Abstract
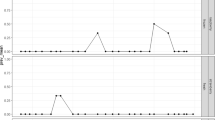
Berries have been implicated as the probable vehicle of infection in multiple outbreaks of norovirus and hepatitis A virus (HAV). These foods often receive minimal or no processing and may be exposed to virus contamination at each stage of production. In an increasingly globalized world, berries have a wide distribution and can give rise to the spread of diseases in distant parts of the world. With the aim of describing the virological quality of the berries cultivated in Argentina, a total of 184 soft fruits of different varieties (strawberries, blueberries, raspberries, blackberries, currants, pomegranate arils, cassis, and elder) were collected during the periods 2016–2018 and 2020. Viral particles were eluted and concentrated by polyethylene glycol precipitation according to ISO 15216-2:2019 guidelines. Genome detection of norovirus (NoV) genogroups I (GI) and II (GII), HAV, rotavirus, and enterovirus was performed by real-time RT-PCR with TaqMan probes. Positive samples were amplified by conventional RT-PCR and the amplicons were purified and sequenced in both directions. Phylogenetic analysis was performed using the Neighbor-Joining method based on the evolutionary model Kimura-2-parameters. NoV GII.6 was detected in 1/184 (0.5%) of the soft fruits, corresponding to a raspberry sample obtained during the fall of 2017. No presence of other human enteric viruses was found in the other berries analyzed. The collected data are the first in Argentina in relation to the prevalence of enteric viruses in berries and is useful as reference data for a risk assessment of soft fruits as vehicles of foodborne pathogenic viruses.

Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Atmar, R. L., Opekun, A. R., Gilger, M. A., Estes, M. K., Crawford, S. E., Neill, F. H., Ramani, S., Hill, H., Ferreira, J., & Graham, D. Y. (2014). Determination of the 50% human infectious dose for Norwalk virus. Journal of Infectious Diseases, 209(7), 1016–1022.
Baert, L., Mattison, K., Loisy-Hamon, F., Harlow, J., Martyres, A., Lebeau, B., Stals, A., Van Coillie, E., Herman, L., & Uyttendaele, M. (2011). Review: Norovirus prevalence in Belgian, Canadian and French fresh produce: A threat to human health? International Journal of Food Microbiology, 151(3), 261–269.
Bennett, S. D., Sodha, S. V., Ayers, T. L., Lynch, M. F., Gould, L. H., & Tauxe, R. V. (2018). Produce-associated foodborne disease outbreaks, USA, 1998–2013. Epidemiology and Infection, 146(11), 1397–1406.
Boone, I., Rosner, B., Lachmann, R., D’Errico, M. L., Iannetti, L., Van der Stede, Y., Boelaert, F., Ethelberg, S., Eckmanns, T., Stark, K., Haller, S., & Wilking, H. (2021). Healthcare-associated foodborne outbreaks in high-income countries: A literature review and surveillance study, 16 OECD countries, 2001 to 2019. Eurosurveillance Weekly, 26(41), 2001278.
Bozkurt, H., Phan-Thien, K. Y., van Ogtrop, F., Bell, T., & McConchie, R. (2021). Outbreaks, occurrence, and control of norovirus and hepatitis a virus contamination in berries: A review. Critical Reviews in Food Science and Nutrition, 61(1), 116–138.
Butot, S., Zuber, S., & Baert, L. (2014). Sample preparation prior to molecular amplification: 325 Complexities and opportunities. Current Opinion in Virology, 4, 66–70.
Centers for Disease Control and Prevention (CDC). (2018). National Outbreak Reporting System (NORS). Retrieved February 26, 2021, from https://wwwn.cdc.gov/norsdashboard/
Collier, M. G., Khudyakov, Y. E., Selvage, D., Adams-Cameron, M., Epson, E., Cronquist, A., Jervis, R. H., Lamba, K., Kimura, A. C., Sowadsky, R., Hassan, R., Park, S. Y., Garza, E., Elliott, A. J., Rotstein, D. S., Beal, J., Kuntz, T., Lance, S. E., Dreisch, R., Wise, M. E., Nelson, N. P., Suryaprasad, A., Drobeniuc, J., Holmberg, S. D., Xu, F., & Hepatitis A Outbreak Investigation Team. (2014). Outbreak of hepatitis A in the USA associated with frozen pomegranate arils imported from Turkey: An epidemiological case study. The Lancet Infectious Diseases, 14(10), 976–981.
Cook, N., Williams, L., & D’Agostino, M. (2019). Prevalence of Norovirus in produce sold at retail in the United Kingdom. Food Microbiology, 79, 85–89.
Costafreda, M. I., Bosch, A., & Pinto, R. M. (2006). Development, evaluation, and standardization of a real-time TaqMan reverse transcription-PCR assay for quantification of hepatitis A virus in clinical and shellfish samples. Applied and Environment Microbiology, 72(6), 3846–3855.
D’Souza, D. H., Moe, C. L., & Jaykus, L. (2007). Foodborne viral pathogens. In M. P. Doyle & L. R. Beuchat (Eds.), Food microbiology: Fundamentals and frontiers. ASM Press.
da Silva, A. K., Le Saux, J. C., Parnaudeau, S., Pommepuy, M., Elimelech, M., & Le Guyader, F. S. (2007). Evaluation of removal of noroviruses during wastewater treatment, using real-time reverse transcription-PCR: Different behaviors of genogroups I and II. Applied and Environment Microbiology, 73(24), 7891–7897.
De Keuckelaere, A., Li, D., Deliens, B., Stals, A., & Uyttendaele, M. (2015). Batch testing for noroviruses in frozen raspberries. International Journal of Food Microbiology, 192, 43–50.
Diakoudi, G., Lanave, G., Catella, C., Medici, M. C., De Conto, F., Calderaro, A., Loconsole, D., Chironna, M., Bonura, F., Giammanco, G. M., Bányai, K., Tohma, K., Parra, G. I., Martella, V., & De Grazia, S. (2019). Analysis of GIIP7 and GII6 noroviruses circulating in Italy during 2011–2016 reveals a replacement of lineages and complex recombination history. Infection Genetics and Evolution, 75, 103–991.
Di Pasquale, S., Paniconi, M., De Medici, D., Suffredini, E., & Croci, L. (2010). Duplex real time PCR for the detection of hepatitis A virus in shellfish using Feline Calicivirus as a process control. Journal of Virological Methods, 163(1), 96–100.
Eden, J.-S., Tanaka, M. M., Boni, M. F., Rawlinson, W. D., & White, P. A. (2013). Recombination within the pandemic norovirus GII.4 lineage. Journal of Virology, 87(11), 6270–6282.
El-Senousy, W. M., Ragab, A. M., & Handak, E. M. (2015). Prevalence of rotaviruses groups A and C in Egyptian children and aquatic environment. Food and Environmental Virology, 7(2), 132–141.
Etherington, G. J., Ring, S. M., Charleston, M. A., Dicks, J., Rayward-Smith, V. J., & Roberts, I. N. (2006). Tracing the origin and co-phylogeny of the caliciviruses. Journal of General Virology, 87(5), 1229–1235.
Evans, E. A., & Ballen, F. H. (2017). An overview of US blueberry production, trade, and consumption, with special reference to Florida. Food and Resource Economics, 952, 1–8.
Farruggia, D., Crescimanno, M., Galati, A., & Tinervia, S. (2016). The quality perception of fresh berries: An empirical survey in the German market. Agr Agr Sci Proc, 8, 566–575.
Fischer, N., Bourne, A., & Plunkett, D. (2015). Outbreak alert! 2015: A review of foodborne illnesses in the US from 2004–2013. Center for Science in the Public Interest.
Gao, X., Wang, Z., Wang, Y., Liu, Z., Guan, X., Ma, Y., Zhou, H., Jiang, Y., Cui, W., Wang, L., & Xu, Y. (2019). Surveillance of norovirus contamination in commercial fresh/frozen berries from Heilongjiang Province, China, using a TaqMan real-time RT-PCR assay. Food Microbiology, 82, 119–126.
Gould, L. H., Walsh, K. A., Vieira, A. R., Herman, K., Williams, I. T., Hall, A. J., Cole, D., & Centers for Disease Control and Prevention. (2013). Surveillance for foodborne disease outbreaks—United States, 1998–2008. MMWR Surveillance Summaries, 62(2), 1–34.
ISO 15216-2. (2013). Microbiology of food and animal feed—Horizontal method for determination of hepatitis A virus and norovirus in food using real-time RT-PCR—Part 2: Method for qualitative detection.
Kageyama, T., Kojima, S., Shinohara, M., Uchida, K., Fukushi, S., Hoshino, F. B., Takeda, N., & Katayama, K. (2003). Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. Journal of Clinical Microbiology, 41(4), 1548–1557.
Kimura, M. (1980). A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. Journal of Molecular Evolution, 16(2), 111–120.
Kitajima, M., Oka, T., Haramoto, E., Takeda, N., Katayama, K., & Katayama, H. (2010). Seasonal distribution and genetic diversity of genogroups I, II, and IV noroviruses in the Tamagawa River, Japan. Environmental Science & Technology, 44(18), 7116–7122.
Kumar, S., Stecher, G., Li, M., Knyaz, C., & Tamura, K. (2018). MEGA X: Molecular evolutionary genetics analysis across computing platforms. Molecular Biology and Evolution, 35(6), 1547–1549.
Li, Y., Sun, H., & Chen, L. (2017). The blueberry industry of China: The past 10 years and the future. Acta Hortic (wagening), 1180, 531–536.
Li, D., Butot, S., Zuber, S., & Uyttendaele, M. (2018). Monitoring of foodborne viruses in berries and considerations on the use of RT-PCR methods in surveillance. Food Control, 89, 235–240.
Loisy, F., Atmar, R. L., Guillon, P., Le Cann, P., Pommepuy, M., & Le Guyader, F. S. (2005). Real-time RT-PCR for norovirus screening in shellfish. Journal of Virological Methods, 123(1), 1–7.
Luo, L. F., Qiao, K., Wang, X. G., Ding, K. Y., Su, H. L., Li, C. Z., & Yan, H. J. (2015). Acute gastroenteritis outbreak caused by a GII.6 norovirus. World Journal of Gastroenterology, 21(17), 5295–5302.
Macori, G., Gilardi, G., Bellio, A., Bianchi, D. M., Gallina, S., Vitale, N., Gullino, M. L., & Decastelli, L. (2018). Microbiological parameters in the primary production of berries: A pilot study. Foods, 7(7), 105.
Maunula, L., Kaupke, A., Vasickova, P., Soderberg, K., Kozyra, I., Lazic, S., van der Poel, W. H., Bouwknegt, M., Rutjes, S., Willems, K. A., Moloney, R., D’Agostino, M., de Roda Husman, A. M., von Bonsdorff, C. H., Rzezutka, A., Pavlik, I., Petrovic, T., & Cook, N. (2013). Tracing enteric viruses in the European berry fruit supply chain. International Journal of Food Microbiology, 167(2), 177–185.
Nasheri, N., Vester, A., & Petronella, N. (2019). Foodborne viral outbreaks associated with frozen produce. Epidemiology and Infection, 147, e291.
Parada-Fabian, J. C., Juarez-Garcia, P., Natividad-Bonifacio, I., Vazquez-Salinas, C., & Quinones-Ramirez, E. I. (2016). Identification of enteric viruses in foods from Mexico city. Food and Environmental Virology, 8(3), 215–220.
Purcell, R. H., Wong, D. C., & Shapiro, M. (2002). Relative infectivity of hepatitis A virus by the oral and intravenous routes in 2 species of nonhuman primates. Journal of Infectious Diseases, 185(11), 1668–1671.
Read, S. J., & Kurtz, J. B. (1999). Laboratory diagnosis of common viral infections of the central nervous system by using a single multiplex PCR screening assay. Journal of Clinical Microbiology, 37(5), 1352–1355.
Red de Seguridad Alimentaria (RSA-CONICET). (2016). Foodborne viruses. Technical report. https://rsa.conicet.gov.ar/wp-content/uploads/2016/09/Informe-virus-transmitidos-por-alimentos.pdf
Ryu, S., You, H. J., Kim, Y. W., Lee, A., Ko, G. P., Lee, S. J., & Song, M. J. (2015). Inactivation of norovirus and surrogates by natural phytochemicals and bioactive substances. Molecular Nutrition & Food Research, 59, 65–74.
Scavia, G., Alfonsi, V., Taffon, S., Escher, M., Bruni, R., Medici, D., Pasquale, S. D., Guizzardi, S., Cappelletti, B., Iannazzo, S., Losio, N. M., Pavoni, E., Decastelli, L., Ciccaglione, A. R., Equestre, M., Tosti, M. E., Rizzo, C., & National Italian Task Force On Hepatitis A. (2017). A large prolonged outbreak of hepatitis A associated with consumption of frozen berries, Italy, 2013–14. Journal of Medical Microbiology, 66(3), 342–349.
Sharma, S., Hagbom, M., Carlsson, B., NederbyÖhd, J., Insulander, M., Eriksson, R., Simonsson, M., Widerström, M., & Nordgren, J. (2020). Secretor status is associated with susceptibility to disease in a large GII.6 norovirus foodborne outbreak. Food and Environmental Virology, 12(1), 28–34.
Svraka, S., Duizer, E., Vennema, H., de Bruin, E., van der Veer, B., Dorresteijn, B., & Koopmans, M. (2007). Etiological role of viruses in outbreaks of acute gastroenteritis in The Netherlands from 1994 through 2005. Journal of Clinical Microbiology, 45(5), 1389–1394.
Torok, V. A., Hodgson, K. R., Jolley, J., Turnbull, A., & McLeod, C. (2019). Estimating risk associated with human norovirus and hepatitis A virus in fresh Australian leafy greens and berries at retail. International Journal of Food Microbiology, 309, 108327.
World Health Organization (WHO). (2015). World Health Organization. Estimates of the global burden of foodborne diseases: foodborne disease burden epidemiology reference group 2007–2015. www.who.int: WHO Press.
Zhang, X. F., Chen, J. R., Song, C. L., Xie, D. J., Tan, M., Wang, L., Koroma, M. M., Hou, Y. Z., Dong, Z. P., Yu, J. R., Duan, W. T., Zhao, D. D., Du, J. R., Zhu, L., & Dai, Y. C. (2020). Characterization of a hospital-based gastroenteritis outbreak caused by GII.6 norovirus in Jinshan China. Epidemiology & Infection, 148, 289.
Zeng, S. Q., Halkosalo, A., Salminen, M., Szakal, E. D., Puustinen, L., & Vesikari, T. (2008). One-step quantitative RT-PCR for the detection of rotavirus in acute gastroenteritis. Journal of Virological Methods, 153(2), 238–240.
Funding
This work was supported by the National Agency for Scientific and Technical Promotion (PICT 2018-04224). J.M.O. and P.B. are members of the researcher career program of CONICET, Argentina. V.E.P. is a recipient of a CONICET fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oteiza, J.M., Prez, V.E., Pereyra, D. et al. Occurrence of Norovirus, Rotavirus, Hepatitis a Virus, and Enterovirus in Berries in Argentina. Food Environ Virol 14, 170–177 (2022). https://doi.org/10.1007/s12560-022-09518-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12560-022-09518-z