Abstract
Triple whammy (TW) is a potentially dangerous drug combination that can lead to acute kidney injury (AKI). This drug interaction (DI) occurs when angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin II receptor blockers (ARBs) are used together with diuretics and non-steroidal anti-inflammatory drugs (NSAIDs). One of the most serious consequences of TW DI is an increased risk of developing pre-renal acute kidney injury (pr-AKI). The term TW, in the context of a DI affecting kidney function, is not very widespread. The aim of this article was to gather information on this interaction. Previous knowledge on the mechanism of TW and how to increase patient awareness of this interaction is described. In addition, the specific nature of the acute kidney injury (AKI) caused by triple whammy (AKITW) is presented. On the basis of the current state of knowledge, recommendations on how to manage the TW DI are also demonstrated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The drug interaction called triple whammy (TW) can occur when combining an angiotensin-converting enzyme inhibitor (ACEI) or angiotensin II receptor blocker (ARB) with a diuretic and non-steroidal anti-inflammatory drug (NSAID). |
In certain cases, this interaction can result into acute kidney injury (AKI) and hence contributes to quality of life and reduction of life expectancy, even death of patients. |
TW in a nephrological context has been reported in the literature, but there are very few publications on this subject. |
Our goal was to summarize the current knowledge on this topic, including the characteristics of TW, its consequences and recommendations for clinical practice. |
Introduction
Polypharmacy in chronic patients can lead to numerous dangerous drug interactions (DI) [1,2,3]. Among the many DI there is a peculiar one called triple whammy (TW), seldom described in the literature because of its complexity and its unique way of affecting kidney function [4,5,6]. TW refers to the concurrent use of three types of medications [7]:
-
1.
Angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin II receptor blockers (ARBs)
-
2.
Diuretics
-
3.
Non-steroidal anti-inflammatory drugs (NSAIDs)
This can alter the renal haemodynamic autoregulation system, leading to an increased risk of developing acute pre-renal acute kidney injury (pr-AKI) and AKI [4, 8,9,10]. The main objective of this article is to indicate the mechanisms and features of AKI due to TW DI (AKITW), as well as to describe the prevalence and consequences of the concurrent use of TW drugs and recommendations regarding these products, such as diagnostic evaluations of patients diagnosed with AKI.
Methods
A literature review performed using the search term ‘triple whammy’ in the PubMed medical database gave 48 results, 18 of which were related to the TW DI (as of 24 April 2021). To enhance the number of results we also used the following search term “triple whammy” and the combinations “triple whammy AND AKI”, “triple whammy AND Acute Kidney Injury”, “NSAIDs AND ACEIs AND diuretics AND AKI”, “RAAS AND inhibitors AND NSAID AND diuretics AND AKI”, “renin–angiotensin AND system AND inhibitors AND nonsteroidal AND anti-inflammatory AND drugs AND AKI”, “drugs AND interactions AND AKI”.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Results and Discussion
Phenomenon of Polypharmacy vs the Occurrence of Triple Whammy
Polypharmacy is the concurrent use of five or more medications per day [2, 3]. This is usually a consequence of coexisting chronic diseases requiring pharmacological treatment. The use of evidence-based guidelines for prescribing medications to treat patients suffering from multiple diseases can result in complex and mutually exclusive treatment regimens [1].
The phenomenon of polypharmacy is most common among older adults. Studies show that polypharmacy affects 40–50% of all older adults [1]. This situation is particularly important given that people over 65 years of age (especially those over 80 years of age) account for an increasing share of the total population in Europe. According to Eurostat, in 2019, more than one fifth (20.3%) of the EU-27 population was aged 65 and over [11]. Older adults are prescribed an average of 5.3–6.9 medications [12] and this number continues to grow [3].
The concurrent use of multiple medicinal products can lead to serious adverse event (SAE), poor adherence to therapy [13, 14] and numerous drug–drug interactions (DDIs) and drug–disease interactions (DDisIs). DI are one of the potential mechanisms leading to adverse events (AE) that can be prevented through early intervention [1, 15].
Older adults are more susceptible to the risk of AE due to DI not only because of the number of administered medications but also because the ageing process can cause physiological changes like decreased renal and liver function, which can consequently affect drug pharmacokinetics (i.e. through reduced renal clearance or reduced liver metabolism) [16]. In addition, the frequent occurrence of chronic diseases among older adults increases the risk of DDisIs [1] in which the medication used to treat a disease or symptom can have an influence on another disease or symptom. DDisIs include the use of anti-inflammatory drugs in people with high blood pressure or kidney diseases [1, 2].
Triple Whammy: Mechanism and Risk
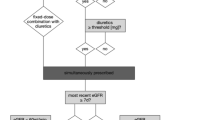
The term “triple whammy”, in relation to an adverse reaction reducing renal function, was first used in 2000 in Australia [9]. It was applied to a specific group of patients who took medications causing triple whammy—they were older adults with comorbidities that included cardiovascular disease, poor perfusion, congestive heart failure (CHF), chronic kidney disease and osteoarthritis. Inhibition of the renin–angiotensin–aldosterone system (RAAS), along with the aforementioned comorbidities, enhances the role of prostaglandins in the regulation and maintenance of haemodynamic glomerular function. The synthesis of prostaglandins is inhibited by NSAIDs.
The mechanism of triple whammy begins with the use of renin–angiotensin system inhibitors (RASI) such as ACEIs or ARBs that dilate efferent arterioles, leading to a decrease in glomerular filtration rate (GFR) and kidney function. The administration of diuretics to such a group of patients can lead to hypovolaemia, which can cause a so-called double whammy and increase the risk of pr-AKI [4, 8, 17, 18]. However, all three drug families (ACEIs, ARBs and diuretics) remain as well-established and safe primary medications of choice in the treatment of various cardiovascular diseases (CVDs) like CHF or peripheral artery disease (PADs) [19]. NSAIDs block the cyclooxygenase (COX) enzymes involved in the synthesis of prostacyclin, prostanoids and prostaglandins (PGE2, PGI2), which play a key role in the physiological regulation of different renal processes. Some of these processes include the regulation of ion and water transport through the nephron and dilation of afferent arteriole increasing the GFR [4, 8, 17, 18]. Therefore, by decreasing the synthesis of prostacyclin, prostanoids and prostaglandins, NSAIDs can constrict the blood flow into the glomerulus via the afferent arteriole and reduce GFR. This increases the risk of TW and AKI [4, 8, 17, 18].
RASI like ACEIs or ARBs are often prescribed together with diuretics for CVDs, while NSAIDs are often used for acute and chronic pain within the group of patients suffering from both conditions. The estimated risk level for patients taking ACEIs or ARBs, diuretics and NSAIDs differs depending on the study. The risk of AKI in the case of NSAIDs used in triple combination is higher than in double combinations with the RASI or just diuretics [8, 18]. In a study [8] involving 2215 identified cases of AKI with an incidence rate 7/10,000 person years during a mean follow-up to 5.9 (SD 3.4) years, the current use of a TW combination (24.6% of all 2215 identified cases) was associated with an increased (31% higher) rate of AKI (rate ratio 1.31, 95% confidence interval 1.12–1.53) in comparison to dual therapy (DT) (18.7% of all 2215 identified cases) containing either diuretics or ACEIs/ARBs with NSAIDs. Another study [18] involved 2226 of identified cases of AKI with incidence rate of 68/10,000 person years. The absolute increase in AKI risk was higher for NSAIDs used in TW versus DT with RASIs or diuretics alone. AKI risk was highest among users of loop diuretic/aldosterone antagonist combinations, in patients over 75 years of age, and in those with pre-existing renal impairment. It has not been unequivocally determined if the occasional short-term use of NSAIDs by patients taking ACEIs or ARBs and diuretics increases the risk. However, it has been estimated that the highest risk of AKI (nearly twice as high) occurs within the first 30 days in patients taking a triple combination [8, 17, 18].
Triple Whammy: Prevalence
Triple whammy has been identified in a variety of studies and the relationship between the occurrence of this combination and adverse events has been proven several times, particularly in the context of pre-existing kidney impairment and the influence on the efficacy of anti-hypertension medications [5, 20]. However, a retrospective cohort study by Lapi et al. did not observe an increased risk of AKI among patients taking diuretics, ACEIs, and ARBs with NSAIDs.
Between 2000 and 2012, the Centre for Adverse Reaction Monitoring (CARM) in New Zealand registered 119 adverse effects by NSAIDs relating to kidney injuries, including 4 deaths and 12 life-threatening cases. The majority of the reports concerned adults over 50 years of age [21]. The cases of acute kidney injury caused by NSAIDs were noted primarily among those patients who took above maximum recommended daily doses of these medications [22].
In 2017, in New Zealand, 115,980 patients (100%) received treatment with an ACE inhibitor or ARB together with a diuretic, which shows the size of the double whammy phenomenon. Out of those who received long-term treatment with an ACE inhibitor or ARB and a diuretic, 26,526 patients (23%) were also dispensed an NSAID [17]. A Polish study [23] based on the analysis of 14,243 prescriptions identified 12 patients aged 56–91 years who had received concurrent medications from the groups likely to cause a triple whammy interaction.
Analysis of the pharmacotherapy based on data from National Healthcare Provider of the Polish patients indicates that in 2018, 6.5 million people bought ARBs or ACEIs, 4.283 million bought diuretics, and 5.9 million bought NSAIDs. Out of 11 million patients taking ARBs or ACEIs and diuretics or NSAIDs, over 660,000 bought medications from all three of the groups within a month, 23.6 thousand of which (6.0%) bought a quantity for at least half a year, being at potential risk of developing AKITW [24].
Medication problems are particularly important among older adults because of the various routes of drug metabolism and the higher risk of adverse events compared to other groups of patients. Older adults are especially susceptible to triple whammy as they are often a priori characterised by impaired renal function and have a tendency for dehydration and hypotension caused by diuretics and inappropriate decreased intake of liquids [25].
Apart from age, the risk factors from AKI caused by triple whammy include any stage of chronic renal failure, decreased hydration, ethnic origin (Māori, Pacific and Indo-Asian peoples), diabetes, heart failure and liver diseases [17, 18, 26, 27].
Specificity of AKI Caused by TW
The term AKI covers a spectrum from relatively decreased urine output and retention of metabolic waste products to complete renal failure. AKI often happens as a complication of hypovolaemia or sepsis, and it should be suspected in all patients taking ACEIs or ARBs and diuretics with NSAIDs who are likely to experience adverse effects [1].
AKI is difficult to verify and analyse as a result of the International Classification of Diseases (ICD-10) term acute renal failure (ARF: N17, N17.8, N17.9). According to KDIGO guidelines, AKI is a subcategory of acute kidney diseases (AKD) and is a broader term that should replace ARF [28].
The KDIGO diagnostic criteria for AKI include [29, 30]:
-
1.
Urine output less than 0.5 ml/kg/h for 6 h
-
2.
Increase in serum creatinine (SCr) (by 0.3 mg/dL observed within 48 h or > 50% within the last 7 days)
It should be highlighted that serum creatinine levels sufficient to diagnose AKI can take 24–36 h to rise [17].
Literature studies on AKI seldom relate to AKI caused by TW. The following studies should be distinguished among them:
-
Studies with a double pharmaceutical intervention [6] consisting of
-
(a) Conducting an educational session among doctors
-
(b) Reviewing medical documentation in which a doctor carried out an intervention in accordance with a pharmacist’s instructions
-
To the best of our knowledge, in previous studies no attention was paid to the parameters differentiating pre-renal AKI from intra-renal AKI, which should be reflected in laboratory parameters (Table 1 [34]).
Triple Whammy: Recommendations
The main objective of treatment of patients with AKITW is to restore renal perfusion. The easiest way to minimise the risk of triple whammy is to avoid the additional therapy with NSAIDs in risk group patients. If the therapy is required, it is recommended to use the smallest effective dose for the shortest period of time possible. It is worth emphasising that the most common reason for prescribing NSAIDs in patients taking ACEIs or ARBs with diuretics is joint pain [8, 20]. In addition, it is recommended to consider discontinuing any medications that can impair kidney function, including those requiring adjusting the dose in the case of deterioration in kidney function, e.g. metformin, gabapentin, atenolol and morphine. In many situations, any unnecessary medications will need to be discontinued until the patient condition improves. It is also recommended to regularly assess kidney function, mainly by a regular monitoring of blood pressure and electrolytes [17, 23].
If it is necessary to combine TW medications, it is advised to check the initial concentration of serum creatinine and electrolytes, and advise the patient to maintain an appropriate intake of fluid [5, 17]. The patient body weight and blood pressure should also be evaluated. It is important to measure body weight, blood pressure, serum creatinine and electrolytes within the first month of treatment because of the increased risk of AKI in this period. In the case of malaise, more frequent patient monitoring is required [17].
Of great importance is the wide spread use of NSAIDs among people with relative contraindications. A study carried out to evaluate the use of NSAIDs in people with identified contraindications showed that 22% of the participants had bought NSAIDs over the counter. Every fifth respondent did not inform the medical staff of taking NSAIDs [27]. That data shows that many patients can consider these medications harmless.
In order to decrease the risk of AKI, patient education is essential. NSAIDs are often patient initiated as most are available without prescription. Doctors or pharmacists often do not know which other medications the patient is taking. In addition, doctors may lack information of medications prescribed by other specialists [2]. Here, the great role of pharmacists should be emphasised. They have essential knowledge to observe the risk of triple whammy in patients. Knowing that patients take medications from ACEIs or ARBs groups with diuretics, they can effectively educate patients on the risks that result from concurrently taking these groups of medications and counsel them against the potential harms of long-term use of NSAIDs.
Conclusions
Triple whammy is one example of a triple interaction. Despite identifying this phenomenon, detailed guidelines regarding monitoring patients taking ACEIs or ARBs with diuretics and NSAIDs have not been developed to this date. The objective of treatment of patients with AKI is to recover renal perfusion by restoration of fluid balance and discontinuation of nephrotoxic medications. AKI can be considered a condition caused among others by a lack of supervision over patient pharmacotherapy. Increasing the role of the pharmacist in clinical supervision over pharmacotherapy (clinical governance) connected with best clinical practice (BCP) can avoid AKI caused by TW (AKITW).
References
Morin L, Johnell K, Laroche ML, Fastbom J, Wastesson JW. The epidemiology of polypharmacy in older adults: register-based prospective cohort study. Clin Epidemiol. 2018;10:289–98.
Onder G, Marengoni A. Polypharmacy. JAMA. 2017;318(17):1728.
Molokhia M, Majeed A. Current and future perspectives on the management of polypharmacy. BMC Fam Pract. 2017;18:70.
Prieto-García L, Pericacho M, Sancho-Martínez SM, et al. Mechanisms of triple whammy acute kidney injury. Pharmacol Ther. 2016;167:132–45.
Camin RMG, Cols M, Chevarria JL, et al. Acute kidney injury secondary to a combination of renin-angiotensin system inhibitors, diuretics and NSAIDS: the triple whammy. Nefrologia. 2015;35(2):197–206.
Arrufat-Goterris G, do Pazo-Oubiña F, Malpartida-Flores M, Rodríguez-Rincón RM. Intervención farmacéutica para reducir el riesgo de iatrogenia asociada a la combinación triple whammy [Pharmaceutical intervention to reduce the iatrogenic risk associated with the triple whammy combination]. Aten Primaria. 2017;49(3):150–55.
Fuentes AV, Pineda MD, Venkata KCN. Comprehension of top 200 prescribed drugs in the US as a resource for pharmacy teaching, training and practice. Pharmacy (Basel). 2018;6(2):43.
Lapi F, Azoulay L, Yin H, Nessim SJ, Suissa S. Concurrent use of diuretics, angiotensin converting enzyme inhibitors, and angiotensin receptor blockers with non-steroidal anti-inflammatory drugs and risk of acute kidney injury: nested case-control study. BMJ. 2013;346:e8525.
Thomas MC. Diuretics, ACE inhibitors and NSAIDs—the triple whammy. Med J Aust. 2000;172(4):184–5.
Prieto-García L, Vicente-Vicente L, Blanco-Gozala V, et al. Pathophysiological mechanisms underlying a rat model of triple whammy acute kidney injury. Lab Invest. 2020;100:1455–64.
Eurostat. Population structure and ageing. https://ec.europa.eu/eurostat/statistics-explained/index.php/Population_structure_and_ageing#Further_Eurostat_information. Accessed Mar 17, 2021.
Rieckert A, Trampisch US, Klaaßen-Mielke R, et al. Polypharmacy in older patients with chronic diseases: a cross-sectional analysis of factors associated with excessive polypharmacy. BMC Fam Pract. 2018;19:113.
Beuscart JB, Petit S, Gautier S, et al. Polypharmacy in older patients: identifying the need for support by a community pharmacist. BMC Geriatr. 2019;19:277.
Patton DE, Hughes CM, Cadogan CA, Ryan CA. Theory-based interventions to improve medication adherence in older adults prescribed polypharmacy: a systematic review. Drugs Aging. 2017;34(2):97–113.
Mousavi S, Ghanbari G. Potential drug-drug interactions among hospitalized patients in a developing country. Caspian J Intern Med. 2017;8:282.
Joint Formulary Committee. BNF 80: September 2020-March 2021. London: Pharmaceutical Press; 2020.
Best Practice Advisory Centre. Avoiding the ‘triple whammy’ in primary care: ACE inhibitor/ARB + diuretic + NSAID. https://bpac.org.nz/2018/docs/triple-whammy.pdf. Accessed June 16, 2021.
Dreischulte T, Morales DR, Bell S, Guthrie B. Combined use of nonsteroidal anti-inflammatory drugs with diuretics and/or renin-angiotensin system inhibitors in the community increases the risk of acute kidney injury. Kidney Int. 2015;88:396–403.
WHO. Cardiovascular diseases (CVDs). https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds). Accessed June 6, 2021.
Zulkifly H, Abdul WMS, Shaharuddin S, Chiau ML, Mat ZH. Be cautious of triple whammy!!! Value Health. 2014;17(7):A467.
Medsafe (New Zealand Medicines and Medical Devices Safety Authority). NSAIDs and acute kidney injury. Prescr Update. 2013;34(2):14–15.
Savage R. CARM reports acute renal failure. Best Pract J. 2007;6:26–7.
Piekarska M, Świeczkowski D, Harężlak T, et al. The combination of angiotensin converting enzyme inhibitors, diuretics and non-steroidal anti-inflammatory drugs in the routine community pharmacy settings in Poland: the unrecognized problem of “triple whammy”: a pilot analysis. J Health Policy Outcomes Res. 2018;1:14–9.
National Health Fund. NHF on health. Polypharmacy. Warsaw; NHF; 2020.
Savage R. A dangerous trio. Prescr Update. 2002;23(2):20.
Huria T, Palmer S, Beckert L, Williman J, Pitama S. Inequity in dialysis related practices and outcomes in Aotearoa/New Zealand: a Kaupapa Māori analysis. Int J Equity Health. 2018;17:27.
Adams RJ, Appleton SL, Gill TK, Taylor AW, Wilson DH, Gill CL. Cause for concern in the use of non-steroidal anti-inflammatory medications in the community—a population-based study. BioMed Central Family Pract. 2011;12:70.
Ostermann M, Joannidis M. Acute kidney injury 2016: diagnosis and diagnostic workup. Crit Care. 2016;20:299.
Levey AS, Eckardt KU, Dorman NM, Christiansen SL, Hoorn EJ, Ingelfinger JR. Nomenclature for kidney function and disease: report of a Kidney Disease: Improving Global Outcomes (KDIGO) Consensus Conference. Kidney Int. 2020;97(6):1117–29.
Chawla LS, Bellomo R, Bihorac A, et al. Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol. 2017;13:241–57.
Loboz KK, Shenfield GM. Drug combinations and impaired renal function—the “triple whammy.” Br J Clin Pharmacol. 2005;59(2):239–43.
Fournier JP, Sommet A, Durrieu G, et al. More on the “triple whammy”: antihypertensive drugs, non-steroidal anti-inflammatory agents and acute kidney injury—a case/non-case study in the French pharmacovigilance database. Ren Fail. 2014;36(7):1166–8.
Piccinni C, Lionello L, Raschi E, et al. Hospitalisations by drug interactions with nsaids in elderly poly-treated patients: outcome research on administrative databases. Clin Ther. 2015;37(8 suppl):E101–2.
Szczeklik A, Gajewski P. Interna szczeklika. Kraków: Medycyna Praktyczna; 2020.
Acknowledgements
Thank you to Dr Matthew Allan (MBBS, DCH, BPharm, FRACGP) from Ulladulla Medical Clinic, Philip Centre Shop 6 / Princes Highway Ulladulla, NSW 2539 Australia for proofreading of the final version of the manuscript and language corrections as well as Dr Régis Vaillancourt (OMM, CD, B.Pharm, Pharm D, FCSHP, FFIP, FOPQ), Director of Pharmacy at Children’s Hospital of Eastern Ontario Centre , 401 Smyth Road Ottawa, ON K1H 8L1 for inspiration to undertake this project in Poland.
Funding
The journal’s Rapid Service Fee and open access fee were funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Conceptualization: TH; methodology: TH; writing—original draft preparation: UR; writing—review and editing: TH, PM, DH, FS, ANP, AB and UR; visualization: UR; supervision: TH and MA; language proofreading: MA.
Disclosures
Tomasz Harężlak, Urszula Religioni, Filip M. Szymański, Dagmara Hering, Agnieszka Barańska, Agnieszka Neumann-Podczaska, Matthew Allan, and Piotr Merks have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Harężlak, T., Religioni, U., Szymański, F.M. et al. Drug Interactions Affecting Kidney Function: Beware of Health Threats from Triple Whammy. Adv Ther 39, 140–147 (2022). https://doi.org/10.1007/s12325-021-01939-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-021-01939-9