Abstract
Purpose
Maternal diabetes leads to increased blood glucose concentration in the mother and consequently in the foetus, causing various neonatal problems. This study was conducted to evaluate the effects of maternal diabetes on foetal ovarian structure.
Methods
Sixteen adult female rats were allocated into two equal groups. Diabetes was induced in one group by alloxan. Both groups became pregnant by natural mating. Thirty days after birth, the female offspring were terminated, the body weight and blood glucose of the animals measured and their ovaries removed. Various histological and cellular parameters were determined using histological and electron microscopy techniques.
Results
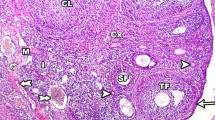
Results revealed a significant increase in body weight and blood glucose in the offspring of the diabetic mothers (ODM) compared to that of the controls. The weight, volume and diameter of the ovary and the ovarian capsule thickness were inclined to decrease in ODM compared to that of controls. The number and diameter of primary, pre-antral and antral follicles were decreased in ovaries in the ODM. The electro-micrographs have demonstrated the organelle alterations in oocytes and granulosa cells that suggest the apoptosis progress and oxidative stress.
Conclusions
Maternal hyperglycaemia exhibited deleterious effects on the female reproductive system in the offspring.



Similar content being viewed by others
References
Jones CW. Gestational diabetes and its impact on the neonate. Neonatal Netw. 2001;20:17–23.
Persson B, Hanson U. Neonatal morbidities in gestational diabetes mellitus. Diabetes Care. 1998;2:79–84.
Van Assche FA, Aerts L, De Prins FA. Degranulation of the insulin-producing B cells in an infant of a diabetic mother. Br J Obstet Gynecol. 1983;90:182–5.
Ballester J, Muñoz MC, Domínguez J, Palomo MJ, Rivera M, Rigau T, Guinovart JJ, Rodríguez-Gil JE. Tungstate administration improves the sexual and reproductive function in female rats with streptozotocin-induced diabetes. Hum Reprod. 2007;22:2128–235.
Ahmed RG. Evolutionary interactions between diabetes and development. Diabetes Res Clin Pract. 2011;92:153–67.
Bestetti G, Locatelli V, Tirone F, Rossi GL, Müller EE. One month of streptozotocin-diabetes induces different neuroendocrine and morphological alterations in the hypothalamo-pituitary axis of male and female rats. Endocrinology. 1985;117:208–16.
Blades RA, Bryant KR, Whitehead SA. Feedback effects of steroids and gonadotrophin control in adult rats with streptozotocin-induced diabetes mellitus. Diabetologia. 1985;28:348–54.
Moley KH, Chi MM, Knudson CM, Korsmeyer SJ, Mueckler MM. Hyperglycemia induces apoptosis in pre-implantation embryos through cell death effector pathways. Nat Med. 1998;4:1421–4.
Goswami D, Conway GS. Premature ovarian failure. Hum Reprod Update. 2005;11:391–410.
Garris DR, Garris BL. Diabetes (db/db) mutation-induced ovarian involution: progressive hypercytolipidemia. Exp Biol Med (Maywood). 2003;228:1040–50.
Garris DR, Garris BL. Diabetes (db/db) mutation-induced female reproductive tract hypercytolipidemia: estrogenic restoration of utero-ovarian indices. Reprod Toxicol. 2004;18:641–51.
Lin S, Lin K, Li W, Zhou X, Huang T. Maternal diabetes increases apoptosis in mice oocytes, not 2-cell embryos. Endocrine. 2010;37:460–6.
Garris DR, Williams SK, West L. Morphometric evaluation of diabetes-associated ovarian atrophy in the C57BL/KsJ mouse: relationship to age and ovarian function. Anat Rec. 1985;211:434–43.
Jelodar G, Khaksar Z, Pourahmadi M. Endocrine profile and testicular histomorphometry in adult rat offspring of diabetic mothers. J Physiol Sci. 2009;59:377–82.
SzkuDelski T. The mechanism of Alloxan and Streptozotocin action in B Cells of the rat pancreas. Physiol Res. 2001; 50: 536–46.
Damas J, Hallet C, Lefebvre PJ. Changes in blood glucose and plasma insulin levels induced by bradykinin in anaesthetized rats. Br J Pharmacol. 2001;134:1312–8.
Turner CD, Bagnara JT. General endocrinology. Philadelphia: WB Saunders Company; 1971.
Elias H, Hyde DM. An elementary introduction to stereology (quantitative microscopy). Am J Anat. 1980;159:411–7.
Luna LG. Manual of histologic staining methods of the Armed forces institute of pathology. USA: McGraw Hill; 1968.
Wrotniak BH, Shults J, Butts S, Stettler N. Gestational weight gain and risk of overweight in the offspring at age 7 y in a multicenter, multiethnic cohort study. Am J Clin Nutr. 2008;87:1818–24.
Khaksar Z, Jelodar G, Hematian H. Cerebrum malformation in offspring of diabetic mothers. Comp Clin Pathol. 2011;10:1160–9.
Chang SA, Dale NA, Oley HK. Maternal diabetes adversely affects pre-ovulatory oocyte maturation, development, and granulosa cell apoptosis. Endocrinology. 2005;146:2445–53.
Wang Q, Frolova AI, Purcell S, Adastra K, Schoeller E, Chi MM, Schedl T, Moley KH. Mitochondrial dysfunction and apoptosis in cumulus cells of type I diabetic mice. PLoS ONE. 2010;5:e15901.
Sugiura K, Eppig JJ. Control of metabolic cooperativity between oocytes and their companion granulosa cells by mouse oocytes. Reprod Fertil Dev. 2005;17:667–74.
Ishikawa S, Bensaoula T, Uga S, Mukuno K. Electron-microscopic study of iris nerves and muscles in diabetes. Ophthalmologica. 1985;191:172–83.
Schramm E, Wagner M, Nellessen U, Inselmann G. Ultrastructural changes of human cardiac atrial nerve endings in diabetes mellitus. Eur J Clin Invest. 2000;30:311–6.
Acknowledgments
The authors would like to express their thanks to Mr Mogheisi, Mrs Ahmadi and Mrs Shams for their technical assistance.
Conflict of interest
We have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.


About this article
Cite this article
Khaksar, Z., Jelodar, G., Hematian, H. et al. Alterations of the ovarian histomorphometry at pre-puberty in rat offspring from diabetic mothers. Reprod Med Biol 12, 173–178 (2013). https://doi.org/10.1007/s12522-013-0151-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12522-013-0151-3