Abstract
Introduction
Robotic radical prostatectomy requires prolonged pneumoperitoneum and a steep Trendelenburg position. Magnesium can attenuate the stress response and hemodynamic perturbations. This study aimed to evaluate the effects of intravenous magnesium administration on hemodynamics and the stress response in patients undergoing robotic radical prostatectomy.
Methods
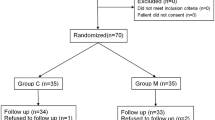
In this prospective, double-blind, randomized controlled study, 52 patients undergoing robotic radical prostatectomy were randomized into two groups: 26 in the magnesium group and 26 in the control group. The patients in the magnesium group received magnesium sulfate 50 mg/kg intravenously, followed by infusion at a rate of 10 mg/kg/h during surgery. The patients in the control group received an equal volume of 0.9% saline. The primary outcomes were the changes in heart rate and mean arterial pressure (MAP) during surgery. The serum stress hormones (adrenocorticotropic hormone, cortisol, epinephrine, and norepinephrine) were also measured.
Results
MAP showed a significant intergroup difference over time (Pgroup*time = 0.017); it increased significantly at 5 min after Trendelenburg position in the control group and decreased significantly at 30 min after Trendelenburg position in the magnesium group. The intergroup difference in the change in cortisol concentrations was significant over time (Pgroup*time = 0.006). The cortisol concentration decreased significantly from baseline to 24 h after surgery in the magnesium group but did not change significantly in the control group. The requirement for intraoperative remifentanil was 35% lower in the magnesium group (P = 0.011), and the severity of postoperative pain at 30 min and 6 h after surgery was also lower in the magnesium group (P = 0.024 and P = 0.015).
Conclusion
There is a possibility that intravenous magnesium administration during robotic radical prostatectomy reduces the increases in arterial pressure, cortisol concentrations, opioid requirements, and postoperative pain.
Trial Registration
ClinicalTrials.gov identifier, NCT02833038



Similar content being viewed by others
References
Cao L, Yang Z, Qi L, Chen M. Robot-assisted and laparoscopic vs open radical prostatectomy in clinically localized prostate cancer: perioperative, functional, and oncological outcomes: a systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e15770.
Demasi CL, Porpiglia F, Tempia A, D’Amelio S. Ocular blood flow in steep Trendelenburg positioning during robotic-assisted radical prostatectomy. Eur J Ophthalmol. 2018;28:333–8.
Chen K, Wang L, Wang Q, et al. Effects of pneumoperitoneum and steep Trendelenburg position on cerebral hemodynamics during robotic-assisted laparoscopic radical prostatectomy: a randomized controlled study. Medicine (Baltimore). 2019;98:e15794.
Gibbison B, Angelini GD, Lightman SL. Dynamic output and control of the hypothalamic-pituitary-adrenal axis in critical illness and major surgery. Br J Anaesth. 2013;111:347–60.
Porcaro AB, de Luyk N, Corsi P, et al. Robotic assisted radical prostatectomy accelerates postoperative stress recovery: final results of a contemporary prospective study assessing pathophysiology of cortisol peri-operative kinetics in prostate cancer surgery. Asian J Urol. 2016;3:88–95.
Porcaro AB, Molinari A, Terrin A, et al. Robotic-assisted radical prostatectomy is less stressful than the open approach: results of a contemporary prospective study evaluating pathophysiology of cortisol stress-related kinetics in prostate cancer surgery. J Robot Surg. 2015;9:249–55.
Queiroz Rangel Micuci AJ, Verçosa N, Filho PAG, de Boer HD, Barbosa DD, Cavalcanti IL. Effect of pretreatment with magnesium sulphate on the duration of intense and deep neuromuscular blockade with rocuronium: a randomised controlled trial. Eur J Anaesthesiol. 2019;36:502–8.
Albrecht E, Kirkham KR, Liu SS, Brull R. Peri-operative intravenous administration of magnesium sulphate and postoperative pain: a meta-analysis. Anaesthesia. 2013;68:79–90.
Sohn HM, Jheon SH, Nam S, Do SH. Magnesium sulphate improves pulmonary function after video-assisted thoracoscopic surgery: a randomised double-blind placebo-controlled study. Eur J Anaesthesiol. 2017;34:508–14.
Mesbah Kiaee M, Safari S, Movaseghi GR, et al. The effect of intravenous magnesium sulfate and lidocaine in hemodynamic responses to endotracheal intubation in elective coronary artery bypass grafting: a randomized controlled clinical trial. Anesth Pain Med. 2014;4:e15905.
Honarmand A, Safavi M, Badiei S, Daftari-Fard N. Different doses of intravenous magnesium sulfate on cardiovascular changes following the laryngoscopy and tracheal intubation: a double-blind randomized controlled trial. J Res Pharm Pract. 2015;4:79–84.
Kutlesic MS, Kutlesic RM, Mostic-Ilic T. Magnesium in obstetric anesthesia and intensive care. J Anesth. 2017;31:127–39.
El Mourad MB, Arafa SK. Effect of intravenous versus intraperitoneal magnesium sulfate on hemodynamic parameters and postoperative analgesia during laparoscopic sleeve gastrectomy-A prospective randomized study. J Anaesthesiol Clin Pharmacol. 2019;35:242–7.
Tan W, Qian DC, Zheng MM, Lu X, Han Y, Qi DY. Effects of different doses of magnesium sulfate on pneumoperitoneum-related hemodynamic changes in patients undergoing gastrointestinal laparoscopy: a randomized, double-blind, controlled trial. BMC Anesthesiol. 2019;19:237.
Zhang J, Wang Y, Xu H, Yang J. Influence of magnesium sulfate on hemodynamic responses during laparoscopic cholecystectomy: a meta-analysis of randomized controlled studies. Medicine (Baltimore). 2018;97:e12747.
Zarif P, Abdelaal Ahmed Mahmoud A, Abdelhaq MM, Mikhail HM, Farag A. Dexmedetomidine versus magnesium sulfate as adjunct during anesthesia for laparoscopic colectomy. Anesthesiol Res Pract. 2016. https://doi.org/10.1155/2016/7172920.
Del Giorno R, Lavorato Hadjeres S, Stefanelli K, et al. Consequences of supraphysiological dialysate magnesium on arterial stiffness, hemodynamic profile, and endothelial function in hemodialysis: a randomized crossover study followed by a non-controlled follow-up phase. Adv Ther. 2020;37:4848–65.
Jahnen-Dechent W, Ketteler M. Magnesium basics. Clin Kidney J. 2012;5:i3–14.
Kalmar AF, Foubert L, Hendrickx JF, et al. Influence of steep Trendelenburg position and CO(2) pneumoperitoneum on cardiovascular, cerebrovascular, and respiratory homeostasis during robotic prostatectomy. Br J Anaesth. 2010;104:433–9.
Falabella A, Moore-Jeffries E, Sullivan MJ, Nelson R, Lew M. Cardiac function during steep Trendelenburg position and CO2 pneumoperitoneum for robotic-assisted prostatectomy: a trans-oesophageal Doppler probe study. Int J Med Robot. 2007;3:312–5.
Haas S, Haese A, Goetz AE, Kubitz JC. Haemodynamics and cardiac function during robotic-assisted laparoscopic prostatectomy in steep Trendelenburg position. Int J Med Robot. 2011;7:408–13.
Lestar M, Gunnarsson L, Lagerstrand L, Wiklund P, Odeberg-Wernerman S. Hemodynamic perturbations during robot-assisted laparoscopic radical prostatectomy in 45° Trendelenburg position. Anesth Analg. 2011;113:1069–75.
Meininger D, Westphal K, Bremerich DH, et al. Effects of posture and prolonged pneumoperitoneum on hemodynamic parameters during laparoscopy. World J Surg. 2008;32:1400–5.
Rosendal C, Markin S, Hien MD, Motsch J, Roggenbach J. Cardiac and hemodynamic consequences during capnoperitoneum and steep Trendelenburg positioning: lessons learned from robot-assisted laparoscopic prostatectomy. J Clin Anesth. 2014;26:383–9.
D’Alonzo RC, Gan TJ, Moul JW, et al. A retrospective comparison of anesthetic management of robot-assisted laparoscopic radical prostatectomy versus radical retropubic prostatectomy. J Clin Anesth. 2009;21:322–8.
Alfonsi P, Vieillard-Baron A, Coggia M, et al. Cardiac function during intraperitoneal CO2 insufflation for aortic surgery: a transesophageal echocardiographic study. Anesth Analg. 2006;102:1304–10.
Dubé L, Granry JC. The therapeutic use of magnesium in anesthesiology, intensive care and emergency medicine: a review. Can J Anaesth. 2003;50:732–46.
Laurant P, Touyz RM. Physiological and pathophysiological role of magnesium in the cardiovascular system: implications in hypertension. J Hypertens. 2000;18:1177–91.
Beers R, Camporesi E. Remifentanil update: clinical science and utility. CNS Drugs. 2004;18:1085–104.
Albrecht S, Hering W, Schüttler J, Schwilden H. New intravenous anesthetics. Remifentanil, S(+)-ketamine, eltanolone and target controlled infusion. Anaesthesist. 1996;45:1129–41.
Quinto D, Reis ST, Zampolli LJ, et al. Robotically assisted laparoscopic radical prostatectomy induces lower tissue trauma than radical retropubic prostatectomy. J Robot Surg. 2020. https://doi.org/10.1007/s11701-020-01150-y.
De Oliveira GS Jr, Castro-Alves LJ, Khan JH, McCarthy RJ. Perioperative systemic magnesium to minimize postoperative pain: a meta-analysis of randomized controlled trials. Anesthesiology. 2013;119:178–90.
Lennon FE, Moss J, Singleto PA. The μ-opioid receptor in cancer progression: is there a direct effect? Anesthesiology. 2012;116:940–5.
Gröber U, Schmidt J, Kisters K. Magnesium in prevention and therapy. Nutrients. 2015;7:8199–226.
Blaszczyk U, Duda-Chodak A. Magnesium: its role in nutrition and carcinogenesis. Rocz Panstw Zakl Hig. 2013;64:165–71.
Dai Q, Motley SS, Smith JA Jr, et al. Blood magnesium, and the interaction with calcium, on the risk of high-grade prostate cancer. PLoS One. 2011;6:e18237.
Neeman E, Ben-Eliyahu S. Surgery and stress promote cancer metastasis: new outlooks on perioperative mediating mechanisms and immune involvement. Brain Behav Immun. 2013;30(Suppl):S32-40.
Kumagai Y, Ohzawa H, Miyato H, et al. Surgical stress increases circulating low-density neutrophils which may promote tumor recurrence. J Surg Res. 2020;246:52–61.
Acknowledgements
The authors thank the participants and participating staff for their cooperation in the study.
Funding
This study and the Rapid Service Fee were sponsored by the new faculty research fund of Ajou University School of Medicine, Suwon, South Korea.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Ha Yeon Kim, Sook Young Lee, Hye Sun Lee, Bo Kyeong Jun, Jong Bum Choi, and Ji Eun Kim have nothing to disclose.
Compliance with Ethics Guidelines
The study protocol was approved by the Institutional Review Board of Ajou University School of Medicine. The study was performed in accordance with the Declaration of Helsinki of 1964 and its later amendments. Written informed consent was obtained from all study participants prior to participation.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H.Y., Lee, S.Y., Lee, H.S. et al. Beneficial Effects of Intravenous Magnesium Administration During Robotic Radical Prostatectomy: A Randomized Controlled Trial. Adv Ther 38, 1701–1712 (2021). https://doi.org/10.1007/s12325-021-01643-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-021-01643-8