Abstract
Background
Previous studies investigated the association between gestational anemia and neonatal outcomes. However, few studies explored whether the effects of gestational anemia could be eliminated by subsequent correction of anemia in the later stages of pregnancy. This study aimed to investigate the relationship between anemia in different trimesters and neonatal outcomes.
Methods
The study was conducted in Shanghai, China, with a sample of 46,578 pregnant women who delivered between January 1, 2016 and July 1, 2019. A multivariable logistic regression model was adopted to analyse the associations between maternal anemia and neonatal outcomes.
Results
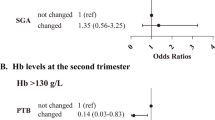
The incidence of gestational anemia was 30.2%, including 4.4% in the first trimester, 9.6% in the second trimester, and 16.2% in the third trimester. Only 24.5% (507/2066) of anemia that occurred in the first trimester and 29.6% (1320/4457) that occurred in the second trimester could be corrected in the later stages of pregnancy. Anemia occurring in the first trimester was associated with small for gestational age [odds ratio (OR) 1.46; 95% confidence interval (CI) 1.20–1.78] and with fetal distress (OR 1.23; 95% CI 1.08–1.40). Anemia corrected in the first trimester also was associated with a higher risk of small for gestational age.
Conclusions
Gestational anemia is a public health problem in China impacting neonatal health. Anemia in pregnancy could be corrected in only about a quarter of the women. Anemia in the first trimester, whether corrected or not, still led to lower birth weight; therefore, the prevention of anemia prior to pregnancy is important.

Similar content being viewed by others
References
Churchill D, Nair M, Stanworth SJ, Knight M. The change in haemoglobin concentration between the first and third trimesters of pregnancy: a population study. BMC Pregnancy Childbirth. 2019;19:359–64.
Fisher AL, Nemeth E. Iron homeostasis during pregnancy. Am J Clin Nutr. 2017;106:1567–74.
Milman N, Bergholt T, Byg KE, Eriksen L, Graudal N. Iron status and iron balance during pregnancy. A critical reappraisal of iron supplementation. Acta Obstet Gynecol Scand. 1999;78:749–57.
Marangoni F, Cetin I, Verduci E, Canzone G, Giovannini M, Scollo P, et al. Maternal diet and nutrient requirements in pregnancy and breastfeeding. An Italian consensus document. Nutrients. 2016;8:629–45.
Stevens GA, Finucane MM, De-Regil LM, Paciorek CJ, Flaxman SR, Branca F, et al. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995–2011: a systematic analysis of population-representative data. Lancet Glob Health. 2013;1:e16-25.
Lin L, Wei Y, Zhu W, Wang C, Su R, Feng H, et al. Prevalence, risk factors and associated adverse pregnancy outcomes of anaemia in Chinese pregnant women: a multicentre retrospective study. BMC Pregnancy Childbirth. 2018;18:111–8.
Tan J, He G, Qi Y, Yang H, Xiong Y, Liu C, et al. Prevalence of anemia and iron deficiency anemia in Chinese pregnant women (IRON WOMEN): a national cross-sectional survey. BMC Pregnancy Childbirth. 2020;20:670–81.
Yin A, Li B, Luo M, Xu L, Wu L, Zhang L, et al. The prevalence and molecular spectrum of alpha- and beta-globin gene mutations in 14,332 families of Guangdong Province, China. PLoS ONE. 2014;9:e89855.
Sun CC, Chou HH, Chuang LL. Trends and risk factors of stillbirth in Taiwan 2006–2013: a population-based study. Arch Gynecol Obstet. 2019;299:961–7.
Zhang Q, Ananth CV, Li Z, Smulian JC. Maternal anaemia and preterm birth: a prospective cohort study. Int J Epidemiol. 2009;38:1380–9.
Figueiredo ACMG, Gomes-Filho IS, Silva RB, Pereira PPS, Mata FAFD, Lyrio AO, et al. Maternal anemia and low birth weight: a systematic review and meta-analysis. Nutrients. 2018;10:601–17.
Lelic M, Bogdanovic G, Ramic S, Brkicevic E. Influence of maternal during pregnancy on placenta and newborns. Med Arch. 2014;68:184–7.
Stangret A, Skoda M, Wnuk A, Pyzlak M, Szukiewicz D. Mild anemia during pregnancy upregulates placental vascularity development. Med Hypotheses. 2017;102:37–40.
Larsen S, Bjelland EK, Haavaldsen C, Eskild A. Placental weight in pregnancies with high or low hemoglobin concentrations. Eur J Obstet Gynecol Reprod Biol. 2016;206:48–52.
Embryogenesis and fetal morphological development. In: Cunningham FG, Leveno KJ, Bloom SL, Spong CY, Dashe JS, Hoffman BL, et al., editors. Williams obstetrics. 24th ed. McGraw-Hill; 2014. p. 127–53.
Liu X, Wang C, Liu W, Li J, Li C, Kou X, et al. Distinct features of H3K4me3 and H3K27me3 chromatin domains in pre-implantation embryos. Nature. 2016;537:558–62.
Wang C, Liu X, Gao Y, Yang L, Li C, Liu W, et al. Reprogramming of H3K9me3-dependent heterochromatin during mammalian embryo development. Nat Cell Biol. 2018;20:620–31.
Li L, Guo F, Gao Y, Ren Y, Yuan P, Yan L, et al. Single-cell multi-omics sequencing of human early embryos. Nat Cell Biol. 2018;20:847–58.
Tian S, Lin XH, Xiong YM, Liu ME, Yu TT, Lv M, et al. Prevalence of prediabetes risk in offspring born to mothers with hyperandrogenism. Ebiomedicine. 2017;16:275–83.
Rooij SR, Painter RC, Roseboom TJ, Phillips DI, Osmond C, Barker DJ, et al. Glucose tolerance at age 58 and the decline of glucose tolerance in comparison with age 50 in people prenatally exposed to the Dutch famine. Diabetologia. 2006;49:637–43.
Li J, Liu S, Li S, Feng R, Na L, Chu X, et al. Prenatal exposure to famine and the development of hyperglycemia and type 2 diabetes in adulthood across consecutive generations: a population-based cohort study of families in Suihua, China. Am J Clin Nutr. 2017;105:221–7.
Barker DJ, Osmond C. Infant mortality, childhood nutrition, and ischaemic heart disease in England and Wales. Lancet. 1986;1:1077–81.
Fowkes FJI, Moore KA, Opi DH, Simpson JA, Langham F, Stanisic DI, et al. Iron deficiency during pregnancy is associated with a reduced risk of adverse birth outcomes in a malaria-endemic area in a longitudinal cohort study. BMC Med. 2018;16:156–65.
Symington EA, Baumgartner J, Malan L, Wise AJ, Ricci C, Zandberg L, et al. Maternal iron-deficiency is associated with premature birth and higher birth weight despite routine antenatal iron supplementation in an urban South African setting: the NuPED prospective study. PLoS ONE. 2019;14:e221299.
Smith C, Teng F, Branch E, Chu S, Joseph KS. Maternal and perinatal morbidity and mortality associated with anemia in pregnancy. Obstet Gynecol. 2019;134:1234–44.
Oaks BM, Jorgensen JM, Baldiviez LM, Adu-Afarwuah S, Maleta K, Okronipa H, et al. Prenatal iron deficiency and replete iron status are associated with adverse birth outcomes, but associations differ in Ghana and Malawi. J Nutr. 2019;149:513–21.
Haider BA, Olofin I, Wang M, Spiegelman D, Ezzati M, Fawzi WW. Anaemia, prenatal iron use, and risk of adverse pregnancy outcomes: systematic review and meta-analysis. BMJ. 2013;346:f3443.
Hamalainen H, Hakkarainen K, Heinonen S. Anaemia in the first but not in the second or third trimester is a risk factor for low birth weight. Clin Nutr. 2003;22:271–5.
WHO. WHO recommendations on antenatal care for a positive pregnancy experience. Geneva: World Health Organization; 2016.
Zhu L, Zhang R, Zhang S, Shi W, Yan W, Wang X, et al. Chinese neonatal birth weight curve for different gestational age. Zhonghua Er Ke Za Zhi. 2015;53:97–103.
Kumar KJ, Asha N, Murthy DS, Sujatha M, Manjunath V. Maternal anemia in various trimesters and its effect on newborn weight and maturity: an observational study. Int J Prev Med. 2013;4:193–9.
Gabriela B, Christian B. Mild anemia and pregnancy outcome in a Swiss collective. J Pregnancy. 2014;2014:307535.
Byg KE, Milman N, Hansen S, Agger AO. Serum ferritin is a reliable, non-invasive test for iron status in pregnancy: comparison of ferritin with other iron status markers in a longitudinal study on healthy pregnant women. Hematology. 2000;5:319–25.
Means RT. Iron deficiency and iron deficiency anemia: implications and impact in pregnancy, fetal development, and early childhood parameters. Nutrients. 2020;12:447–61.
Pavord S, Myers B, Robinson S, Allard S, Strong J, Oppenheimer C. UK guidelines on the management of iron deficiency in pregnancy. Br J Haematol. 2012;156:588–600.
WHO. Serum and red blood cell folate concentrations for assessing folate status in populations. Geneva: World Health Organization; 2012.
WHO. Optimal serum and red blood cell folate concentrations in women of reproductive age for prevention of neural tube defects. Geneva: World Health Organization, 2015.
Julia LF, Anura VK, Beena B, Tinku T, Krishnamachari S, Christopher D. Anaemia and iron deficiency in pregnancy and adverse perinatal outcomes in Southern India. Eur J Clin Nutr. 2018;74:112–25.
Barker DJ. Fetal origins of coronary heart disease. BMJ. 1995;311:171–4.
Katz J, Lee AC, Kozuki N, Lawn JE, Cousens S, Blencowe H, et al. Mortality risk in preterm and small-for-gestational-age infants in low-income and middle-income countries: a pooled country analysis. Lancet. 2013;382:417–25.
Ludvigsson JF, Lu D, Hammarstrom L, Cnattingius S, Fang F. Small for gestational age and risk of childhood mortality: a Swedish population study. PLoS Med. 2018;15:e1002717.
Acknowledgements
The authors would like to thank Lei Chen and Ping Wang in International Peace Maternity and Child Health Hospital for their efforts in data collection.
Funding
This work was supported by the Special Fund for the National Key Research and Development Plan Grant (2017YFC1001300), the International Cooperation Project of China and Canada NSFC (81661128010), the Major Program of National Natural Science Foundation of China (81490742), the Natural Science Foundation of China (31571556), Innovative research team of high-level local universities in Shanghai, and Shanghai Municipal Key Clinical Speciality, Shanghai, China.
Author information
Authors and Affiliations
Contributions
HFH, GLD, CFS, XML, and ZYZ participated in the conceptualization of the study. HHF had full access to all data in the study and take responsibility for the integrity of the data and accuracy of the data analysis. CFS wrote the original draft. CFS and HL conducted the formal analysis. GLD, HFH, and JZS reviewed and edited the manuscript. YHH, KXZ, and HTH provided supports on methodology. All authors commented on the drafts and approved the final draft.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Ethics Committee of the International Peace Maternity and Child Health Hospital (GKLW) 2017–81.
Conflict of interest
No financial or nonfinancial benefits have been received or will be received from any party related directly or indirectly to the subject of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, CF., Liu, H., Hao, YH. et al. Association between gestational anemia in different trimesters and neonatal outcomes: a retrospective longitudinal cohort study. World J Pediatr 17, 197–204 (2021). https://doi.org/10.1007/s12519-021-00411-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-021-00411-6