin recombinant proteins. Protein Expression and Purification, 63, 12–17.
Li, X. Q., Wei, J. Z., Tan, A., & Aroian, R. V. (2007). Resistance to root-knot nematode in tomato roots expressing a nematicidal Bacillus thuringiensis crystal protein. Plant Biotechnology Journal, 5, 455–464.
Lilley, C. J., Devlin, P., Urwin, P. E., & Atkinson, H. J. (1999). Parasitic nematodes, proteinases and transgenic plants. Parasitology Today, 15, 414–417.
Liu, Y., Schiff, M., & Dinesh-Kumar, S. P. (2002). Virus-induced gene silencing in tomato. The Plant Journal, 31, 777–786.
Lopez-Solanilla, E., Gonzalez-Zorn, B., Novella, S., Vazquez-Boland, J. A., & Rodriguez-Palenzuela, P. (2003). Susceptibility of Listeria monocytogenes to antimicrobial peptides. FEMS Microbiology Letters, 226, 101–105.
Mao, Z. C., Xie, B. Y., Yang, Y. H., Feng, D. X., Feng, L. X., & Yang, Z. W. (2007). Analysis of early expression genes resistance to root knot nematode in N gene pepper by SSH. Acta Horticulturae Sinica, 34, 629–636 (in Chinese).
Marroquin, L. D., Elyassnia, D., Griffitts, J. S., Feitelson, J. S., & Aroian, R. V. (2000). Bacillus thuringiensis (Bt) toxin susceptibility and isolation of resistance mutants in the nematode Caenorhabditis elegans. Genetics, 155, 1693–1699.
McCarter, J. P. (2009). Molecular approaches toward resistance to plant-parasitic nematodes. In R. H. Berg and C. G. Taylor (Eds.), Cell biology of plant nematode parasitism. Plant Cell Monographs, 15, 239–267.
Pegard, A., Brizzard, G., Fazari, A., Soucaze, O., Abad, P., & Djian-Caporalino, C. (2005). Histological characterization of resistance to different root-knot nematode species related to phenolics accumulation in Capsicum annuum. Phytopathology, 95, 158–165.
Plate, N. A., Valuev, L. I., Valueva, T. A., & Chupov, V. V. (1993). Biospecific haemosorbents based on proteinase inhibitor. I. Synthesis and properties. Biomaterials, 14, 51–56.
Segura, A., Moreno, M., Madueno, F., Molina, A., & Garcia-Olmedo, F. (1999). Snakin-1, a peptide from potato that is active against plant pathogens. Molecular Plant-Microbe Interactions, 12, 16–23.
Shai, Y. (2002). From innate immunity to de-novo designed antimicrobial peptides. Current Pharmaceutical Design, 8, 715–725.
Silverstein, K. A., Moskal, W. A., Jr., Wu, H. C., Underwood, B. A., Graham, M. A., Town, C. D., et al. (2007). Small cysteine-rich peptides resembling antimicrobial peptides have been under-predicted in plants. The Plant Journal, 51, 262–280.
Theis, J. A., & Fery, R. L. (2002). Host plant resistance as an alternative to methyl bromide for managing Meloidogyne incognita in pepper. Journal of Nematology, 34, 374–377.
Thevissen, K., Ferket, K. K., Francois, I. E., & Cammue, B. P. (2003). Interactions of antifungal plant defensins with fungal membrane components. Peptides, 24, 1705–1712.
Urwin, P. E., Atkinson, H. J., Waller, D. A., & McPherson, M. J. (1995). Engineered oryzacystatin-I expressed in transgenic hairy roots confers resistance to Globodera pallida. The Plant Journal, 8, 121–131.
Urwin, P. E., Lilley, C. J., McPherson, M. J., & Atkinson, H. J. (1997). Resistance to both cyst and root-knot nematodes conferred by transgenic Arabidopsis expressing a modified plant cystatin. The Plant Journal, 12, 455–461.
Valentine, T., Shaw, J., Blok, V., Phillips, M., Oparka, K., & Lacomme, C. (2004). Efficient virus-induced gene silencing in roots using a modified tobacco rattle virus vector. Plant Physiology, 136, 3999.
Valueva, T. A., Revina, T. A., Gvozdeva, E. L., Gerasimova, N. G., & Ozeretskovskaia, O. L. (2003). Role of proteinase inhibitors in potato protection. Bioorganic Khimica, 29, 499–504.
Vishnudasan, D., Tripathi, M. N., Rao, U., & Khurana, P. (2005). Assessment of nematode resistance in wheat transgenic plants expressing potato proteinase inhibitor (PIN2) gene. Transgenic Research, 14, 665–675.
Vivanco, J. M., Savary, B. J., & Flores, H. E. (1999). Characterization of two novel type I ribosome-inactivating proteins from the storage roots of the Andean crop Mirabilis expansa. Plant Physiology, 119, 1447–1456.
Wei, J. Z., Hale, K., Carta, L., Platzer, E., Wong, C., Fang, S. C., et al. (2003). Bacillus thuringiensis crystal proteins that target nematodes. Proceedings of the National Academy of Sciences of the United States of America, 100, 2760–2765.
Acknowledgments
This work was supported by the National Basic Research Program of China (2009CB11900), the National Natural Science Foundation of China (30971905, 30900925), Special Fund for Agro-scientific Research in the Public Interest (2011303018) and Modern Agro-industry Technology Research System. The authors would like to thank D. P. Zhang and Massimo Pugliese (University of Torino) for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mao, Z., Zheng, J., Wang, Y. et al. The new CaSn gene belonging to the snakin family induces resistance against root-knot nematode infection in pepper. Phytoparasitica 39, 151–164 (2011). https://doi.org/10.1007/s12600-011-0149-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-011-0149-5