Abstract
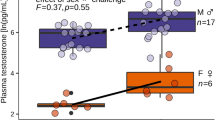
The aquaculture of greater amberjack Seriola dumerili is of considerable research interest worldwide. The larviculture methods employed to culture this species, however, are still under development, and the majority of farms still rely on wild-caught juveniles. One of the problems associated with the hatchery production of this species is the optimal selection of broodstock to ensure a stable supply of high-quality eggs. Specifically, no reliable low-stress sex-discrimination technique is currently available for selecting broodstock of this species. This study investigated the efficacy of a hormone-based sex-discrimination method in full-cycle cultured S. dumerili, ranging in age from 412 to 1150 days after hatching (DAH). Plasma concentrations of the female hormone 17β-estradiol (E2) and the male hormone 11-ketotestosterone (11-KT) were measured in both spawning and non-spawning seasons, and the optimal threshold levels for sex discrimination were estimated using a receiver operating characteristic curve. Sex discrimination using E2 produced several false positives in younger fish, and had an overall accuracy of 78.7%. However, sex discrimination using 11-KT had an accuracy of 96.7%, even in 412 DAH fish. This study demonstrated that sex discrimination using 11-KT is a reliable method for optimizing the sex ratio of S. dumerili broodstock, even before the broodstock mature.




Similar content being viewed by others
References
Agawa Y, Iwaki M, Komiya T, Honryo T, Tamura K, Okada T, Yagishita N, Kobayashi T, Sawada Y (2015) Identification of male sex-linked DNA sequence of the cultured Pacific bluefin tuna Thunnus orientalis. Fish Sci 81:113–121
Akobeng AK (2006) Understanding diagnostic tests. 3. Receiver operating characteristic curves. Acta Paediatr 96:644–647
Baroiller JF, Guiguen Y, Iseki K, Fostier A (1998) Physiological role of androgens on gonadal sex differentiation in two teleost fish, Oncorhynchus mykiss and Oreochromis niloticus. J Exp Zool 281:506–507
Borg B (1994) Androgens in teleost fishes. Comp Biochem Phys C 109:219–245
Ceapa C, Williot P, Le Menn F, Davail-Cuisset B (2002) Plasma sex steroids and vitellogenin levels in stellate sturgeon (Acipenser stellatus Pallas) during spawning migration in the Danube River. J Appl Ichthyol 18:391–396
Chu-Koo F, Dugue R, Alvan Aguilar M, Casanova Daza A, Alcantara Bocanegra F, Chavez Veintemilla C, Duponchelle F, Renno JF, Tello S, Nunez J (2009) Gender determination in the paiche or pirarucu (Arapaima gigas) using plasma vitellogenin, 17beta-estradiol, and 11-ketotestosterone levels. Fish Physiol Biochem 35:125–136
Corriero A, Medina A, Mylonas CC, Abascal FJ, Deflorio M, Aragón L (2007) Histological study of the effects of treatment with gonadotropin-releasing hormone agonist (GnRHa) on the reproductive maturation of captive-reared Atlantic bluefin tuna (Thunnus thynnus L.). Aquaculture 272:675–686
Food and Agriculture Organization of the United Nations, Fisheries and Aquaculture—Cultured Aquatic Species Information Programme, 2–3 pp—Seriola dumerili (Risso, 1810).pdf http://www.fao.org/fishery/culturedspecies/Seriola_dumerili/en. Accessed 12 June 2019
Godwin J (2010) Neuroendocrinology of sexual plasticity in teleost fishes. Front Neuroendocr 31:203–216
Grober MS, Jackson IM, Bass AH (1991) Gonadal steroids affect LHRH preoptic cell number in a sex/role changing fish. J Neurobiol 22:734–741
Hara A, Hiramatsu N, Fujita T (2016) Vitellogenesis and choriogenesis in fishes. Fish Sci 82:187–202
Koyama T, Nakamoto M, Morishima K, Toyoda A, Kikuchi K, Sakamoto T (2019) A SNP in a steroidogenic enzyme is associated with phenotypic sex in Seriola fishes. Curr Biol 29:1901–1909
Kucherka WD, Thomas P, Khan IA (2006) Sex differences in circulating steroid hormone levels in red drum, Sciaenops ocellatus L. Aquac Res 37:1464–1472
Lorenzi V, Earley RL, Grober MS (2012) Differential responses of brain, gonad and muscle steroid levels to changes in social status and sex in a sequential and bidirectional hermaphroditic fish. PLOS ONE 7:e51158
Lubzens E, Young G, Bobe J, Cerdà J (2010) Oogenesis in teleosts: how fish eggs are formed. Gen Comp Endocr 165:367–389
Miura T, Yamauchi K, Takahashi H, Nagahama Y (1991) Hormonal induction of all stages of spermatogenesis in vitro in the male Japanese eel (Anguilla japonica). Proc Natl Acad Sci USA 88:5774–5778
Nakada M (2000) Yellowtail and related species culture. In: Stickney RR (ed) Encyclopedia of aquaculture. Wiley, London, pp 1007–1036
Nakamura M, Kobayashi Y, Miura S, Alam MA, Bhandari RK (2005) Sex change in coral reef fish. Fish Physiol Biochem 31:117–122
Nakamura M, Alam MA, Kobayashi Y, Bhandari RK (2007) Role of sex hormonnes in sex change of grouper. J Mar Sci Technol 15(SPEC. ISS.):23–27
Nazeri S, Amiri BM, Nazeri MR, Mirvaghefi AR (2014) Sexing of farmed immature beluga (Huso huso) using steroid hormone levels as indicators. Comp Clin Pathol 23:631–635
Ouchi K, Adachi S, Nagahama Y (1987a) Changes in plasma levels of steroid hormones during sexual maturation of female red seabream Pagrus major. Nippon Suisan Gakkaishi 54:585–591 (in Japanese with English abstract)
Ouchi K, Adachi S, Nagahama Y (1987b) Changes in plasma levels of steroid hormones during sexual maturation of male red seabream Pagrus major. Nippon Suisan Gakkaishi 54:593–597 (in Japanese with English abstract)
Penman DJ, Piferrer F (2008) Fish gonadogenesis. Part I. Genetic and environmental mechanisms of sex determination. Rev Fish Sci 16(S1):16–34
Reinboth R (1975) In vitro studies on steroid metabolism of testicular tissue in ambisexual teleost fish. J Steroid Biochem 6:341–344
Rougeot C, Krim A, Mandiki SNM, Kestemont P, Melard C (2007) Sex steroid dynamics during embryogenesis and sexual differentioation in Eurasian perch, Perca fluviatilis. Theriogenology 67:1046–1052
Smith GH, Murie DJ, Parkyn DC (2014) Nonlethal sex discrimination of the greater amberjack, with direct application to sex ratio analysis of the Gulf of Mexico stock. Mar Coast Fish 6:200–210
Statistics of Agriculture, Forestry and Fisheries (Ministry of Agriculture, Forestry and Fisheries, 38 pp). http://www.maff.go.jp/j/tokei/kouhyou/kaimen_gyosei/attach/pdf/index-24.pdf. Accessed 12 June 2019
Strüssmann CA, Nakamura M (2002) Morphology, endocrinology, and environmental modulation of gonadal sex differentiation in teleost fishes. Fish Physiol Biochem 26:13–29
Sudo R, Tosaka R, Ijiri S, Adachi S, Aoyama J, Tsukamoto K (2012) 11-Ketotestosterone synchronously induces oocyte development and silvering-related changes in the Japanese eel, Anguilla japonica. Zool Sci 29:254–259
Takahashi N (2017) Production and sales of amberjack artificial seedlings by Kindai University. Aqua Net 2:34–37 (in Japanese)
Takaoka O (2005) Seriola dumerili. In: Kumai H (ed) Aquaculture system saltwater fish. Kouseisha-Kouseikaku, Tokyo, pp 31–43 (in Japanese)
Takemura A (1996) Early sex distinction in greater amberjack Seriola dumerili using skin mucus. Nippon Suisan Gakkaishi 62:62–67 (in Japanese with English abstract)
Torati LS, Varges APS, Galvao JA, Mesquita PE, Migaud H (2016) Endoscopy application in broodstock management of Arapaima gigas (Schinz, 1822). J Appl Ichthyol 32:353–355
Acknowledgements
We thank the staff of the Aquaculture Research Institute at Kindai University for their cooperation and guidance in conducting this research. The authors would also like to thank the staff of the Kindai University Aquaculture Seedling Center for providing the fish used in the experiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aoki, R., Chuda, H., Washio, Y. et al. Sex discrimination of cultured greater amberjack Seriola dumerili using steroid hormones. Fish Sci 86, 57–64 (2020). https://doi.org/10.1007/s12562-019-01379-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-019-01379-z