Abstract
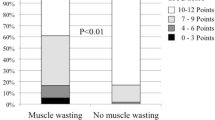
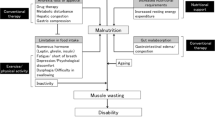
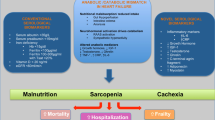
Malnutrition in patients with chronic heart failure (CHF) is not always as severe as muscle wasting in the same patients. Our data showed that 24% of patients with CHF had malnutrition (serum albumin < 3.5 g/dl) while 68% had muscle atrophy. This apparent discrepancy can be explained by considering the metabolic role of the striate muscle. The striate muscle maintains body metabolic performance by continuous exchange of fuel (amino acids) with other organs such as the liver. This happens when the subject is suffering from malnutrition or is starving, and it is regulated by the ratio of catabolic to anabolic molecules such as hormones or cytokines. Glucose is produced when amino acids are metabolized in the liver by gluconeogenesis. Malnutrition, muscle wasting and the frequent progression to cachexia can be reduced by the use of specific therapeutic agents such as cytokines and/or catabolic hormone antagonists. This is because cytokines and catabolic hormones, with consequent insulin resistance (IR), can cause muscle wasting. An alternative and/or complementary therapy may be exogenous supplementation with amino acids. Amino acids: are rapidly absorbed independent of pancreatic activity, reduce IR, induce production in the liver of anabolic molecules such as growth hormone and insulin-like growth factor, and modulate catabolic hormone-mediated effects on adipocytes. Research on the most suitable qualitative and quantitative amino acid composition for an alternative and/or complementary therapy is being undertaken in different research centres.
Similar content being viewed by others
References
Mancini DM, Walter G, Reichek N et al (1999) Contribution of skeletal muscle atrophy to exercise intolerance and altered muscle metabolism in heart failure. Circulation 85:1364–1373
Aquilani R, Opasich C, Cobelli F et al (1994) Lo stato nutrizionale nello scompenso cardiaco cronico. Riv Ital Nutr Parenter Enter 12:1–9
Rady MY, Ryan T, Starr NJ (1997) Clinical characteristics of preoperative hypoalbuminemia predict outcome of cardiovascular surgery. JPEN 21(2):81–90
Gariballa SE, Stuart GP, Taub N et al (1998) Influence of nutritional status on clinical outcome after acute stroke. Am J Clin Nutr 68:275–281
Daniel PM, Pratt OE, Spargo E (1977) The metabolic homeostatic role of muscle and its function as a store of protein. Lancet 2:446–448
Lehninger A (1975) Biochemistry. Worth, New York
Anker SD, Swan JW, Volterrani M et al (1997) The influence of muscle mass, strength, fatigability and blood flow on exercise capacity in cachectic and non-cachectic patients with chronic heart failure. Eur Heart J 18:259–269
Anker SD, Rauchhaus M (1999) Insights into the pathogenesis of chronic heart failure: immune activation and cachexia. Curr Opin Cardiol 14(3):211–216
Neubauer S (2007) The failing heart-an engine out of fuel. N Engl J Med 356:1140–1151
Tartaglia LA, Goeddel DV (1992) Two TNF receptors. Immunol Today 13(5):151–153
Levine B, Kalman J, Mayer L et al (1990) Elevated circulating levels of tumor necrosis factor in severe heart failure. N Engl J Med 323:236–241
Packer M (1988) Neurohormonal interactions in congestive heart failure. Circulation 77:721–730
Sasayama S, Matsumori A, Kihara Y (1999) New insights into the pathophysiological role for cytokines in heart failure. Cardiovasc Res 42:557–564
Hunter JJ, Chien KR (1999) Signalling pathways for cardiac hypertrophy and failure. N Engl J Med 21:1276–1283
Swan JW, Anker SD, Walton C et al (1997) Insulin resistance in chronic heart failure: relation to severity and etiology of heart failure. J Am Coll Cardiol 30(2):527–532
Brouns F, van der Vusse GJ (1998) Utilization of lipids during exercise in human subjects: metabolic and dietary constraints. Br J Nutr 79:117–128
Timmons JA, Gustafsson T, Sundberg CJ et al (1998) Substrate availability limits human skeletal muscle oxidative ATP regeneration at the onset of ischemic exercise. J Clin Invest 101:79–85
Dioguardi FS (1997) Influence of the ingestion of branched chain amino acids on plasma concentrations of ammonia and free fatty acids. J Strength Cond Res 11(4):242–245
Patti ME, Brambilla E, Luzi L et al (1998) Bidirectional modulation of insulin action by amino acids. J Clin Invest 101:1519–1529
Mosoni L, Houlier ML, Mirand PP et al (1993) Effect of amino acids alone or with insulin on muscle and liver protein syntheses in adult and old rats. Am J Physiol 264(4 Pt 1):E614–E620
Giordano M, Castellino P, De Fronzo A (1996) Differential responsiveness of protein synthesis and degradation to amino acid availability in humans. Diabetes 45:393–399
Le Roith D (1996) Insulin-like growth factors. N Engl J Med 336(9):633–640
Quiong L, Baosheng L, Xiaowei W et al (1997) Overexpression of insulin-like growth factor-1 in mice protects from myocyte death after infarction, attenuating ventricular dilation, wall stress and cardiac hypertrophy. J Clin Invest 100:1991–1999
Hesketh JE, Vasconcelos MH, Bermano G (1998) Regulatory signals in messenger RNA: determinants of nutrient-gene interaction and metabolic compartmentation. Br J Nutr 80:307–321
Traxinger RR, Marshall S (1989) Role of amino acids in modulating glucose-induced desensitization of the glucose transport system. J Biol Chem 264:20910–20916
Garlick PJ, McNurlan MA, Patlack CS (1999) Adaptation of protein metabolism in relation to limits to high dietary protein intake. Eur J Clin Nutr 53(S1):S34–S43
Millward DJ, Rivers JP (1988) The nutritional role of indispensable amino acids and the metabolic basis for their requirements. Eur J Clin Nutr 42:367–393
Waterlow JC (1996) The requirements of adult man for indispensable amino acids. Eur J Clin Nutr 50[Suppl 1]:S151–S176
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Pasini, E., Aquilani, R. & Dioguardi, F.S. Malnutrition and chronic heart failure. Mediterr J Nutr Metab 1, 95–98 (2008). https://doi.org/10.1007/s12349-008-0013-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12349-008-0013-4