Abstract
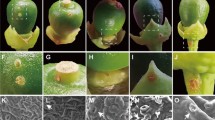
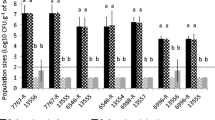
Anthracnose of tomato caused by Colletotrichum coccodes is a devastating disease of ripe fruits. This pathogen may also infect tomato roots, stems and leaves. In the present study, C. coccodes is shown to be capable of contaminating seeds collected from artificially inoculated tomato fruits. Seedlings germinating from these infected seeds exhibited disease symptoms and therefore may transmit the pathogen to the next crop. The proportion of infected seeds ranged between 20% and 63% in all C. coccodes isolates tested and correlated with the aggressiveness of the isolates to tomato fruits. Fungicidal treatment of the collected seeds reduced, but did not eliminate, seed infection. A transgenic C. coccodes isolate expressing green fluorescent protein was used to visualize the pathogen. Mycelium was observed both on surfaces of the seed coat and within 1% of the embryos.



Similar content being viewed by others
References
Ben-Daniel, B., Bar Zvi, D., & Tsror (Lahkim), L. (2009). An improved large-scale screening method for assessment of Colletotrichum coccodes aggressiveness using mature green tomato. Plant Pathology, 58, 497–503.
Byrne, J. M., Hausbeck, M. K., & Hammerschmidt, R. (1997). Conidial germination and appressorium formation of Colletotrichum coccodes on tomato foliage. Plant Disease, 81, 715–718.
Byrne, J. M., Hausbeck, M. K., Meloche, C., & Jarosz, A. M. (1998). Influence of dew period and temperature on foliar infection of greenhouse-grown tomato by Colletotrichum coccodes. Plant Disease, 82, 639–641.
Chapin, L. J., Wang, Y., Lutton, E., & Gardener, B. B. M. (2006). Distribution and fungicide sensitivity of fungal pathogens causing anthracnose-like lesions on tomatoes grown in Ohio. Plant Disease, 90, 397–403.
Dillard, H. (1992). Colletotrichum coccodes: the pathogen and its hosts. In J. Bailey & M. Jeger (Eds.), Colletotrichum: Biology, pathology and control (pp. 225–236). Wallingford, UK: CAB International.
Dillard, H. R., & Cobb, A. C. (1997). Disease progress of black dot on tomato roots and reduction in incidence with foliar applied fungicides. Plant Disease, 81, 1439–1442.
Dillard, H. R., & Cobb, A. C. (1998). Survival of Colletotrichum coccodes in infected tomato tissue and soil. Plant Disease, 82, 235–238.
Dunn, E., & Roy, A. K. (1970). Infection of tomato seedlings by Colletotrichum coccodes (Wallr.) Hughes. Plant Pathology, 19, 196–197.
Fokunang, C. N., Ikotun, T. A., Dixon, G. O., & Akem, C. N. (1997). First report of Colletotrichum gloeosporioides f. sp. manihotis, cause of cassava anthracnose disease, being seed-borne and seed-transmitted in cassava. Plant Disease, 81, 695.
Horowitz, S., Freeman, S., & Sharon, A. (2002). Use of green fluorescent protein-transgenic strains to study pathogenic and nonpathogenic lifestyles in Colletotrichum acutatum. Phytopathology, 92, 743–749.
Rubin, E., Baider, A., & Cohen, Y. (2001). Phytophthora infestans produces oospores in fruits and seeds of tomato. Phytopathology, 91, 1074–1080.
Sanogo, S., & Pennypacker, S. P. (1997). Factors affecting sporogenic and myceliogenic germination of sclerotia of Colletotrichum coccodes. Plant Disease, 81, 333–336.
Tsror (Lahkim), L., Peretz-Alon, I., & van de Haar, J. J. (2005). Epidemiology and control of blemish diseases on potato. Proceedings of the European Association of Potato Research (Bilbao, Basque Country), pp. 119–120 (abstr.).
Tu, J. C. (1980). The ontogeny of the sclerotia of Colletotrichum coccodes. Canadian Journal of Botany, 58, 631–636.
Acknowledgments
We thank M. Hazanovsky for excellent technical assistance and C. Weinstein for editing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ben-Daniel, Bh., Bar-Zvi, D. & Tsror (Lahkim), L. Transmission of Colletotrichum coccodes via tomato seeds. Phytoparasitica 38, 167–174 (2010). https://doi.org/10.1007/s12600-010-0085-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-010-0085-9