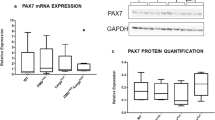
Abstract
Satellite cells exist in postnatal muscle tissue and constitute the main source of muscle precursor cells for growth and repair. These cells carry out important roles for skeletal muscle formation postnatally during growth of muscle mass as well as damage-induced regenerative processes. Muscle regeneration supports muscle function in aging and has a role in the functional impairment caused by progressive neuromuscular diseases. Major substances controlling this process are growth factors and extracellular matrix. Myostatin, a member of TGF-β family, was mainly expressed in muscle tissue. Decorin, a member of the small leucine-rich proteoglycan gene family, is composed of a core protein and a dermatan/chondroitin sulfate chain. Recent studies have shown that decorin enhanced the proliferation and differentiation of myogenic cells by suppressing myostatin activity. Thus, decorin appears to be a new molecule in the myostatin signaling pathway and a promising target for treatment of progressive neuromuscular diseases. Therefore, in this study, we examined the localization of decorin as well as myostatin in a muscular dystrophy model in mdx mice and B10 Scott Snells mice as a control to elucidate the differences between decorin and myostatin messages as well as protein distribution. This study revealed increased expression of decorin protein as well as mRNA at the regenerative stage of mdx mice compared to early stages, while only weak expression of decorin was detected in the control mice. Our study contributes to identifying the relationship between decorin and myostatin as well as the development of a therapeutic strategy for progressive neuromuscular diseases.





Similar content being viewed by others
References
Allen RE, Rankin LL (1990) Regulation of satellite cells during skeletal muscle growth and development. Proc Soc Exp Biol Med 194:81–86
Attal P, Lambert F, Marchand-Adam S, Bobin S, Pourny JC, Chemla D, Lecarpentier Y, Coirault C (2000) Severe mechanical dysfunction in pharyngeal muscle from adult mdx mice. Am J Respir Crit Care Med 162:278–281
Bernasconi P, Torchiana E, Confalonieri P, Brugnoni R, Barresi R, Mora M, Cornelio F, Morandi L, Mantegazza R (1995) Expression of transforming growth factor-beta 1 in dystrophic patient muscles correlates with fibrosis. Pathogenetic role of a fibrogenic cytokine. J Clin Invest 96:1137–1144
Bidanset DJ, Guidry C, Rosenberg LC, Choi HU, Timpl R, Hook M (1992) Binding of the proteoglycan decorin to collagen type VI. J Biol Chem 267:5250–5256
Bogdanovich S, Krag TO, Barton ER, Morris LD, Whittemore LA, Ahima RS, Khurana TS (2002) Functional improvement of dystrophic muscle by myostatin blockade. Nature 420:418–421
Border WA, Noble NA (1994) Transforming growth factor beta in tissue fibrosis. N Engl J Med 331:1286–1292
Bulfield G, Siller WG, Wight PA, Moore KJ (1984) X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc Natl Acad Sci USA 81:1189–1192
Cáceres S, Cuellar C, Casar JC, Garrido J, Schaefer L, Kresse H, Brandan E (2000) Synthesis of proteoglycans is augmented in dystrophic mdx mouse skeletal muscle. Eur J Cell Biol 79:173–181
Dangain J, Vrbova G (1984) Muscle development in mdx mutant mice. Muscle Nerve 7:700–704
Danielson KG, Baribault H, Holmes DF, Graham H, Kadler KE, Iozzo RV (1997) Targeted disruption of decorin leads to abnormal collagen fibril morphology and skin fragility. J Cell Biol 136:729–743
DiMario JX, Uzman A, Strohman RC (1991) Fiber regeneration is not persistent in dystrophic (MDX) mouse skeletal muscle. Dev Biol 148:314–321
Gillis JM (1996) Membrane abnormalities and Ca homeostasis in muscles of the mdx mouse, an animal model of the Duchenne muscular dystrophy: a review. Acta Physiol Scand 156:397–406
Gosselin LE, Williams JE, Deering M, Brazeau D, Koury S, Martinez DA (2004) Localization and early time course of TGF-beta 1 mRNA expression in dystrophic muscle. Muscle Nerve 30:645–653
Hawke TJ, Garry DJ (2001) Myogenic satellite cells: physiology to molecular biology. J Appl Physiol 91:534–551
Hill JJ, Davies MV, Pearson AA, Wang JH, Hewick RM, Wolfman NM, Qiu Y (2002) The myostatin propeptide and the follistatin-related gene are inhibitory binding proteins of myostatin in normal serum. J Biol Chem 277:40735–40741
Hoffman EP, Brown RH Jr, Kunkel LM (1987) Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 51:919–928
Kirk S, Oldham J, Kambadur R, Sharma M, Dobbie P, Bass J (2000) Myostatin regulation during skeletal muscle regeneration. J Cell Physiol 184:356–363
Kishioka Y, Thomas M, Wakamatsu J, Hattori A, Sharma M, Kambadur R, Nishimura T (2008) Decorin enhances the proliferation and differentiation of myogenic cells through suppressing myostatin activity. J Cell Physiol 215:856–867
Lee WH, Abe S, Kim HJ, Usami A, Honda A, Sakiyama K, Ide Y (2006) Characteristics of muscle fibers reconstituted in the regeneration process of masseter muscle in an mdx mouse model of muscular dystrophy. J Muscle Res Cell Motil 27:235–240
Li Y, Foster W, Deasy BM, Chan Y, Prisk V, Tang Y, Cummins J, Huard J (2004) Transforming growth factor-beta1 induces the differentiation of myogenic cells into fibrotic cells in injured skeletal muscle: a key event in muscle fibrogenesis. Am J Pathol 164:1007–1019
Li ZF, Shelton GD, Engvall E (2005) Elimination of myostatin does not combat muscular dystrophy in dy mice but increases postnatal lethality. Am J Pathol 166:491–497
Lijnen PJ, Petrov VV, Fagard RH (2000) Induction of cardiac fibrosis by transforming growth factor-beta (1). Mol Genet Metab 71:418–435
McLennan IS (1996) Degenerating and regenerating skeletal muscles contain several subpopulations of macrophages with distinct spatial and temporal distributions. J Anat 188(Pt 1):17–28
McPherron AC, Lawler AM, Lee SJ (1997) Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387:83–90
Miura T, Kishioka Y, Wakamatsu J, Hattori A, Hennebry A, Berry CJ, Sharma M, Kambadur R, Nishimura T (2006) Decorin binds myostatin and modulates its activity to muscle cells. Biochem Biophys Res Commun 340:675–678
Sara VR, Hall K (1990) Insulin-like growth factors and their binding proteins. Physiol Rev 70:591–614
Schönherr E, Hausser H, Beavan L, Kresse H (1995) Decorin-type I collagen interaction. Presence of separate core protein-binding domains. J Biol Chem 270:8877–8883
Schönherr E, Broszat M, Brandan E, Bruckner P, Kresse H (1998) Decorin core protein fragment Leu155-Val260 interacts with TGF-beta but does not compete for decorin binding to type I collagen. Arch Biochem Biophys 355:241–248
Takeuchi Y, Kodama Y, Matsumoto T (1994) Bone matrix decorin binds transforming growth factor-beta and enhances its bioactivity. J Biol Chem 269:32634–32638
Velleman SG (1999) The role of the extracellular matrix in skeletal muscle development. Poult Sci 78:778–784
Wagner KR, McPherron AC, Winik N, Lee SJ (2002) Loss of myostatin attenuates severity of muscular dystrophy in mdx mice. Ann Neurol 52:832–836
Yamaguchi Y, Mann DM, Ruoslahti E (1990) Negative regulation of transforming growth factor-beta by the proteoglycan decorin. Nature 346:281–284
Acknowledgements
This study was supported by grants-in-aid for scientific research (19592131: Shinichi Abe) from the Ministry of Education, Culture, Sports, Science and Technology, Japan, by the Foundation of the Japan Medical Association, by the Oral Health Science Center Grant HRC7 (Shinichi Abe) from Tokyo Dental College, and by a “High-Tech Research Center” Project for Private Universities: matching fund subsidy from MEXT (Ministry of Education, Culture, Sports, Science and Technology) of Japan, 2006-2011.
Author information
Authors and Affiliations
Corresponding author
Additional information
S. Abe and D. Hirose contributed equally to this work.
Rights and permissions
About this article
Cite this article
Abe, S., Hirose, D., Kado, S. et al. Increased expression of decorin during the regeneration stage of mdx mouse. Anat Sci Int 84, 305–311 (2009). https://doi.org/10.1007/s12565-009-0039-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-009-0039-9