Abstract
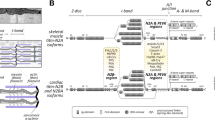
Sarcomere assembly in striated muscles has long been described as a series of steps leading to assembly of individual proteins into thick filaments, thin filaments and Z-lines. Decades of previous work focused on the order in which various structural proteins adopted the striated organization typical of mature myofibrils. These studies led to the view that actin and α-actinin assemble into premyofibril structures separately from myosin filaments, and that these structures are then assembled into myofibrils with centered myosin filaments and actin filaments anchored at the Z-lines. More recent studies have shown that particular scaffolding proteins and chaperone proteins are required for individual steps in assembly. Here, we review the evidence that N-RAP, a LIM domain and nebulin repeat protein, scaffolds assembly of actin and α-actinin into I-Z-I structures in the first steps of assembly; that the heat shock chaperone proteins Hsp90 & Hsc70 cooperate with UNC-45 to direct the folding of muscle myosin and its assembly into thick filaments; and that the kelch repeat protein Krp1 promotes lateral fusion of premyofibril structures to form mature striated myofibrils. The evidence shows that myofibril assembly is a complex process that requires the action of particular catalysts and scaffolds at individual steps. The scaffolds and chaperones required for assembly are potential regulators of myofibrillogenesis, and abnormal function of these proteins caused by mutation or pathological processes could in principle contribute to diseases of cardiac and skeletal muscles.



Similar content being viewed by others
References
Adams J, Kelso R, Cooley L (2000) The kelch repeat superfamily of proteins: propellers of cell function. Trends Cell Biol 10:17–24
Barral JM, Hutagalung AH, Brinker A, Hartl FU, Epstein HF (2002) Role of the myosin assembly protein UNC-45 as a molecular chaperone for myosin. Science 295:669–671
Bloor JW, Brown NH (1998) Genetic analysis of the Drosophila alphaPS2 integrin subunit reveals discrete adhesive, morphogenetic and sarcomeric functions. Genetics 148:1127–1142
Carroll SL, Horowits R (2000) Myofibrillogenesis and formation of cell contacts mediate the localization of N-RAP in cultured chick cardiomyocytes. Cell Motil Cytoskeleton 47:63–76
Carroll SL, Herrera AH, Horowits R (2001) Targeting and functional role of N-RAP, a nebulin-related LIM protein, during myofibril assembly in cultured chick cardiomyocytes. J Cell Sci 114:4229–4238
Carroll S, Lu S, Herrera AH, Horowits R (2004) N-RAP scaffolds I-Z-I assembly during myofibrillogenesis in cultured chick cardiomyocytes. J Cell Sci 117:105–114
Chow D, Srikakulam R, Chen Y, Winkelmann DA (2002) Folding of the striated muscle myosin motor domain. J Biol Chem 277:36799–36807
Clark KA, McElhinny AS, Beckerle MC, Gregorio CC (2002) Striated muscle cytoarchitecture: an intricate web of form and function. Annu Rev Cell Dev Biol 18:637–706
Critchley DR (2000) Focal adhesions - the cytoskeletal connection. Curr Opin Cell Biol 12:133–139
Dabiri GA, Turnacioglu KK, Sanger JM, Sanger JW (1997) Myofibrillogenesis visualized in living embryonic cardiomyocytes. Proc Natl Acad Sci USA 94:9493–9498
Dhume A, Lu S, Horowits R (2006) Targeted disruption of N-RAP gene function by RNA interference: a role for N-RAP in myofibril organization. Cell Motil Cytoskeleton 63:493–511
Dlugosz AA, Antin PB, Nachmias VT, Holtzer H (1984) The relationship between stress fiber-like structures and nascent myofibrils in cultured cardiac myocytes. J Cell Biol 99:2268–2278
Du A, Sanger JM, Linask KK, Sanger JW (2003) Myofibrillogenesis in the first cardiomyocytes formed from isolated quail precardiac mesoderm. Dev Biol 257:382–394
Du A, Sanger JM, Sanger JW (2008a) Cardiac myofibrillogenesis inside intact embryonic hearts. Dev Biol 318:236–246
Du SJ, Li H, Bian Y, Zhong Y (2008b) Heat-shock protein 90alpha1 is required for organized myofibril assembly in skeletal muscles of zebrafish embryos. Proc Natl Acad Sci USA 105:554–559
Ehler E, Rothen BM, Hämmerle SP, Komiyama M, Perriard J-C (1999) Myofibrillogenesis in the developing chicken heart: assembly of the z-disk, m-line and thick filaments. J Cell Sci 112:1529–1539
Ehler E, Horowits R, Zuppinger C, Price RL, Perriard E, Leu M, Caroni P, Sussman M, Eppenberger HM, Perriard JC (2001) Alterations at the intercalated disk associated with the absence of muscle LIM protein. J Cell Biol 153:763–772
Epstein HF, Thomson JN (1974) Temperature-sensitive mutation affecting myofilament assembly in Caenorhabditis elegans. Nature 250:579–580
Etard C, Behra M, Fischer N, Hutcheson D, Geisler R, Strahle U (2007) The UCS factor Steif/Unc-45b interacts with the heat shock protein Hsp90a during myofibrillogenesis. Dev Biol 308:133–143
Etard C, Roostalu U, Strahle U (2008) Shuttling of the chaperones Unc45b and Hsp90a between the A band and the Z line of the myofibril. J Cell Biol 180:1163–1175
Feuerstein R, Wang X, Song D, Cooke NE, Liebhaber SA (1994) The LIM/double zinc-finger motif functions as a protein dimerization domain. Proc Natl Acad Sci USA 91:10655–10659
Gehmlich K, Geier C, Osterziel KJ, Van Der Ven PF, Furst DO (2004) Decreased interactions of mutant muscle LIM protein (MLP) with N-RAP and alpha-actinin and their implication for hypertrophic cardiomyopathy. Cell Tissue Res 317:129–136
Geyer R, Wee S, Anderson S, Yates J, Wolf DA (2003) BTB/POZ domain proteins are putative substrate adaptors for cullin 3 ubiquitin ligases. Mol Cell 12:783–790
Gray CH, McGarry LC, Spence HJ, Riboldi-Tunnicliffe A, Ozanne BW (2009) Novel beta-propeller of the BTB-Kelch protein Krp1 provides a binding site for Lasp-1 that is necessary for pseudopodial extension. J Biol Chem 284:30498–30507
Greenberg CC, Connelly PS, Daniels MP, Horowits R (2008) Krp1 (Sarcosin) promotes lateral fusion of myofibril assembly intermediates in cultured mouse cardiomyocytes. Exp Cell Res 314:1177–1191
Gregorio CC, Granzier H, Sorimachi H, Labeit S (1999) Muscle assembly: a titanic achievement? Curr Opin Cell Biol 11:18–25
Handel SE, Greaser ML, Schultz E, Wang SM, Bulinski JC, Lin JJ, Lessard JL (1991) Chicken cardiac myofibrillogenesis studied with antibodies specific for titin and the muscle and nonmuscle isoforms of actin and tropomyosin. Cell Tissue Res 263:419–430
Herrera AH, Elzey B, Law DJ, Horowits R (2000) Terminal regions of mouse nebulin: sequence analysis and complementary localization with N-RAP. Cell Motil Cytoskeleton 45:211–222
Holtzer H, Hijikata T, Lin ZX, Zhang ZQ, Holtzer S, Protasi F, Franzini-Armstrong C, Sweeney HL (1997) Independent assembly of 1.6 microns long bipolar MHC filaments and I-Z-I bodies. Cell Struct Funct 22:83–93
Hutagalung AH, Landsverk ML, Price MG, Epstein HF (2002) The UCS family of myosin chaperones. J Cell Sci 115:3983–3990
Imanaka-Yoshida K (1997) Myofibrillogenesis in precardiac mesoderm explant culture. Cell Struct Funct 22:45–49
Imanaka-Yoshida K, Knudsen KA, Linask KK (1998) N-cadherin is required for the differentiation and initial myofibrillogenesis of chick cardiomyocytes. Cell Motil Cytoskeleton 39:52–62
Kim IF, Mohammadi E, Huang RC (1999) Isolation and characterization of IPP, a novel human gene encoding an actin-binding, kelch-like protein. Gene 228:73–83
Kinose F, Wang SX, Kidambi US, Moncman CL, Winkelmann DA (1996) Glycine 699 is pivotal for the motor activity of skeletal muscle myosin. J Cell Biol 134:895–909
Kontrogianni-Konstantopoulos A, Ackermann MA, Bowman AL, Yap SV, Bloch RJ (2009) Muscle giants: molecular scaffolds in sarcomerogenesis. Physiol Rev 89:1217–1267
Lin X, Miller JW, Mankodi A, Kanadia RN, Yuan Y, Moxley RT, Swanson MS, Thornton CA (2006) Failure of MBNL1-dependent post-natal splicing transitions in myotonic dystrophy. Hum Mol Genet 15:2087–2097
Liu Y, Steinacker JM (2001) Changes in skeletal muscle heat shock proteins: pathological significance. Front Biosci 6:D12–D25
Liu Y, Gampert L, Nething K, Steinacker JM (2006) Response and function of skeletal muscle heat shock protein 70. Front Biosci 11:2802–2827
Lu S, Horowits R (2008) Role of nonmuscle myosin IIB and N-RAP in cell spreading and myofibril assembly in primary mouse cardiomyocytes. Cell Motil Cytoskeleton 65:747–761
Lu S, Carroll SL, Herrera AH, Ozanne B, Horowits R (2003) New N-RAP-binding partners α-actinin, filamin and Krp1 detected by yeast two-hybrid screening: implications for myofibril assembly. J Cell Sci 116:2169–2178
Lu S, Borst DE, Horowits R (2005) N-RAP expression during mouse heart development. Dev Dyn 233:201–212
Lu S, Borst DE, Horowits R (2008) Expression and alternative splicing of N-RAP during mouse skeletal muscle development. Cell Motil Cytoskeleton 65:945–954
Luo G, Herrera AH, Horowits R (1999) Molecular interactions of N-RAP, a nebulin-related protein of striated muscle myotendon junctions and intercalated disks. Biochemistry 38:6135–6143
Luo G, Zhang JQ, Nguyen TP, Herrera AH, Paterson B, Horowits R (1997) Complete cDNA sequence and tissue localization of N-RAP, a novel nebulin-related protein of striated muscle. Cell Motil Cytoskeleton 38:75–90
Manisastry SM, Zaal KJ, Horowits R (2009) Myofibril assembly visualized by imaging N-RAP, alpha-actinin, and actin in living cardiomyocytes. Exp Cell Res 315:2126–2139
Mohiddin SA, Lu S, Cardoso J-P, Carroll SL, Jha S, Horowits R, Fananapazir L (2003) Genomic organization, alternative splicing, and expression of human and mouse N-RAP, a nebulin-related LIM protein of striated muscle. Cell Motil Cytoskeleton 55:200–212
Morano KA (2007) New tricks for an old dog: the evolving world of Hsp70. Ann NY Acad Sci 1113:1–14
Perriard JC, Hirschy A, Ehler E (2003) Dilated cardiomyopathy: a disease of the intercalated disc? Trends Cardiovasc Med 13:30–38
Price MG, Landsverk ML, Barral JM, Epstein HF (2002) Two mammalian UNC-45 isoforms are related to distinct cytoskeletal and muscle-specific functions. J Cell Sci 115:4013–4023
Rhee D, Sanger JM, Sanger JW (1994) The premyofibril: evidence for its role in myofibrillogenesis. Cell Motil Cytoskeleton 28:1–24
Risebro CA, Searles RG, Melville AA, Ehler E, Jina N, Shah S, Pallas J, Hubank M, Dillard M, Harvey NL, Schwartz RJ, Chien KR, Oliver G, Riley PR (2009) Prox1 maintains muscle structure and growth in the developing heart. Development 136:495–505
Rudy DE, Yatskievych TA, Antin PB, Gregorio CC (2001) Assembly of thick, thin, and titin filaments in chick precardiac explants. Dev Dyn 221:61–71
Sanchez-Garcia I, Axelson H, Rabbitts TH (1995) Functional diversity of LIM proteins: amino-terminal activation domains in the oncogenic proteins RBTN1 and RBTN2. Oncogene 10:1301–1306
Sanger JW, Wang J, Holloway B, Du A, Sanger JM (2009) Myofibrillogenesis in skeletal muscle cells in zebrafish. Cell Motil Cytoskeleton 66:556–566
Schultheiss T, Lin ZX, Lu MH, Murray J, Fischman DA, Weber K, Masaki T, Imamura M, Holtzer H (1990) Differential distribution of subsets of myofibrillar proteins in cardiac nonstriated and striated myofibrils. J Cell Biol 110:1159–1172
Schwander M, Leu M, Stumm M, Dorchies OM, Ruegg UT, Schittny J, Muller U (2003) Beta1 integrins regulate myoblast fusion and sarcomere assembly. Dev Cell 4:673–685
Soltysik-Espanola M, Rogers RA, Jiang S, Kim TA, Gaedigk R, White RA, Avraham H, Avraham S (1999) Characterization of Mayven, a novel actin-binding protein predominantly expressed in brain. Mol Biol Cell 10:2361–2375
Sparrow JC, Schock F (2009) The initial steps of myofibril assembly: integrins pave the way. Nat Rev Mol Cell Biol 10:293–298
Spence HJ, Johnston I, Ewart K, Buchanan SJ, Fitzgerald U, Ozanne BW (2000) Krp1, a novel kelch related protein that is involved in pseudopod elongation in transformed cells. Oncogene 19:1266–1276
Spence HJ, McGarry L, Chew CS, Carragher NO, Scott-Carragher LA, Yuan Z, Croft DR, Olson MF, Frame M, Ozanne BW (2006) AP-1 differentially expressed proteins Krp1 and fibronectin cooperatively enhance Rho-ROCK-independent mesenchymal invasion by altering the function, localization, and activity of nondifferentially expressed proteins. Mol Cell Biol 26:1480–1495
Srikakulam R, Winkelmann DA (1999) Myosin II folding is mediated by a molecular chaperonin. J Biol Chem 274:27265–27273
Srikakulam R, Winkelmann DA (2004) Chaperone-mediated folding and assembly of myosin in striated muscle. J Cell Sci 117:641–652
Srikakulam R, Liu L, Winkelmann DA (2008) Unc45b forms a cytosolic complex with Hsp90 and targets the unfolded myosin motor domain. PLoS ONE 3:e2137
Stogios PJ, Prive GG (2004) The BACK domain in BTB-kelch proteins. Trends Biochem Sci 29:634–637
Taylor A, Obholz K, Linden G, Sadiev S, Klaus S, Carlson KD (1998) DNA sequence and muscle-specific expression of human sarcosin transcripts. Mol Cell Biochem 183:105–112
Tullio AN, Accili D, Ferrans VJ, Yu ZX, Takeda K, Grinberg A, Westphal H, Preston YA, Adelstein RS (1997) Nonmuscle myosin II-B is required for normal development of the mouse heart. Proc Natl Acad Sci USA 94:12407–12412
Volk T, Fessler LI, Fessler JH (1990) A role for integrin in the formation of sarcomeric cytoarchitecture. Cell 63:525–536
Wang K (1985) Sarcomere-associated cytoskeletal lattices in striated muscle. Review and hypothesis. Cell Muscle Motil 6:315–369
Wang SM, Greaser ML, Schultz E, Bulinski JC, Lin JJ, Lessard JL (1988) Studies on cardiac myofibrillogenesis with antibodies to titin, actin, tropomyosin, and myosin. J Cell Biol 107:1075–1083
Wang K, Knipfer M, Huang QQ, van Heerden A, Hsu LC, Gutierrez G, Quian XL, Stedman H (1996) Human skeletal muscle nebulin sequence encodes a blueprint for thin filament architecture: sequence motifs and affinity profiles of tandem repeats and terminal SH3. J Biol Chem 271:4304–4314
Wegele H, Wandinger SK, Schmid AB, Reinstein J, Buchner J (2006) Substrate transfer from the chaperone Hsp70 to Hsp90. J Mol Biol 356:802–811
Whitesell L, Lindquist SL (2005) HSP90 and the chaperoning of cancer. Nat Rev Cancer 5:761–772
Willis MS, Schisler JC, Portbury AL, Patterson C (2009) Build it up-Tear it down: protein quality control in the cardiac sarcomere. Cardiovasc Res 81:439–448
Wohlgemuth SL, Crawford BD, Pilgrim DB (2007) The myosin co-chaperone UNC-45 is required for skeletal and cardiac muscle function in zebrafish. Dev Biol 303:483–492
Xu L, Wei Y, Reboul J, Vaglio P, Shin TH, Vidal M, Elledge SJ, Harper JW (2003) BTB proteins are substrate-specific adaptors in an SCF-like modular ubiquitin ligase containing CUL-3. Nature 425:316–321
Zhang JQ, Elzey B, Williams G, Lu S, Law DJ, Horowits R (2001) Ultrastructural and biochemical localization of N-RAP at the interface between myofibrils and intercalated disks in the mouse heart. Biochemistry 40:14898–14906
Zhang DD, Lo SC, Sun Z, Habib GM, Lieberman MW, Hannink M (2005) Ubiquitination of Keap1, a BTB-Kelch substrate adaptor protein for Cul3, targets Keap1 for degradation by a proteasome-independent pathway. J Biol Chem 280:30091–30099
Zhao R, Houry WA (2007) Molecular interaction network of the Hsp90 chaperone system. Adv Exp Med Biol 594:27–36
Acknowledgement
This research was supported by the Intramural Research Program of the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Crawford, G.L., Horowits, R. Scaffolds and chaperones in myofibril assembly: putting the striations in striated muscle. Biophys Rev 3, 25–32 (2011). https://doi.org/10.1007/s12551-011-0043-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-011-0043-x