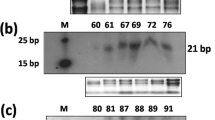
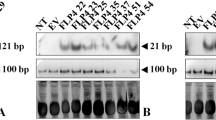
Abstract
The fungus Fusarium verticillioides is a maize pathogen that can produce fumonisin mycotoxins in ears under certain environmental conditions. Because fumonisins pose health risks to humans and livestock, control strategies with minimal risk to the environment are needed to reduce fumonisin contamination. Host-induced gene silencing is a promising technique in which double-stranded RNA expressed in the plant host is absorbed by an invading fungus and down-regulates genes critical for pathogenicity or mycotoxin production in the fungus. A key preliminary step of this technique is identification of DNA segments within the targeted fungal gene that can effectively silence the gene. Here, we used segments of the fumonisin biosynthetic gene FUM1 to generate double-stranded RNA in F. verticillioides. Several of the resulting transformants exhibited reduced FUM1 gene expression and fumonisin production (24- to 3675-fold reduction in fumonisin FB1). Similar reductions in fumonisin production resulted from double-stranded RNA constructs with segments of FUM8, another fumonisin biosynthetic gene (3.5- to 2240-fold reduction in fumonisin FB1). FUM1 or FUM8 silencing constructs were transformed into three isolates of F. verticillioides. Whole genome sequence analysis of seven transformants revealed that reductions in fumonisin production were not due to mutation of the fumonisin biosynthetic gene cluster and revealed a complex pattern of plasmid integration. These results suggest the cloned FUM1 or FUM8 gene segments could be expressed in maize for host-induced gene silencing of fumonisin production.


Similar content being viewed by others
References
Baulcombe D (2004) RNA silencing in plants. Nature 431:356–363. https://doi.org/10.1038/nature02874
Baum JA, Bogaert T, Clinton W, Heck GR, Feldmann P, Ilagan O, Johnson S, Plaetinck G, Munyikwa T, Pleau M, Vaughn T, Roberts J (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25:1322–1326. https://doi.org/10.1038/nbt1359
Brown DW, Lee S-H, Kim L-H, Ryu J-G, Lee S, Seo Y, Kim YH, Busman M, Yun S-H, Proctor RH, Lee T (2015) Identification of a 12-gene fusaric acid biosynthetic gene cluster in Fusarium species through comparative and functional genomics. Mol Plant-Microbe Interact 28:319–332. https://doi.org/10.1094/MPMI-09-14-0264-R
Chen Y, Gao Q, Huang M, Liu Y, Liu Z, Liu X, Ma Z (2015) Characterization of RNA silencing components in the plant pathogenic fungus Fusarium graminearum. Sci Rep 5:12500. https://doi.org/10.1038/srep12500
Cheng W, Song X-S, Li H-P, Cao L-H, Sun K, Qiu X-L, Xu Y-B, Yang P, Huang T, Zhang J-B, Qu B, Liao Y-C (2015) Host-induced gene silencing of an essential chitin synthase gene confers durable resistance to Fusarium head blight and seedling blight in wheat. Plant Biotechnol J 13:1335–1345. https://doi.org/10.1111/pbi.12352
da Silva VN, de Araujo J, Durigon EL, Corrêa B (2007) Sequence variability in the FUM1 gene of Fusarium verticillioides strains. Can J Microbiol 53:446–449. https://doi.org/10.1139/W06-135
Desjardins AE, Proctor RH (1999) Biochemistry and genetics of Fusarium toxins. In: Summerell B, Leslie JF, Backhouse D, Bryden WL, Burgess LW (eds) Fusarium: Paul E Nelson Symposium. APS Press, St. Paul, MN
Dowd PF, Johnson ET (2015) Environmental effects on resistance gene expression in milk stage popcorn kernels and associations with mycotoxin production. Mycotoxin Res 31:63–82. https://doi.org/10.1007/s12550-014-0215-1
Fagard M, Boutet S, Morel JB, Bellini C, Vaucheret H (2000) AGO1, QDE-2, and RDE-1 are related proteins required for post-transcriptional gene silencing in plants, quelling in fungi, and RNA interference in animals. Proc Natl Acad Sci U S A 97:11650–11654. https://doi.org/10.1073/pnas.200217597
Fernandes JS, Angelo PCS, Cruz JC, Santos JMM, Sousa NR, Silva GF (2016) Post-transcriptional silencing of the SGE1 gene induced by a dsRNA hairpin in Fusarium oxysporum f. sp cubense, the causal agent of Panama disease. Genet Mol Res. https://doi.org/10.4238/gmr.15027941
Fire AZ (2007) Gene silencing by double-stranded RNA. Cell Death Differ 14:1998–2012. https://doi.org/10.1038/sj.cdd.4402253
Govindarajulu M, Epstein L, Wroblewski T, Michelmore RW (2015) Host-induced gene silencing inhibits the biotrophic pathogen causing downy mildew of lettuce. Plant Biotechnol J 13:875–883. https://doi.org/10.1111/pbi.12307
Howard PC, Eppley RM, Stack ME, Warbritton A, Voss KA, Lorentzen RJ, Kovach RM, Bucci TJ (2001) Fumonisin B1 carcinogenicity in a two-year feeding study using F344 rats and B6C3F1 mice. Environ Health Perspect 109(Suppl 2):277–282
Hu Y, Hao X, Lou J, Zhang P, Pan J, Zhu X (2012) A PKS gene, pks-1, is involved in chaetoglobosin biosynthesis, pigmentation and sporulation in Chaetomium globosum. Sci China Life Sci 55:1100–1108. https://doi.org/10.1007/s11427-012-4409-5
Jiménez M, Mateo JJ, Hinojo MJ, Mateo R (2003) Sugars and amino acids as factors affecting the synthesis of fumonisins in liquid cultures by isolates of the Gibberella fujikuroi complex. Int J Food Microbiol 89:185–193
Johnson ET, Dowd PF (2016) A quantitative method for determining relative colonization rates of maize callus by Fusarium graminearum for resistance gene evaluations. J Microbiol Methods 130:73–75. https://doi.org/10.1016/j.mimet.2016.08.026
Ketting RF (2011) The many faces of RNAi. Dev Cell 20:148–161. https://doi.org/10.1016/j.devcel.2011.01.012
Knip M, Constantin ME, Thordal-Christensen H (2014) Trans-kingdom cross-talk: small RNAs on the move. PLoS Genet 10:e1004602. https://doi.org/10.1371/journal.pgen.1004602
Koch A, Kumar N, Weber L, Keller H, Imani J, Kogel KH (2013) Host-induced gene silencing of cytochrome P450 lanosterol C14α-demethylase-encoding genes confers strong resistance to Fusarium species. Proc Natl Acad Sci U S A 110:19324–19329. https://doi.org/10.1073/pnas.1306373110
Leslie JF, Plattner RD, Desjardins AE, Klittich CJ (1992) Fumonisin B1 production by strains from different mating populations of Gibberella fujikuroi (Fusarium section Liseola). Mycotoxicology 82:341–345
Liu Q, Paroo Z (2010) Biochemical principles of small RNA pathways. Annu Rev Biochem 79:295–319. https://doi.org/10.1146/annurev.biochem.052208.151733
Liu W, Saint DA (2002) A new quantitative method of real time reverse transcription polymerase chain reaction assay based on simulation of polymerase chain reaction kinetics. Anal Biochem 302:52–59. https://doi.org/10.1006/abio.2001.5530
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Majumdar R, Rajasekaran K, Cary JW (2017) RNA interference (RNAi) as a potential tool for control of mycotoxin contamination in crop plants: concepts and considerations. Front Plant Sci 8:200. https://doi.org/10.3389/fpls.2017.00200
Marasas WFO, Riley RT, Hendricks KA et al (2004) Fumonisins disrupt sphingolipid metabolism, folate transport, and neural tube development in embryo culture and in vivo: a potential risk factor for human neural tube defects among populations consuming fumonisin-contaminated maize. J Nutr 134:711–716
Masanga JO, Matheka JM, Omer RA, Ommeh SC, Monda EO, Alakonya AE (2015) Downregulation of transcription factor aflR in Aspergillus flavus confers reduction to aflatoxin accumulation in transgenic maize with alteration of host plant architecture. Plant Cell Rep 34:1379–1387. https://doi.org/10.1007/s00299-015-1794-9
McDonald T, Brown D, Keller NP, Hammond TM (2005) RNA silencing of mycotoxin production in Aspergillus and Fusarium species. Mol Plant-Microbe Interact 18:539–545. https://doi.org/10.1094/MPMI-18-0539
Morgavi DP, Riley RT (2007) An historical overview of field disease outbreaks known or suspected to be caused by consumption of feeds contaminated with Fusarium toxins. Anim Feed Sci Technol 137:201–212. https://doi.org/10.1016/j.anifeedsci.2007.06.002
Munkvold GP (2003) Epidemiology of Fusarium diseases and their mycotoxins in maize ears. Eur J Plant Pathol 109:705–713. https://doi.org/10.1023/A:1026078324268
Narva K, Geng C, Olson MB, Li H, Larrinua IM, Elango NJ (2015) United States Patent: 9102946—Nucleic acid molecules that confer resistance to coleopteran pests
Niño-Sánchez J, Casado-Del Castillo V, Tello V, de Vega-Bartol JJ, Ramos B, Sukno SA, Díaz Mínguez JM (2016) The FTF gene family regulates virulence and expression of SIX effectors in Fusarium oxysporum. Mol Plant Pathol 17:1124–1139. https://doi.org/10.1111/mpp.12373
Plattner RD (1999) HPLC/MS analysis of Fusarium mycotoxins, fumonisins and deoxynivalenol. Nat Toxins 7:365–370. https://doi.org/10.1002/1522-7189(199911/12)7:6<365::AID-NT85>3.0.CO;2-0
Proctor RH, Desjardins AE, Plattner RD, Hohn TM (1999) A polyketide synthase gene required for biosynthesis of fumonisin mycotoxins in Gibberella fujikuroi mating population A. Fungal Genet Biol 27:100–112. https://doi.org/10.1006/fgbi.1999.1141
Shimizu T, Ito T, Kanematsu S (2014) Functional analysis of a melanin biosynthetic gene using RNAi-mediated gene silencing in Rosellinia necatrix. Fungal Biol 118:413–421. https://doi.org/10.1016/j.funbio.2014.02.006
Thakare D, Zhang J, Wing RA, Cotty PJ, Schmidt MA (2017) Aflatoxin-free transgenic maize using host-induced gene silencing. Sci Adv 3:e1602382. https://doi.org/10.1126/sciadv.1602382
Tinoco MLP, Dias BBA, Dall’Astta RC, Pamphile JA, Aragão FJL (2010) In vivo trans-specific gene silencing in fungal cells by in planta expression of a double-stranded RNA. BMC Biol 8:27. https://doi.org/10.1186/1741-7007-8-27
Urban M, King R, Hassani-Pak K, Hammond-Kosack KE (2015) Whole-genome analysis of Fusarium graminearum insertional mutants identifies virulence associated genes and unmasks untagged chromosomal deletions. BMC Genomics 16:261. https://doi.org/10.1186/s12864-015-1412-9
Zhang G, Sun Z, Ren A, Shi L, Shi D, Li X, Zhao M (2017) The mitogen-activated protein kinase GlSlt2 regulates fungal growth, fruiting body development, cell wall integrity, oxidative stress and ganoderic acid biosynthesis in Ganoderma lucidum. Fungal Genet Biol 104:6–15. https://doi.org/10.1016/j.fgb.2017.04.004
Zhao S, Fernald RD (2005) Comprehensive algorithm for quantitative real-time polymerase chain reaction. J Comput Biol 12:1047–1064. https://doi.org/10.1089/cmb.2005.12.1047
Acknowledgements
We are grateful to Marcie Moore, Heather Walker and Mark Doehring for technical assistance. The mention of trade names or commercial products in this paper is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U. S. Department of Agriculture over other firms or similar products not mentioned. USDA is an equal opportunity provider and employer.
Funding
This study was supported by USDA CRIS Projects 5010-42000-047 (-049 and -050)-00D base funds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments comply with current laws of the USA.
Conflict of interest
None. The authors have full control over primary data, which is available for review by the journal.
Electronic supplementary material
ESM 1
(DOCX 13963 kb)
Rights and permissions
About this article
Cite this article
Johnson, E.T., Proctor, R.H., Dunlap, C.A. et al. Reducing production of fumonisin mycotoxins in Fusarium verticillioides by RNA interference. Mycotoxin Res 34, 29–37 (2018). https://doi.org/10.1007/s12550-017-0296-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12550-017-0296-8