Abstract
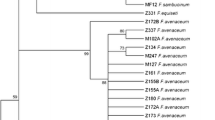
The increasing incidence of pokkah boeng has been observed throughout the sugarcane-producing areas in the Philippines. Sugarcane plants exhibiting characteristic symptoms of the disease were collected from different areas in the three major geographical regions: Luzon, Visayas and Mindanao. Symptoms surveyed in Visayas and Mindanao fields were more severe and diverse than those observed in Luzon. A total of 306 Fusarium spp. were isolated from these diseased plant samples on potato dextrose medium and water agar medium, and molecular identification using the genus-specific primer pair (ITS-Fu-f/r) and translation elongation factor (TEF)-1α gene sequencing were performed. Chlorosis after 30 days of inoculation and leaf necrosis after 45 days of inoculation in sugarcane seedlings (VMC 86-550) was observed, wherein 37% (112 out of 306) isolates were proven pathogenic. Traditional taxonomic characterization of the 112 pathogenic isolates identified them as F. sacchari, F. proliferatum, F. verticillioides, F.subglutinans, F. graminareum and F. incarnatum. This was further supported by molecular identification of the representative 56 isolates based on TEF-1α sequences as F. sacchari (66%), F. proliferatum (16%), F. incarnatum (7%), F. verticillioides (5%) and F. subglutinans (5%). Furthermore, these fungal pathogens were established as closely related species regardless of geographical origin based on the established clustering pattern in the constructed phylogenetic tree. These findings are important information on the etiology of pokkah boeng in the Philippines, and in formulating studies for control measures of the pathogen to prevent the disease from becoming an epidemic which may cause extensive loss to the sugarcane industry in the future. This is the first report on characterizing the pathogenic Fusarium spp. on sugarcane pokkah boeng in the Philippines.





Similar content being viewed by others
References
Abd-Elsalam, K.A., I.N. Aly, M.A. Abdel-Satar, M.S. Khalil, and J.A. Verreet. 2003. PCR identification of Fusarium genus based on nuclear ribosomal-DNA sequence data. African Journal of Biotechnology 2 (4): 82–85.
Abramoff, M.D., P.J. Magalhes, and S.J. Ram. 2004. Image processing with ImageJ. Biophotonics International 11 (7): 36–42.
Cullings, K.W. 1992. Design and testing of a plant-specific PCR primer for ecological and evolutionary studies. Molecular Ecology 1: 233–240.
Cumagun, C.J.R. 2007. Population genetic analysis of plant pathogenic fungi with emphasis on Fusarium species. The Philippine Agricultural Scientist 90 (3): 244–256.
Doyle, J.J., and J.L. Doyle. 1987. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemistry Bulletin 19: 11–15.
Fourie, G., E.T. Steenkamp, T.R. Gordon, and A. Viljoen. 2009. Evolutionary relationships among Fusarium oxysporum f. sp. cubense vegetative compatibility groups. Applied and Environmental Microbiology 75 (14): 4770–4781.
Hilton, A., H. Zhang, W. Yu, and W.B. Shim. 2017. Identification and characterization of pathogenic and endophytic fungal species associated with Pokkah Boeng Disease of Sugarcane. Plant Pathology Journal 33 (3): 238–248.
Hsuan, H.M., L. Zakaria, and B. Salleh. 2010. Characterization of Fusarium isolates from rice, sugarcane and maize using RFLP-IGS. Journal of Plant Protection Research 50 (4): 409–411.
Israel, D.C. 2012. Typhoons, floods and droughts: Regional occurrence and value of damages to rice farming in the Philippines. Philippine Institute for Developmental Studies Policy Notes (2012–15):1–6.
Kaur, A., V.K. Sharma, A. Sirari, J. Kaur, G. Singh, and P. Kumar. 2015. Variability in Fusarium oxysporum f. sp. ciceris causing wilt in chickpea. African Journal of Microbiology Research 9 (15): 1089–1097.
Kearse, M., R. Moir, A. Wilson, S. Stones-Havas, M. Cheung, S. Sturrock, S. Buxton, A. Cooper, S. Markowitz, C. Duran, T. Thierer, B. Ashton, P. Mentjies, and A. Drummond. 2012. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28 (12): 1647–1649.
Khani, K.T., A. Alizadeh, R.F. Nejad, and A.S. Tehrani. 2013. Pathogenicity of Fusarium proliferatum, a new causal agent of pokkah boeng in sugarcane. Proceedings of International Society of Sugar Cane 1–5.
Leslie, J.F., and B.A. Summerell. 2006. The Fusarium laboratory manual. Ames, IA: Blackwell Publishing.
Lin, Z., Y. Zhang, Y. Que, R. Chen, B. Chen, and M. Zhang. 2015. Characterization of Fusarium verticilloides isolates from pokkah boeng on sugarcane and the disease incidence in field. Journal of Microbiology & Experimentation 2 (5): 1–7.
Lin, Z., S. Xu, Y. Que, J. Wang, J.C. Comstock, J. Wei, P.H. McCord, B. Chen, R. Chen, and M. Zhang. 2014. Species-specific detection and identification of Fusarium species complex, the causal agent of sugarcane pokkah boeng in China. PLoS ONE 9 (8): 1–12.
Martin, J.P., H. Handojo, and C.A. Wismer. 1989. Pokkah boeng. In: Diseases of sugarcane: Major diseases, pp. 157–165. Netherlands: Elsevier Science Publishers.
Mohammadi, A., R.F. Nejad, and N.N. Mofrad. 2012. Fusarium verticilloides from sugarcane, vegetative compatibility groups and pathogenicity. Plant Protection Science 48 (2): 80–84.
Padilla-Fernandez, M.D., and P.L. Nuthall. 2009. Technical efficiency in the production of sugarcane in Central Negros area, Philippines: An application of data envelopment analysis. Journal of ISSAAS 15 (1): 77–90.
Pascual, C.B., A.K. Barcos, J.L. Mandap, and E.M. Ocampo. 2016. Fumonisin-producing Fusarium species causing ear rot of corn in the Philippines. Philippine Journal of Crop Science 41 (1): 12–21.
Singh, A., S.S. Chauhan, A. Singh, and S.B. Singh. 2006. Deterioration in sugarcane due to pokkah boeng disease. Sugar Tech 8 (2&3): 187–190.
Siti Nordahliawate, M.S., M.Z. Nur Ain Izzati, A.R. Azmi, and B. Salleh. 2008. Distribution, morphological characterization and pathogenicity of Fusarium sacchari associated with pokkah boeng disease of sugarcane in Peninsular Malaysia. Pertanika Journal of Tropical Agricultural Science 31 (2): 279–286.
Vishwakarma, S.K., P. Kumar, A. Nigam, A. Singh, and A. Kumar. 2013. Pokkah boeng: An emerging disease of sugarcane. Journal of Plant Pathology & Microbiology 4 (3): 1–5.
Vishwakarma, S.K., A. Nigam, and A. Singh. 2016. Molecular phylogenetic analysis of Fusarium isolates causing pokkah boeng disease in sugarcane based on RAPD marker. International Journal of Agricultural Science and Research 6 (3): 177–186.
Viswanathan, R., and G.P. Rao. 2011. Disease scenario and management of major sugarcane diseases in India. Sugar Tech 13 (4): 336–353.
Viswanathan, R., P. Malathi, A. Annadurai, C. Naveen Prasanth, and M. Scindiya. 2014. Sudden occurrence of wilt and pokkah boeng in sugarcane and status of resistance in the parental clones in national hybridization garden to these diseases. Journal of Sugarcane Research 4 (1): 62–81.
Viswanathan, R., C.G. Balaji, R. Selvakumar, P. Malathi, A. Ramesh Sundar, C. Naveen Prasanth, M.L. Chhabra, and B. Parameswari. 2017. Epidemiology of Fusarium diseases in sugarcane: A new discovery of same Fusarium sacchari causing two distinct diseases, wilt and pokkah boeng. Sugar Tech 19 (6): 638–646.
Zhang, C., J. Wang, H. Tao, X. Dang, Y. Wang, M. Chen, Z. Zhai, W. Yu, L. Xu, W.B. Shim, G. Lu, and Z. Wang. 2015. FvBck1, a component of cell wall integrity MAP kinase pathway, required for virulence and oxidative stress response in sugarcane Pokkah Boeng pathogen. Frontiers in Microbiology 6 (1096): 1–12.
Acknowledgements
This study has been funded by the Philippine Sugar Research Foundation, Inc. (PHILSURIN). The authors would also like to acknowledge the following institutions and persons for their invaluable technical contributions in the formulation of this study: Sugar Regulatory Administration (SRA), BUSCO Sugar Milling Corporation, Bureau of Plant Industry—National Plant Quarantine Service (BPI-NPQS), Ms. Rizalina L. Tiongco, Ms. Rosalyn T. Luzaran, Ms. Chona Untal, Ms. Fatima Florie May Silva, Ms. Aira Waje, Mr. Eddie M. Bueta. The research grant was received from the Philippine Sugar Research Institute Foundation, Inc. and the organization declares no conflict of interest in the subject matter or materials present in the manuscript
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human or Animal Participants
This study does not involve human or animal participants
Rights and permissions
About this article
Cite this article
Samaco, M.A., dela Cueva, F.M. Molecular Characterization of Fusarium spp. Associated with Sugarcane Pokkah Boeng from the Philippines Using Partial Translation Elongation Factor-1α (TEF-1α) Gene Sequences. Sugar Tech 21, 619–630 (2019). https://doi.org/10.1007/s12355-018-0662-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-018-0662-7