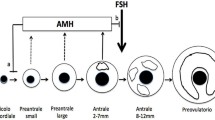
Abstract
Ovarian reserve reflects the quality and quantity of available oocytes. This reserve has become indispensable for the better understanding of reproductive potential. Measurement of the serum anti-Müllerian hormone (AMH) level allows quantitative evaluation of ovarian reserve. It has been applied to a wide range of clinical conditions, and it is well established that the measurement of serum AMH levels is more useful than qualitative evaluation based on the menstrual cycle. AMH levels are monitored during infertility treatments; in patients undergoing medically assisted reproductive technology; and in the diagnosis of ovarian failure, polycystic ovarian syndrome, and granulosa cell tumor. It is also useful in the evaluation of iatrogenic ovarian damage. Population-based studies have indicated a potential role for serum AMH in the planning of reproductive health management. While AMH is currently the best measure of ovarian reserve, its predictive value for future live births remains controversial. Furthermore, there is a serious practical issue in the interpretation of test results, as currently available assay kits use different assay ranges and coefficients of variation due to the absence of an international reference standard. The pros and cons of the serum AMH level as a definitive measure of ovarian reserve merits further review in order to guide future research.
Similar content being viewed by others
References
Broekmans FJ, Kwee J, Hendriks DJ, Mol BW, Lambalk CB. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update. 2006;12(6):685–718.
Durlinger AL, Gruijters MJ, Kramer P, Karels B, Ingraham HA, Nachtigal MW, Uilenbroek JT, Grootegoed JA, Themmen AP. Anti-Müllerian hormone inhibits initiation of primordial follicle growth in the mouse ovary. Endocrinology. 2002;143(3):1076–84.
Weenen C, Laven JS, Von Bergh AR, Cranfield M, Groome NP, Visser JA, Kramer P, Fauser BC, Themmen AP. Anti-Müllerian hormone expression pattern in the human ovary: potential implications for initial and cyclic follicle recruitment. Mol Hum Reprod. 2004;10(2):77–83.
Anderson RA, Nelson SM, Wallace WH. Measuring anti-Müllerian hormone for the assessment of ovarian reserve: when and for whom is it indicated? Maturitas. 2012;71(1):28–33.
de Vet A, Laven JS, de Jong FH, Themmen AP, Fauser BC. Anti-Müllerian hormone serum levels: a putative marker for ovarian aging. Fertil Steril. 2002;77(2):357–62.
Fanchin R, Schonauer LM, Righini C, Guibourdenche J, Frydman R, Taieb J. Serum anti-Müllerian hormone is more strongly related to ovarian follicular status than serum inhibin B, estradiol, FSH and LH on day 3. Hum Reprod. 2003;18(2):323–7.
van Rooij IA, Broekmans FJ, Scheffer GJ, Looman CW, Habbema JD, de Jong FH, Fauser BJ, Themmen AP, te Velde ER. Serum anti-Müllerian hormone levels best reflect the reproductive decline with age in normal women with proven fertility: a longitudinal study. Fertil Steril. 2005;83(4):979–87.
La Marca A, Sighinolfi G, Radi D, Argento C, Baraldi E, Artenisio AC, Stabile G, Volpe A. Anti-Müllerian hormone (AMH) as a predictive marker in assisted reproductive technology (ART). Hum Reprod Update. 2010;16(2):113–30.
van Disseldorp J, Lambalk CB, Kwee J, Looman CW, Eijkemans MJ, Fauser BC, Broekmans FJ. Comparison of inter- and intra-cycle variability of anti-Müllerian hormone and antral follicle counts. Hum Reprod. 2010;25(1):221–7.
Loh JS, Maheshwari A. Anti-Müllerian hormone–is it a crystal ball for predicting ovarian ageing? Hum Reprod. 2011;26(11):2925–32.
Nelson SM. Biomarkers of ovarian response: current and future applications. Fertil Steril. 2013;99(4):963–9.
Pandian Z, Marjoribanks J, Ozturk O, Serour G, Bhattacharya S. Number of embryos for transfer following in vitro fertilisation or intra-cytoplasmic sperm injection. Cochrane Database Syst Rev. 2013;7:CD003416.
Seifer DB, MacLaughlin DT, Christian BP, Feng B, Shelden RM. Early follicular serum Müllerian-inhibiting substance levels are associated with ovarian response during assisted reproductive technology cycles. Fertil Steril. 2002;77(3):468–71.
Broer SL, Dolleman M, Opmeer BC, Fauser BC, Mol BW, Broekmans FJ. AMH and AFC as predictors of excessive response in controlled ovarian hyperstimulation: a meta-analysis. Hum Reprod Update. 2011;17(1):46–54.
Broer SL, Dolleman M, van Disseldorp J, Broeze KA, Opmeer BC, Bossuyt PM, Eijkemans MJ, Mol BW, Broekmans FJ. Prediction of an excessive response in in vitro fertilization from patient characteristics and ovarian reserve tests and comparison in subgroups: an individual patient data meta-analysis. Fertil Steril. 2013;100(2):420–9 (e7).
Broer SL, Mol BW, Hendriks D, Broekmans FJ. The role of anti-Müllerian hormone in prediction of outcome after IVF: comparison with the antral follicle count. Fertil Steril. 2009;91(3):705–14.
Broer SL, van Disseldorp J, Broeze KA, Dolleman M, Opmeer BC, Bossuyt P, Eijkemans MJ, Mol BW, Broekmans FJ. Added value of ovarian reserve testing on patient characteristics in the prediction of ovarian response and ongoing pregnancy: an individual patient data approach. Hum Reprod Update. 2013;19(1):26–36.
Nelson SM, Yates RW, Lyall H, Jamieson M, Traynor I, Gaudoin M, Mitchell P, Ambrose P, Fleming R. Anti-Müllerian hormone-based approach to controlled ovarian stimulation for assisted conception. Hum Reprod. 2009;24(4):867–75.
Anckaert E, Smitz J, Schiettecatte J, Klein BM, Arce JC. The value of anti-Müllerian hormone measurement in the long GnRH agonist protocol: association with ovarian response and gonadotrophin-dose adjustments. Hum Reprod. 2012;27(6):1829–39.
La Marca A, Sunkara SK. Individualization of controlled ovarian stimulation in IVF using ovarian reserve markers: from theory to practice. Hum Reprod Update. 2014;20(1):124–40.
Lan VT, Linh NK, Tuong HM, Wong PC, Howles CM. Anti-Müllerian hormone versus antral follicle count for defining the starting dose of FSH. Reprod Biomed Online. 2013;27(4):390–9.
La Marca A, Papaleo E, Grisendi V, Argento C, Giulini S, Volpe A. Development of a nomogram based on markers of ovarian reserve for the individualisation of the follicle-stimulating hormone starting dose in in vitro fertilisation cycles. BJOG. 2012;119(10):1171–9.
Yates AP, Rustamov O, Roberts SA, Lim HY, Pemberton PW, Smith A, Nardo LG. Anti-Müllerian hormone-tailored stimulation protocols improve outcomes whilst reducing adverse effects and costs of IVF. Hum Reprod. 2011;26(9):2353–62.
La Marca A, Argento C, Sighinolfi G, Grisendi V, Carbone M, D’Ippolito G, Artenisio AC, Stabile G, Volpe A. Possibilities and limits of ovarian reserve testing in ART. Curr Pharm Biotechnol. 2012;13(3):398–408.
Eldar-Geva T, Ben-Chetrit A, Spitz IM, Rabinowitz R, Markowitz E, Mimoni T, Gal M, Zylber-Haran E, Margalioth EJ. Dynamic assays of inhibin B, anti-Müllerian hormone and estradiol following FSH stimulation and ovarian ultrasonography as predictors of IVF outcome. Hum Reprod. 2005;20(11):3178–83.
Nelson SM, Yates RW, Fleming R. Serum anti-Müllerian hormone and FSH: prediction of live birth and extremes of response in stimulated cycles–implications for individualization of therapy. Hum Reprod. 2007;22(9):2414–21.
Silberstein T, MacLaughlin DT, Shai I, Trimarchi JR, Lambert-Messerlian G, Seifer DB, Keefe DL, Blazar AS. Müllerian inhibiting substance levels at the time of HCG administration in IVF cycles predict both ovarian reserve and embryo morphology. Hum Reprod. 2006;21(1):159–63.
Fanchin R, Mendez Lozano DH, Frydman N, Gougeon A, Di Clemente N, Frydman R, Taieb J. Anti-Müllerian hormone concentrations in the follicular fluid of the preovulatory follicle are predictive of the implantation potential of the ensuing embryo obtained by in vitro fertilization. J Clin Endocrinol Metab. 2007;92(5):1796–802.
Ficicioglu C, Kutlu T, Baglam E, Bakacak Z. Early follicular anti-Müllerian hormone as an indicator of ovarian reserve. Fertil Steril. 2006;85(3):592–6.
Penarrubia J, Fabregues F, Manau D, Creus M, Carmona F, Casamitjana R, Vanrell JA, Balasch J. Previous cycle cancellation due to poor follicular development as a predictor of ovarian response in cycles stimulated with gonadotrophin-releasing hormone agonist-gonadotrophin treatment. Hum Reprod. 2005;20(3):622–8.
Smeenk JM, Sweep FC, Zielhuis GA, Kremer JA, Thomas CM, Braat DD. Anti-Müllerian hormone predicts ovarian responsiveness, but not embryo quality or pregnancy, after in vitro fertilization or intracytoplasmic sperm injection. Fertil Steril. 2007;87(1):223–6.
van Rooij IA, Broekmans FJ, Hunault CC, Scheffer GJ, Eijkemans MJ, de Jong FH, Themmen AP, te Velde ER. Use of ovarian reserve tests for the prediction of ongoing pregnancy in couples with unexplained or mild male infertility. Reprod Biomed Online. 2006;12(2):182–90.
Iliodromiti S, Kelsey TW, Wu O, Anderson RA, Nelson SM. The predictive accuracy of anti-Müllerian hormone for live birth after assisted conception: a systematic review and meta-analysis of the literature. Hum Reprod Update. 2014;20(4):560–70.
Tal R, Tal O, Seifer BJ, Seifer DB. Anti-Müllerian hormone as predictor of implantation and clinical pregnancy after assisted conception: a systematic review and meta-analysis. Fertil Steril. 2015;103(1):119–30 (e3).
Arce JC, La Marca A, Klein BM, Andersen AN, Fleming R. Anti-Müllerian hormone in gonadotropin releasing-hormone antagonist cycles: prediction of ovarian response and cumulative treatment outcome in good-prognosis patients. Fertil Steril. 2013;99(6):1644–53.
Brodin T, Hadziosmanovic N, Berglund L, Olovsson M, Holte J. Anti-Müllerian hormone levels are strongly associated with live-birth rates after assisted reproduction. J Clin Endocrinol Metab. 2013;98(3):1107–14.
La Marca A, Spada E, Grisendi V, Argento C, Papaleo E, Milani S, Volpe A. Normal serum anti-Müllerian hormone levels in the general female population and the relationship with reproductive history. Eur J Obstet Gynecol Reprod Biol. 2012;163(2):180–4.
Leader B, Hegde A, Baca Q, Stone K, Lannon B, Seifer DB, Broekmans F, Baker VL. High frequency of discordance between anti-Müllerian hormone and follicle-stimulating hormone levels in serum from estradiol-confirmed days 2 to 4 of the menstrual cycle from 5,354 women in U.S. fertility centers. Fertil Steril. 2012;98(4):1037–42.
Nelson SM, Iliodromiti S, Fleming R, Anderson R, McConnachie A, Messow CM. Reference range for the anti-Müllerian hormone Generation II assay: a population study of 10,984 women, with comparison to the established Diagnostics Systems Laboratory nomogram. Fertil Steril. 2014;101(2):523–9.
Nelson SM, Messow MC, McConnachie A, Wallace H, Kelsey T, Fleming R, Anderson RA, Leader B. External validation of nomogram for the decline in serum anti-Müllerian hormone in women: a population study of 15,834 infertility patients. Reprod Biomed Online. 2011;23(2):204–6.
Nelson SM, Messow MC, Wallace AM, Fleming R, McConnachie A. Nomogram for the decline in serum anti-Müllerian hormone: a population study of 9,601 infertility patients. Fertil Steril. 2011;95(2):736–41 (e1–3).
Steiner AZ, Herring AH, Kesner JS, Meadows JW, Stanczyk FZ, Hoberman S, Baird DD. Anti-Müllerian hormone as a predictor of natural fecundability in women aged 30–42 years. Obstet Gynecol. 2011;117(4):798–804.
Hagen CP, Vestergaard S, Juul A, Skakkebaek NE, Andersson AM, Main KM, Hjollund NH, Ernst E, Bonde JP, Anderson RA, et al. Low concentration of circulating anti-Müllerian hormone is not predictive of reduced fecundability in young healthy women: a prospective cohort study. Fertil Steril. 2012;98(6):1602–8 (e2).
Burger HG, Dudley EC, Hopper JL, Groome N, Guthrie JR, Green A, Dennerstein L. Prospectively measured levels of serum follicle-stimulating hormone, estradiol, and the dimeric inhibins during the menopausal transition in a population-based cohort of women. J Clin Endocrinol Metab. 1999;84(11):4025–30.
Dolleman M, Faddy MJ, van Disseldorp J, van der Schouw YT, Messow CM, Leader B, Peeters PH, McConnachie A, Nelson SM, Broekmans FJ. The relationship between anti-Müllerian hormone in women receiving fertility assessments and age at menopause in subfertile women: evidence from large population studies. J Clin Endocrinol Metab. 2013;98(5):1946–53.
Broer SL, Eijkemans MJ, Scheffer GJ, van Rooij IA, de Vet A, Themmen AP, Laven JS, de Jong FH, Te Velde ER, Fauser BC, et al. Anti-Müllerian hormone predicts menopause: a long-term follow-up study in normoovulatory women. J Clin Endocrinol Metab. 2011;96(8):2532–9.
Tehrani FR, Solaymani-Dodaran M, Azizi F. A single test of anti-Müllerian hormone in late reproductive-aged women is a good predictor of menopause. Menopause. 2009;16(4):797–802.
Dolleman M, Depmann M, Eijkemans MJ, Heimensem J, Broer SL, van der Stroom EM, Laven JS, Van Rooij IA, Scheffer GJ, Peeters PH, et al. Anti-Müllerian hormone is a more accurate predictor of individual time to menopause than mother’s age at menopause. Hum Reprod. 2014;29(3):584–91.
Ramezani Tehrani F, Dolleman M, van Disseldorp J, Broer SL, Azizi F, Solaymani-Dodaran M, Fauser BC, Laven JS, Eijkemans MJ, Broekmans F. Predicting menopausal age with anti-Müllerian hormone: a cross-validation study of two existing models. Climacteric. 2014;17(5):583–90.
Baker VL. Primary ovarian insufficiency in the adolescent. Curr Opin Obstet Gynecol. 2013;25(5):375–81.
Chao KC, Ho CH, Shyong WY, Huang CY, Tsai SC, Cheng HY, Chou LC, Lin CH, Li HY. Anti-Müllerian hormone serum level as a predictive marker of ovarian function in Taiwanese women. J Chin Med Assoc. 2012;75(2):70–4.
Kallio S, Aittomaki K, Piltonen T, Veijola R, Liakka A, Vaskivuo TE, Dunkel L, Tapanainen JS. Anti-Müllerian hormone as a predictor of follicular reserve in ovarian insufficiency: special emphasis on FSH-resistant ovaries. Hum Reprod. 2012;27(3):854–60.
Visser JA, Schipper I, Laven JS, Themmen AP. Anti-Müllerian hormone: an ovarian reserve marker in primary ovarian insufficiency. Nat Rev Endocrinol. 2012;8(6):331–41.
Knauff EA, Eijkemans MJ, Lambalk CB, ten Kate-Booij MJ, Hoek A, Beerendonk CC, Laven JS, Goverde AJ, Broekmans FJ, Themmen AP, et al. Anti-Müllerian hormone, inhibin B, and antral follicle count in young women with ovarian failure. J Clin Endocrinol Metab. 2009;94(3):786–92.
La Marca A, Marzotti S, Brozzetti A, Stabile G, Artenisio AC, Bini V, Giordano R, De Bellis A, Volpe A, Falorni A. Primary ovarian insufficiency due to steroidogenic cell autoimmunity is associated with a preserved pool of functioning follicles. J Clin Endocrinol Metab. 2009;94(10):3816–23.
Dewailly D, Lujan ME, Carmina E, Cedars MI, Laven J, Norman RJ, Escobar-Morreale HF. Definition and significance of polycystic ovarian morphology: a task force report from the Androgen Excess and Polycystic Ovary Syndrome Society. Hum Reprod Update. 2014;20(3):334–52.
Overbeek A, Lambalk CB. Phenotypic and pharmacogenetic aspects of ovulation induction in WHO II anovulatory women. Gynecol Endocrinol. 2009;25(4):222–34.
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004;81(1):19–25.
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod. 2004;19(1):41–7.
Dewailly D, Gronier H, Poncelet E, Robin G, Leroy M, Pigny P, Duhamel A, Catteau-Jonard S. Diagnosis of polycystic ovary syndrome (PCOS): revisiting the threshold values of follicle count on ultrasound and of the serum AMH level for the definition of polycystic ovaries. Hum Reprod. 2011;26(11):3123–9.
Eilertsen TB, Vanky E, Carlsen SM. Anti-Müllerian hormone in the diagnosis of polycystic ovary syndrome: can morphologic description be replaced? Hum Reprod. 2012;27(8):2494–502.
Li L, Chen X, Mo Y, Chen Y, Wenig M, Yang D. Elevated serum anti-Müllerian hormone in adolescent and young adult Chinese patients with polycystic ovary syndrome. Wien Klin Wochenschr. 2010;122(17–18):519–24.
Pigny P, Jonard S, Robert Y, Dewailly D. Serum anti-Müllerian hormone as a surrogate for antral follicle count for definition of the polycystic ovary syndrome. J Clin Endocrinol Metab. 2006;91(3):941–5.
Lin YH, Chiu WC, Wu CH, Tzeng CR, Hsu CS, Hsu MI. Anti-Müllerian hormone and polycystic ovary syndrome. Fertil Steril. 2011;96(1):230–5.
Hart R, Doherty DA, Norman RJ, Franks S, Dickinson JE, Hickey M, Sloboda DM. Serum anti-Müllerian hormone (AMH) levels are elevated in adolescent girls with polycystic ovaries and the polycystic ovarian syndrome (PCOS). Fertil Steril. 2010;94(3):1118–21.
Woo HY, Kim KH, Rhee EJ, Park H, Lee MK. Differences of the association of anti-Müllerian hormone with clinical or biochemical characteristics between women with and without polycystic ovary syndrome. Endocr J. 2012;59(9):781–90.
Homburg R, Ray A, Bhide P, Gudi A, Shah A, Timms P, Grayson K. The relationship of serum anti-Müllerian hormone with polycystic ovarian morphology and polycystic ovary syndrome: a prospective cohort study. Hum Reprod. 2013;28(4):1077–83.
Li HW, Anderson RA, Yeung WS, Ho PC, Ng EH. Evaluation of serum anti-Müllerian hormone and inhibin B concentrations in the differential diagnosis of secondary oligoamenorrhea. Fertil Steril. 2011;96(3):774–9.
Iliodromiti S, Kelsey TW, Anderson RA, Nelson SM. Can anti-Müllerian hormone predict the diagnosis of polycystic ovary syndrome? A systematic review and meta-analysis of extracted data. J Clin Endocrinol Metab. 2013;98(8):3332–40.
Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W, Janssen OE, Legro RS, Norman RJ, Taylor AE, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91(2):456–88.
Sahmay S, Aydin Y, Atakul N, Aydogan B, Kaleli S. Relation of anti-Müllerian hormone with the clinical signs of hyperandrogenism and polycystic ovary morphology. Gynecol Endocrinol. 2014;30(2):130–4.
Laven JS, Mulders AG, Visser JA, Themmen AP, De Jong FH, Fauser BC. Anti-Müllerian hormone serum concentrations in normoovulatory and anovulatory women of reproductive age. J Clin Endocrinol Metab. 2004;89(1):318–23.
Piouka A, Farmakiotis D, Katsikis I, Macut D, Gerou S, Panidis D. Anti-Müllerian hormone levels reflect severity of PCOS but are negatively influenced by obesity: relationship with increased luteinizing hormone levels. Am J Physiol Endocrinol Metab. 2009;296(2):E238–43.
Thomson RL, Buckley JD, Moran LJ, Noakes M, Clifton PM, Norman RJ, Brinkworth GD. The effect of weight loss on anti-Müllerian hormone levels in overweight and obese women with polycystic ovary syndrome and reproductive impairment. Hum Reprod. 2009;24(8):1976–81.
Farquhar C, Brown J, Marjoribanks J. Laparoscopic drilling by diathermy or laser for ovulation induction in anovulatory polycystic ovary syndrome. Cochrane Database Syst Rev. 2012;6:CD001122.
Amer SA, Li TC, Ledger WL. The value of measuring anti-Müllerian hormone in women with anovulatory polycystic ovary syndrome undergoing laparoscopic ovarian diathermy. Hum Reprod. 2009;24(11):2760–6.
Elmashad AI. Impact of laparoscopic ovarian drilling on anti-Müllerian hormone levels and ovarian stromal blood flow using three-dimensional power Doppler in women with anovulatory polycystic ovary syndrome. Fertil Steril. 2011;95(7):2342–6 (2346 e1).
Bath LE, Wallace WH, Shaw MP, Fitzpatrick C, Anderson RA. Depletion of ovarian reserve in young women after treatment for cancer in childhood: detection by anti-Müllerian hormone, inhibin B and ovarian ultrasound. Hum Reprod. 2003;18(11):2368–74.
Iwase A, Nakamura T, Nakahara T, Goto M, Kikkawa F. Anti-Müllerian hormone and assessment of ovarian reserve after ovarian toxic treatment: a systematic narrative review. Reprod Sci. 2015;22(5):519–26.
Anders C, Marcom PK, Peterson B, Gu L, Unruhe S, Welch R, Lyons P, Behera M, Copland S, Kimmick G, et al. A pilot study of predictive markers of chemotherapy-related amenorrhea among premenopausal women with early stage breast cancer. Cancer Invest. 2008;26(3):286–95.
Anderson RA, Themmen AP, Al-Qahtani A, Groome NP, Cameron DA. The effects of chemotherapy and long-term gonadotrophin suppression on the ovarian reserve in premenopausal women with breast cancer. Hum Reprod. 2006;21(10):2583–92.
Yu B, Douglas N, Ferin MJ, Nakhuda GS, Crew K, Lobo RA, Hershman DL. Changes in markers of ovarian reserve and endocrine function in young women with breast cancer undergoing adjuvant chemotherapy. Cancer. 2010;116(9):2099–105.
Decanter C, Morschhauser F, Pigny P, Lefebvre C, Gallo C, Dewailly D. Anti-Müllerian hormone follow-up in young women treated by chemotherapy for lymphoma: preliminary results. Reprod Biomed Online. 2010;20(2):280–5.
Bozza C, Puglisi F, Lambertini M, Osa EO, Manno M, Del Mastro L. Anti-Müllerian hormone: determination of ovarian reserve in early breast cancer patients. Endocr Relat Cancer. 2014;21(1):R51–65.
Gracia CR, Sammel MD, Freeman E, Prewitt M, Carlson C, Ray A, Vance A, Ginsberg JP. Impact of cancer therapies on ovarian reserve. Fertil Steril. 2012;97(1):134–40 (e1).
Rosendahl M, Andersen CY, la Cour Freiesleben N, Juul A, Lossl K, Andersen AN. Dynamics and mechanisms of chemotherapy-induced ovarian follicular depletion in women of fertile age. Fertil Steril. 2010;94(1):156–66.
Cancer Research UK [Internet]. A trial looking at ovarian protection for premenopausal women having chemotherapy for breast cancer (OPTION). http://www.cancerresearchuk.org/about-cancer/find-a-clinical-trial/a-trial-looking-at-ovarian-protection-for-premenopausal-women-having-chemotherapy-for-breast-cancer. Accessed 28 Sept 2009.
Anderson RA, Hindmarsh PC, Wallace WH. Induction of puberty by autograft of cryopreserved ovarian tissue in a patient previously treated for Ewing sarcoma. Eur J Cancer. 2013;49(13):2960–1.
Wallace WH, Smith AG, Kelsey TW, Edgar AE, Anderson RA. Fertility preservation for girls and young women with cancer: population-based validation of criteria for ovarian tissue cryopreservation. Lancet Oncol. 2014;15(10):1129–36.
Somigliana E, Vercellini P, Vigano P, Ragni G, Crosignani PG. Should endometriomas be treated before IVF-ICSI cycles? Hum Reprod Update. 2006;12(1):57–64.
Chang HJ, Han SH, Lee JR, Jee BC, Lee BI, Suh CS, Kim SH. Impact of laparoscopic cystectomy on ovarian reserve: serial changes of serum anti-Müllerian hormone levels. Fertil Steril. 2010;94(1):343–9.
Iwase A, Hirokawa W, Goto M, Takikawa S, Nagatomo Y, Nakahara T, Manabe S, Kikkawa F. Serum anti-Müllerian hormone level is a useful marker for evaluating the impact of laparoscopic cystectomy on ovarian reserve. Fertil Steril. 2010;94(7):2846–9.
Ercan CM, Duru NK, Karasahin KE, Coksuer H, Dede M, Baser I. Ultrasonographic evaluation and anti-Müllerian hormone levels after laparoscopic stripping of unilateral endometriomas. Eur J Obstet Gynecol Reprod Biol. 2011;158(2):280–4.
Kitajima M, Khan KN, Hiraki K, Inoue T, Fujishita A, Masuzaki H. Changes in serum anti-Müllerian hormone levels may predict damage to residual normal ovarian tissue after laparoscopic surgery for women with ovarian endometrioma. Fertil Steril. 2011;95(8):2589–91 (e1).
Lee DY, Young Kim N, Jae Kim M, Yoon BK, Choi D. Effects of laparoscopic surgery on serum anti-Müllerian hormone levels in reproductive-aged women with endometrioma. Gynecol Endocrinol. 2011;27(10):733–6.
Tsolakidis D, Pados G, Vavilis D, Athanatos D, Tsalikis T, Giannakou A, Tarlatzis BC. The impact on ovarian reserve after laparoscopic ovarian cystectomy versus three-stage management in patients with endometriomas: a prospective randomized study. Fertil Steril. 2010;94(1):71–7.
Hirokawa W, Iwase A, Goto M, Takikawa S, Nagatomo Y, Nakahara T, Bayasula B, Nakamura T, Manabe S, Kikkawa F. The post-operative decline in serum anti-Müllerian hormone correlates with the bilaterality and severity of endometriosis. Hum Reprod. 2011;26(4):904–10.
Kwon SK, Kim SH, Yun SC, Kim DY, Chae HD, Kim CH, Kang BM. Decline of serum anti-Müllerian hormone levels after laparoscopic ovarian cystectomy in endometrioma and other benign cysts: a prospective cohort study. Fertil Steril. 2014;101(2):435–41.
Raffi F, Metwally M, Amer S. The impact of excision of ovarian endometrioma on ovarian reserve: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2012;97(9):3146–54.
Somigliana E, Berlanda N, Benaglia L, Vigano P, Vercellini P, Fedele L. Surgical excision of endometriomas and ovarian reserve: a systematic review on serum anti-Müllerian hormone level modifications. Fertil Steril. 2012;98(6):1531–8.
Sugita A, Iwase A, Goto M, Nakahara T, Nakamura T, Kondo M, Osuka S, Mori M, Saito A, Kikkawa F. One-year follow-up of serum anti-Müllerian hormone levels in patients with cystectomy: are different sequential changes due to different mechanisms causing damage to the ovarian reserve? Fertil Steril. 2013;100(2):516–22 (e3).
Sonmezer M, Taskin S, Gemici A, Kahraman K, Ozmen B, Berker B, Atabekoglu C. Can ovarian damage be reduced using hemostatic matrix during laparoscopic endometrioma surgery? A prospective, randomized study. Arch Gynecol Obstet. 2013;287(6):1251–7.
Zaitoun MM, El Behery MM. Comparing long-term impact on ovarian reserve between laparoscopic ovarian cystectomy and open laprotomy for ovarian endometrioma. J Ovarian Res. 2013;6(1):76.
Hart RJ, Hickey M, Maouris P, Buckett W. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database Syst Rev. 2008;2:CD004992.
Jang WK, Lim SY, Park JC, Lee KR, Lee A, Rhee JH. Surgical impact on serum anti-Müllerian hormone in women with benign ovarian cyst: A prospective study. Obstet Gynecol Sci. 2014;57(2):121–7.
Mohamed ML, Nouh AA, El-Behery MM, Mansour SA. Effect on ovarian reserve of laparoscopic bipolar electrocoagulation versus laparotomic hemostatic sutures during unilateral ovarian cystectomy. Int J Gynaecol Obstet. 2011;114(1):69–72.
Ni L, Sadiq S, Mao Y, Cui Y, Wang W, Liu J. Influence of various tubal surgeries to serum anti-Müllerian hormone level and outcome of the subsequent IVF-ET treatment. Gynecol Endocrinol. 2013;29(4):345–9.
Ercan CM, Sakinci M, Coksuer H, Keskin U, Tapan S, Ergun A. Ovarian reserve testing before and after laparoscopic tubal bipolar electrodesiccation and transection. Eur J Obstet Gynecol Reprod Biol. 2013;166(1):56–60.
Hehenkamp WJ, Volkers NA, Broekmans FJ, de Jong FH, Themmen AP, Birnie E, Reekers JA, Ankum WM. Loss of ovarian reserve after uterine artery embolization: a randomized comparison with hysterectomy. Hum Reprod. 2007;22(7):1996–2005.
Arthur R, Kachura J, Liu G, Chan C, Shapiro H. Laparoscopic myomectomy versus uterine artery embolization: long-term impact on markers of ovarian reserve. J Obstet Gynaecol Can. 2014;36(3):240–7.
Chong YH, Campbell AJ, Farrand S, McLennan IS. Anti-Müllerian hormone level in older women: detection of granulosa cell tumor recurrence. Int J Gynecol Cancer. 2012;22(9):1497–9.
Lane AH, Lee MM, Fuller AF Jr, Kehas DJ, Donahoe PK, MacLaughlin DT. Diagnostic utility of Müllerian inhibiting substance determination in patients with primary and recurrent granulosa cell tumors. Gynecol Oncol. 1999;73(1):51–5.
Long WQ, Ranchin V, Pautier P, Belville C, Denizot P, Cailla H, Lhomme C, Picard JY, Bidart JM, Rey R. Detection of minimal levels of serum anti-Müllerian hormone during follow-up of patients with ovarian granulosa cell tumor by means of a highly sensitive enzyme-linked immunosorbent assay. J Clin Endocrinol Metab. 2000;85(2):540–4.
La Marca A, Volpe A. The Anti-Müllerian hormone and ovarian cancer. Hum Reprod Update. 2007;13(3):265–73.
Hudson PL, Dougas I, Donahoe PK, Cate RL, Epstein J, Pepinsky RB, MacLaughlin DT. An immunoassay to detect human Müllerian inhibiting substance in males and females during normal development. J Clin Endocrinol Metab. 1990;70(1):16–22.
Nelson SM, La Marca A. The journey from the old to the new AMH assay: how to avoid getting lost in the values. Reprod Biomed Online. 2011;23(4):411–20.
Rustamov O, Smith A, Roberts SA, Yates AP, Fitzgerald C, Krishnan M, Nardo LG, Pemberton PW. The measurement of anti-Müllerian hormone: a critical appraisal. J Clin Endocrinol Metab. 2014;99(3):723–32.
Li HW, Ng EH, Wong BP, Anderson RA, Ho PC, Yeung WS. Correlation between three assay systems for anti-Müllerian hormone (AMH) determination. J Assist Reprod Genet. 2012;29(12):1443–6.
Reichman DE, Goldschlag D, Rosenwaks Z. Value of anti-Müllerian hormone as a prognostic indicator of in vitro fertilization outcome. Fertil Steril. 2014;101(4):1012–8 (e1011).
Kedem A, Haas J, Geva LL, Yerushalmi G, Gilboa Y, Kanety H, Hanochi M, Maman E, Hourvitz A. Ongoing pregnancy rates in women with low and extremely low AMH levels. A multivariate analysis of 769 cycles. PLoS One. 2013;8(12):e81629.
Su HI, Sammel MD, Homer MV, Bui K, Haunschild C, Stanczyk FZ. Comparability of anti-Müllerian hormone levels among commercially available immunoassays. Fertil Steril. 2014;101(6):1766–72 (e1761).
Welsh P, Smith K, Nelson SM. A single-centre evaluation of two new anti-Müllerian hormone assays and comparison with the current clinical standard assay. Hum Reprod. 2014;29(5):1035–41.
Chai J, Howie AF, Cameron DA, Anderson RA. A highly-sensitive anti-Müllerian hormone assay improves analysis of ovarian function following chemotherapy for early breast cancer. Eur J Cancer. 2014;50(14):2367–74.
Decanter C, Peigne M, Mailliez A, Morschhauser F, Dassonneville A, Dewailly D, Pigny P. Toward a better follow-up of ovarian recovery in young women after chemotherapy with a hypersensitive anti-Müllerian hormone assay. Fertil Steril. 2014;102(2):483–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Akira Iwase, Tomoko Nakamura, Satoko Osuka, Sachiko Takikawa, Maki Goto, and Fumitaka Kikkawa declare that they have no conflicts of interest.
Human/animal studies
This article does not contain any studies with human or animal subjects performed by any of the authors.
About this article
Cite this article
Iwase, A., Nakamura, T., Osuka, S. et al. Anti-Müllerian hormone as a marker of ovarian reserve: What have we learned, and what should we know?. Reprod Med Biol 15, 127–136 (2016). https://doi.org/10.1007/s12522-015-0227-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12522-015-0227-3