Abstract
Introduction
Peripheral neuropathic pain (PNP) is defined as the neuropathic pain that arises either acutely or in the chronic phase of a lesion or disease affecting the peripheral nervous system. PNP is associated with a remarkable disease burden, and there is an increasing demand for new therapies to be used in isolation or combination with currently available treatments. The aim of this systematic review was to evaluate the current evidence, derived from randomized controlled trials (RCTs) that assess non-pharmacological interventions for the treatment of PNP.
Methods
After a systematic Medline search, we identified 18 papers eligible to be included.
Results
The currently best available evidence (level II of evidence) exist for painful diabetic peripheral neuropathy. In particular, spinal cord stimulation as adjuvant to conventional medical treatment can be effectively used for the management of patients with refractory pain. Similarly, adjuvant repetitive transcranial magnetic stimulation of the motor cortex is effective in reducing the overall pain intensity, whereas adjuvant static magnetic field therapy can lead to a significant decrease in exercise-induced pain. Weaker evidence (level III of evidence) exists for the use of acupuncture as a monotherapy and neurofeedback, either as an add-on or a monotherapy approach, for treatment of painful chemotherapy-induced peripheral neuropathy
Conclusions
Future RCTs should be conducted to shed more light in the use of non-pharmacological approaches in patients with PNP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Non-pharmacological approaches can be used for treatment of peripheral neuropathic pain. |
Adjuvant repetitive transcranial magnetic stimulation of the motor cortex is effective in reducing the overall pain intensity in patients with diabetic polyneuropathy (level II evidence). |
Adjuvant static magnetic field therapy can lead to a significant decrease in exercise-induced pain in patients with diabetic polyneuropathy (level II evidence). |
Acupuncture as a monotherapy and neurofeedback, either as an add-on or a monotherapy approach, can be used for treatment of painful chemotherapy-induced peripheral neuropathy (level III evidence). |
Introduction
The term peripheral neuropathy (PN) refers to any disorder of the peripheral nervous system including single and multiple (asymmetric) mononeuropathy and symmetrical involvement of many nerves (polyneuropathy) [1]. Many aetiological factors have been implicated in the development of peripheral neuropathy including diabetes, cancer, drug toxicity, vitamin deficiencies, excessive alcohol consumption, increased oxidative stress, gluten sensitivity and genetic [2,3,4,5,6,7,8,9].
Sensory symptoms that are seen in PN include tingling, pins and needles sensations, numbness, tightness, burning, itchiness, sensory ataxia and pain. The latter is very prevalent as it affects up to two thirds of patients, independently of aetiology [10,11,12].
Presence of peripheral neuropathic pain (PNP) leads to an overall poor quality of life, regardless of disease severity [13, 14]. PNP is challenging to control, as the response to pharmacological monotherapy is frequently suboptimal, and combination treatments are often necessary for adequate pain control [15]. These regimens may be also associated with potentially serious adverse effects, which may not be well tolerated by the patients. Given the remarkable disease burden associated with PNP and the limited response to the currently available management strategies, there is a demand for new therapies to be used in isolation or combination with current treatments.
Non-pharmacological interventions have been proposed to be used in PNP, mostly as adjuvant to pharmacological treatments [16,17,18]. The aim of this systematic review was to evaluate the currently available evidence, derived from randomized controlled trials (RCTs) that assess non-pharmacological interventions for the treatment of peripheral neuropathic pain.
Methods
Literature Search Strategy
A systematic literature search was performed on April 12, 2020, in the PubMed database. For the PubMEd search, three medical subject heading (MeSH) terms were used. Term A was “pain” OR “painful”. Term B was “neuropathy” OR “polyneuropathy”. Term C was “randomised” OR randomized”. The filter “clinical trial” was applied. We also searched at clinicaltrials.gov, a resource provided by the US National Library of Medicine for unpublished trials. For the search we used the same MeSH terms as above. The filters “with results” AND “completed” were applied. The reference lists of included articles were further screened to identify further studies that may fall within the scope of this review.
Inclusion Criteria
Articles eligible to be included in this review were required to meet the following criteria:
-
1.
Human subjects were involved.
-
2.
The full article was written in English language.
-
3.
They were RCTs investigating non-pharmacological approaches for the management of painful polyneuropathy.
-
4.
Papers were of adequate methodological quality as described below.
Exclusion Criteria
Articles meeting the following criteria were excluded from our review:
-
1.
Studies not referring to polyneuropathy.
-
2.
Non-original articles (i.e. review articles, letters, medical hypotheses, etc.).
-
3.
Non-pharmacological studies where pain relief was not the primary aim.
-
4.
Trials with less than ten patients per treatment arm.
-
5.
Withdrawal trials.
-
6.
Duplicate articles or papers from the same research teams describing the same patient population.
-
7.
Pharmacological trials.
All article abstracts were screened in triplicate in a blinded fashion. Those found to meet any of the exclusion criteria were removed and any controversies were dealt with consensus during a face-to-face meeting, in which the abstracts were reviewed. All remaining papers were screened again as a full article by at least three authors and conflicts were settled as previously noted.
Quality Assessment of Included Studies
All studies were initially screened for bias using the JADAD scoring system [19]. Trials with JADAD score < 3 were excluded. Studies with JADAD score ≥ 3 were further assessed using the Cochrane Collaboration’s tool [20]. This information is available as Supplementary material.
Clinical Recommendations
To determine the grading of evidence we used the classification proposed by the American Society of Interventional Pain Physicians (ASIPP), where applicable [21].
Data Collection Process
Data were extracted from each study in a structured coding scheme using Excel and included population size, gender and age distribution, the type of polyneuropathy, means of diagnosis, means of treatment, duration of RCT, response to treatment, way of assessment of effectiveness, side effects associated with the treatment and the follow-up period of the patients, where applicable. When there was uncertainty regarding how data should be interpreted or utilized, at least three authors discussed the study in question to ensure consensus.
Data Synthesis
This study used aggregate data where possible and it is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [22].
Compliance with Ethics Guidelines
This article is based on previously conducted studies. Thus, there are no ethical concerns in respect to this study.
Results
Study Characteristics
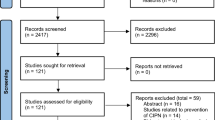
Our search strategy produced a total of 538 results. During the eligibility assessment 520 articles were excluded. Ultimately, a total of 18 completed studies published between 2000 and 2020 were included in the present review [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40]. The selection process is illustrated in Fig. 1 (PRISMA chart). A summary of the characteristics of the included studies is available as Supplementary material.
Spinal Cord Stimulation
Spinal cord stimulation (SCS) is a pain neuromodulation technique. There are two components of a fully implanted SCS system: the electrodes (or lead) and the implantable pulse generator (IPG). Through these implantable electrodes SCS alters the local neurochemistry in dorsal horns, suppressing the hyperexcitability of the neurons with variable pulse-width modulation [41, 42].
Adjuvant SCS for the management of refractory painful diabetic peripheral neuropathy has been used in two open-label controlled studies, showing a significant reduction of pain intensity scores, maintained over at least 6 months [23, 24]. However, SCS has been associated with severe adverse events including infection after the SCS implantation and dural puncture leading to a subdural hematoma and subsequently death in one patient [24].
Transcutaneous Electrical and Electromagnetic Stimulation
The active principle of Scrambler Therapy is that synthetic “non-pain” information is transmitted by C fiber surface receptors. This is a different theoretical mechanism than the traditional electric stimulation of A-beta fibers to produce paresthesia and/or block the conduction of nerve fibers to produce an analgesic effect, which happens via transcutaneous electrical nerve stimulation (TENS) [43].
Electrical stimulation through stocking electrodes as add-on treatment has been studied in a small double-blind crossover study [25] of patients with painful diabetic peripheral neuropathy showing, however, no beneficial effect in reducing pain intensity.
In a 2-week open-label controlled study, Loprinzi et al. studied the efficacy of scrambler therapy as a monotherapy versus TENS in patients with chemotherapy-induced polyneuropathy [26]. Scrambler therapy was well-tolerated and effective in reducing the pain. However, no significant differences in pain reduction were found between the two interventions. In a randomized double-blind sham-controlled trial, the authors failed to show any beneficial effect of adjuvant scrambler therapy compared to sham treatment in patients with chemotherapy-induced polyneuropathy after a 3-month follow-up assessment [27].
Frequency-modulated electromagnetic neural stimulation (FREMS) is a novel transcutaneous electrotherapy that involves sequences of voltage-controlled modulated electrical stimuli which vary on regards with pulse frequency and duration [44] leading to gradual recruitment of membrane potentials in the targeting tissues [45]. This technique was designed on the basis of the hypothesis that the summation of sub-threshold electrical stimuli, conveyed through the skin proximal to a motor nerve in a non-invasive system, would induce composite motor action potentials in excitable tissues [28].
In a randomized double-blind crossover trial, Bosi et al. applied FREMS as a monotherapy on patients with painful diabetic polyneuropathy finding statistically significant improvement in pain intensity that was maintained for at least 4 months. Besides the analgesic effect, an improvement in the overall quality of life as well as a significant improvement of the motor nerve conduction velocity was found [28].
Low-frequency pulsed electromagnetic stimulation as add-on has been studied in three double-blind sham-controlled RCTs, all of which showed no superiority of the active treatment when used in patients with painful diabetic polyneuropathy [29, 31].
However, in a double-blind sham-controlled study, Weintraub et al. showed that adjuvant static magnetic field therapy can lead to a significant decrease in exercise induced pain due to diabetic polyneuropathy [32].
Interferential therapy is another electrophysical treatment, in which electrical stimuli are applied on the painful area via two or four electrodes using medium-frequency alternating current (MFAC) at frequencies between 1 and 1500 Hz [46, 47]. Lindblad et al. studied the therapeutic effect of interferential therapy and long-wave diathermy as a monotherapy in patients with chemotherapy-induced painful polyneuropathy and found that long-wave diathermy at low power is effective in decreasing the pain intensity whereas the combination of interferential therapy and long-wave diathermy at high power is not [33].
Repetitive Magnetic Stimulation
Transcranial magnetic stimulation (TMS) is a neurostimulation and neuromodulation technique, based on the principle of electromagnetic induction of an electric field in the brain [48]. The stimulation parameters as well as the duration of the modulation are variable and can prompt either an amplifying or a suppressing effect [49]. Essentially, low-frequency rTMS (≤ 1 Hz) results in inhibition of motor cortical excitability [50, 51], while application of high-frequency rTMS (5–20 Hz) can lead to long-lasting inhibitory effects [52, 53].
Onesti et al. showed that adjuvant repetitive transcranial magnetic stimulation (rTMS) of the motor cortex is effective in reducing the pain intensity of patients with diabetic polyneuropathy [34], an effect that persisted for at least 3 weeks.
Photon Stimulation
Photon stimulation is regarded as either a pulsed infrared light therapy [54] or a photobiomodulation [55], and these vary in terms of wavelength. At the cellular level, photobiomodulation can improve cellular metabolism and accelerate tissue and cell repair [55, 56]. It has been demonstrated that cytochrome c-oxidase (COX) is responsible for the light absorption [57] and nitric oxide (NO) is responsible for the inhibition of the respiration by binding to COX [58]. Photon stimulation targets the bond between COX and NO leading to segregation and mitochondrial disinhibition or respiration [59].
Low-intensity laser therapy (LILT) is treatment based on photochemical modulation, which triggers biochemical changes [60] at optimal wavelengths and energy densities. This novel noninvasive technique can only deliver monochromatic light in the far-red to near-infrared region of the spectrum (630–1000 nm) [55].
The use of adjuvant photon stimulation for the treatment of peripheral neuropathic pain due to diabetic polyneuropathy of the lower limbs has been studied in a double-blind sham-controlled RCT by Swislocki et al. [35]. Patients receiving photon stimulation reported improved sensation, social functioning and mental health compared to sham stimulation. However, no differences over time in pain intensity or pain relief scores were found between the two groups.
In an 8-week double-blind RCT, Zinman et al. considered the efficacy and safety of adjuvant LILT in 50 subjects suffering from painful diabetic peripheral neuropathy [36]. Initially, all subjects were treated with sham laser therapy for 2 weeks, achieving a decrease in the mean pain score. Then, for 4 weeks patients were randomized to receive LILT or sham laser therapy. Only patients receiving LILT achieved a further reduction in their pain scores, though these were not statistically significant compared to sham therapy (p = 0.07). However, this effect was not maintained 2 weeks after completion of treatment. Although patients had already been treated with multiple medications, no complications were reported with add-on laser therapy, showing that laser therapy is a safe and well-tolerated treatment option in patients with comorbidities.
Vibration Stimulation
Vibratory stimulation can be applied via a device either on the whole body or at a specific painful area with various pulses [61], which results in enhancement of neuromuscular activation and improvement of skeletal muscle function [62, 63] probably through muscle tuning [64]. Whole-body vibration is typically applied indirectly via the lower limbs with the patient standing in a half squat position on a vibrating platform [65].
Paice et al. investigated the efficacy of a 45-min vibration at a frequency of 60 Hz as a monotherapy in a double-blind sham-controlled RCT of 40 patients with moderate to severe HIV-associated polyneuropathy [37]. Statistically significant reduction of pain intensity was achieved in both treatment arms. However, no statistically significant difference of post-treatment benefit was found between the two arms.
Acupuncture
Acupuncture is a minimally invasive procedure, which derives from traditional Chinese medicine and is based on the meridian theory. The meridian theory is based on empirical experience. Although the meridians have not been formally characterized as anatomical structures, they seem to serve as a road map to identify the location of various acupoints [66].
Garrow et al. conducted a 10-week single-blind sham-controlled RCT in patients with intractable peripheral pain in the lower limbs due to diabetes and showed that adjuvant acupuncture improves the pain intensity, but not significantly compared to sham intervention [38].
In an 8-week single-blind RCT, Molassiotis et al. investigated the efficacy of acupuncture as a monotherapy in patients with painful chemotherapy-induced polyneuropathy. A statistically significant improvement was achieved in the acupuncture arm [39].
Neurofeedback
Neurofeedback is a learning intervention that aims to have patients control their brain waves consciously and assess their progress [67]. It has been successfully used in combination with regular medication for a variety of health issues, including pain conditions [68, 69], traumatic brain injuries [70] and sleep difficulties [71].
Prinsloo et al. [40] investigated the efficacy of a neurofeedback intervention as either monotherapy or add-on in a small sample of subjects with chemotherapy-induced polyneuropathy. At 4 months post-intervention, the maximum pain score was significantly reduced in the group that had received the intervention compared to patients that had not. Similarly, significant improvements were demonstrated for other neuropathic symptoms such as numbness and tingling.
Discussion
Our systematic review underscores the lack of good quality studies that are focusing on non-pharmacological approaches for the treatment of PNP. The majority of the available RCTs test non-pharmacological approaches as adjuvant to the available pharmacological treatments.
Using the ASIPP criteria the currently best available evidence (level II evidence) exists for painful diabetic peripheral neuropathy. In particular, spinal cord stimulation as adjuvant to conventional medical treatment can be effectively used for the management of patients with refractory pain. Similarly, adjuvant rTMS of the motor cortex is effective in reducing the overall pain intensity, whereas adjuvant static magnetic field therapy can lead to a significant decrease in exercise-induced pain.
Weaker evidence (level III evidence) exists for the use of acupuncture as a monotherapy and neurofeedback, as either an add-on or a monotherapy approach, for treatment of painful CIPN.
Our results should be interpreted with some caution given the limitations of our design. First, there was a great deal of heterogeneity between the studies, involving different techniques and different treatment approaches, thus not allowing for a meta-analysis to be done. Second, we only searched for publications in PubMed and clinicaltrials.gov, and we therefore might have missed a few more papers that are indexed only in other databases.
Future RCTs should be conducted to shed more light on the use of non-pharmacological approaches in patients with other forms of PNP as currently the available evidence to make clinical recommendations concerns patients with diabetic PN and patients with CIPN.
References
Zis P, Sarrigiannis PG, Rao DG, Hewamadduma C, Hadjivassiliou M. Chronic idiopathic axonal polyneuropathy: a systematic review. J Neurol. 2016;263(10):1903–10.
Gross JL, De Azevedo MJ, Silveiro SP, Canani LH, Caramori ML, Zelmanovitz T. Diabetic nephropathy: diagnosis, prevention, and treatment. Diabetes Care. 2005;28(1):164–76.
Zis P, Varrassi G. Painful peripheral neuropathy and cancer. Pain Ther. 2017;6(2):115–6.
Brozou V, Vadalouca A, Zis P. Pain in platin-induced neuropathies: a systematic review and meta-analysis. Pain Ther. 2018;7(1):105–19.
Zis P, Paladini A, Piroli A, McHugh PC, Varrassi G, Hadjivassiliou M. Pain as a first manifestation of paraneoplastic neuropathies: a systematic review and meta-analysis. Pain Ther. 2017;6(2):143–51.
Julian T, Glascow N, Syeed R, Zis P. Alcohol-related peripheral neuropathy: a systematic review and meta-analysis. J Neurol. 2019;266(12):2907–19.
Zis P, Rao DG, Sarrigiannis PG, Aeschlimann P, Aeschlimann DP, Sanders D, et al. Transglutaminase 6 antibodies in gluten neuropathy. Dig Liver Dis. 2017;49(11):1196–200.
Mallet ML, Hadjivassiliou M, Sarrigiannis PG, Zis P. The role of oxidative stress in peripheral neuropathy. J Mol Neurosci. 2020. https://doi.org/10.1007/s12031-020-01495-x(published online ahead of print, 2020 Feb 26).
Zis P, McHugh PC, Manca M, Sarrigiannis PG, Rao DG, Hadjivassiliou M. Increased oxidative stress as a risk factor in chronic idiopathic axonal polyneuropathy. J Mol Neurosci. 2018;66(4):547–51.
Zis P, Sarrigiannis PG, Rao DG, Hadjivassiliou M. Gluten neuropathy: prevalence of neuropathic pain and the role of gluten-free diet. J Neurol. 2018;265(10):2231–6.
Zis P, Sarrigiannis PG, Rao DG, Hewamadduma C, Hadjivassiliou M. Chronic idiopathic axonal polyneuropathy: prevalence of pain and impact on quality of life. Brain Behav. 2019;9(1):e01171.
Michaelides A, Hadden RDM, Sarrigiannis PG, Hadjivassiliou M, Zis P. Pain in chronic inflammatory demyelinating polyradiculoneuropathy: a systematic review and meta-analysis. Pain Ther. 2019;8(2):177–85.
Girach A, Julian TH, Varrassi G, Paladini A, Vadalouka A, Zis P. Quality of life in painful peripheral neuropathies: a systematic review. Pain Res Manag. 2019;2019:2091960.
Zis P, Sarrigiannis PG, Rao DG, Hadjivassiliou M. Quality of life in patients with gluten neuropathy: a case–controlled study. Nutrients. 2018;10(6):662.
Freynhagen R, Bennett MI. Diagnosis and management of neuropathic pain. BMJ. 2009;339:b3002.
Aamir A, Girach A, Sarrigiannis PG, Hadjivassiliou M, Paladini A, Varassi G, et al. Repetitive magnetic stimulation for the management of peripheral neuropathic pain: a systematic review. Adv Ther. 2020;37(3):998–1012.
Zis P, Shafique F, Hadjivassiliou M, Blackburn D, Venneri A, Iliodromiti S, et al. Safety, tolerability, and nocebo phenomena during transcranial magnetic stimulation: a systematic review and meta-analysis of placebo-controlled clinical trials. Neuromodulation. 2020;23(3):291–300.
Vitoula K, Venneri A, Varrassi G, Paladini A, Sykioti P, Adewusi J, et al. Behavioral therapy approaches for the management of low back pain: an up-to-date systematic review. Pain Ther. 2018;7(1):1–12.
Jadad AR, Moore RA, Caroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12.
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;18(343):d5928.
Manchikanti L, Falco FJ, Benyamin RM, Kaye AD, Boswell MV, Hirsch JA. A modified approach to grading of evidence. Pain Physician. 2014;17(3):E319–E325325.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097.
Slangen R, Schaper NC, Faber CG, Joosten EA, Dirksen CD, van Dongen RT, et al. Spinal cord stimulation and pain relief in painful diabetic peripheral neuropathy: a prospective two-center randomized controlled trial. Diabetes Care. 2014;37(11):3016–24.
de Vos CC, Meier K, Zaalberg PB, Nijhuis HJ, Duyvendak W, Vesper J, et al. Spinal cord stimulation in patients with painful diabetic neuropathy: a multicentre randomized clinical trial. Pain. 2014;155(11):2426–31.
Oyibo SO, Breislin K, Boulton AJ. Electrical stimulation therapy through stocking electrodes for painful diabetic neuropathy: a double blind, controlled crossover study. Diabet Med. 2004;21(8):940–4.
Loprinzi C, Le-Rademacher JG, Majithia N, McMurray RP, O'Neill CR, Bendel MA, et al. Scrambler therapy for chemotherapy neuropathy: a randomized phase II pilot trial. Support Care Cancer. 2020;28(3):1183–97.
ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000 Feb 29. Identifier NCT02111174, scrambler therapy in the treatment of chronic chemotherapy-induced peripheral neuropathy; 2014 Apr 11 [cited 2020 July 23]; [about 4 screens]. https://clinicaltrials.gov/ct2/show/NCT02111174. Accessed 23 July 2020.
Bosi E, Conti M, Vermigli C, Cazzetta G, Peretti E, Cordoni MC, et al. Effectiveness of frequency-modulated electromagnetic neural stimulation in the treatment of painful diabetic neuropathy. Diabetologia. 2005;48(5):817–23.
Wróbel MP, Szymborska-Kajanek A, Wystrychowski G, Biniszkiewicz T, Sieroń-Stołtny K, Sieroń A, et al. Impact of low frequency pulsed magnetic fields on pain intensity, quality of life and sleep disturbances in patients with painful diabetic polyneuropathy. Diabetes Metab. 2008;34(4 Pt 1):349–54.
Weintraub MI, Herrmann DN, Smith AG, Backonja MM, Cole SP. Pulsed electromagnetic fields to reduce diabetic neuropathic pain and stimulate neuronal repair: a randomized controlled trial. Arch Phys Med Rehabil. 2009;90(7):1102–9.
ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000 Feb 29. Identifier NCT03455543, dual field PEMF therapy in lower extremity painful diabetic distal symmetric peripheral neuropathy (RELIEF); 2018 March 6 [cited 2020 July 23]; [about 4 screens]. https://clinicaltrials.gov/ct2/show/NCT03455543. Accessed 23 July 2020.
Weintraub MI, Wolfe GI, Barohn RA, Cole SP, Parry GJ, Hayat G, et al. Static magnetic field therapy for symptomatic diabetic neuropathy: a randomized, double-blind, placebo-controlled trial. Arch Phys Med Rehabil. 2003;84(5):736–46.
Lindblad K, Bergkvist L, Johansson AC. Evaluation of the treatment of chronic chemotherapy-induced peripheral neuropathy using long-wave diathermy and interferential currents: a randomized controlled trial. Support Care Cancer. 2016;24(6):2523–31.
Onesti E, Gabriele M, Cambieri C, Ceccanti M, Raccah R, Di Stefano G, et al. H-coil repetitive transcranial magnetic stimulation for pain relief in patients with diabetic neuropathy. Eur J Pain. 2013;17(9):1347–56.
Swislocki A, Orth M, Bales M, Weisshaupt J, West C, Edrington J, et al. A randomized clinical trial of the effectiveness of photon stimulation on pain, sensation, and quality of life in patients with diabetic peripheral neuropathy. J Pain Symptom Manag. 2010;39(1):88–99.
Zinman LH, Ngo M, Ng ET, Nwe KT, Gogov S, Bril V. Low-intensity laser therapy for painful symptoms of diabetic sensorimotor polyneuropathy: a controlled trial. Diabetes Care. 2004;27(4):921–4.
Paice JA, Shott S, Oldenburg FP, Zeller J, Swanson B. Efficacy of a vibratory stimulus for the relief of HIV-associated neuropathic pain. Pain. 2000;84(2–3):291–6.
Garrow AP, Xing M, Vere J, Verrall B, Wang L, Jude EB. Role of acupuncture in the management of diabetic painful neuropathy (DPN): a pilot RCT. Acupunct Med. 2014;32(3):242–9.
Molassiotis A, Suen LKP, Cheng HL, Mok TSK, Lee SCY, Wang CH, et al. A randomized assessor-blinded wait-list-controlled trial to assess the effectiveness of acupuncture in the management of chemotherapy-induced peripheral neuropathy. Integr Cancer Ther. 2019;18:1534735419836501.
Prinsloo S, Novy D, Driver L, Lyle R, Ramondetta L, Eng C, et al. Randomized controlled trial of neurofeedback on chemotherapy-induced peripheral neuropathy: a pilot study. Cancer. 2017;123(11):1989–97.
Sdrulla D, Guan Y, Raja SN. Spinal cord stimulation: clinical efficacy and potential mechanisms. Pain Pract. 2018;18(8):1048–67.
Shrivastav M, Musley S. Spinal cord stimulation for complex regional pain syndrome. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:2033–6.
Marineo G. Inside the scrambler therapy, a noninvasive treatment of chronic neuropathic and cancer pain: from the gate control theory to the active principle of information. Integr Cancer Ther. 2019;18:1534735419845143.
Barrella M, Toscano R, Goldoni M, Bevilacqua M. Frequency rhythmic electrical modulation system (FREMS) on H-reflex amplitudes in healthy subjects. Eura Medicophys. 2007;43(1):37–47.
Bevilacqua M, Barrella M, Toscano R et al. Disturbances of vasomotion in diabetic (type 2) neuropathy: increase of vascular endothelial growth factor, elicitation of sympathetic efflux and synchronization of vascular flow (vasomotion) during frequency modulated neural stimulation (FREMS). In: 86th annual meeting of the Endocrine Society, p 321, P 2–61 (abstract) (2004).
Ozcan J, Ward AR, Robertson VJ. A comparison of true and premodulated interferential currents. Arch Phys Med Rehabil. 2004;85:409–15.
Pope GD, Mockett SP, Wright JP. A survey of electrotherapeutic modalities: ownership and use in the NHS in England. Physiotherapy. 1995;81:82–91.
Rossi S, Hallett M, Rossini PM, Pascual-Leone A, Safety of TMS Consensus Group. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol. 2009;120(12):2008–399.
Nardone R, Höller Y, Taylor A, Thomschewski A, Orioli A, Frey V, Trinka E, Brigo F. Noninvasive spinal cord stimulation: technical aspects and therapeutic applications. Neuromodulation. 2015;18(7):580–91 (discussion 590–1).
Lee L, Siebner HR, Rowe JB, Rizzo V, Rothwell JC, Frackowiak RSJ, et al. Acute remapping within the motor system induced by low-frequency repetitive transcranial magnetic stimulation. J Neurosci. 2003;23:5308–18.
Pascual-Leone A, Tormos JM, Keenan J, Tarazona F, Cañete C, Catalá MD. Study and modulation of human cortical excitability with transcranial magnetic stimulation. J Clin Neurophysiol. 2009;15:333–43.
Berardelli A, Inghilleri M, Rothwell JC, Romeo S, Currà A, Gilio F, et al. Facilitation of muscle evoked responses after repetitive cortical stimulation in man. Exp Brain Res. 1998;122(1):79–84.
Di Lazzaro V, Oliviero A, Berardelli A, Mazzone P, Insola A, Pilato A, et al. Direct demonstration of the effects of repetitive transcranial magnetic stimulation on the excitability of the human motor cortex. Exp Brain Res. 2002;144(4):549–53.
Pollett HF. Infrared light therapy in the treatment of chronic pain. Today’s Ther Trends. 2002;20:357–71.
Desmet KD, Paz DA, Corry JJ, Eells JT, Wong-Riley MTT, Henry MM, et al. Clinical and experimental applications of NIR-LED photobiomodulation. Photomed Laser Surg. 2006;24:121–8.
Lubart R, Wollman Y, Friedmann H, Rochkind S, Laulicht I. Effects of visible and near infrared lasers on cell cultures. J Photochem Photobiol B. 1992;12(3):305–10.
Wong-Riley MT, Liang HL, Eells JT, Chance B, Henry MM, Buchmann E, et al. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280(6):4761–71.
Brown GC. Nitric oxide regulates mitochondrial respiration and cell functions by inhibiting cytochrome oxidase. FEBS Lett. 1995;369(2–3):136–9.
Lane N. Cell biology: power games. Nature. 2006;443(7114):901–3.
Cotler HB, Chow RT, Hamblin MR, Carroll J. The use of low level laser therapy (LLLT) for musculoskeletal pain. MOJ Orthop Rheumatol. 2015;2(5):00068.
Murillo N, Valls-Sole J, Vidal J, Opisso E, Medina J, Kumru H. Focal vibration in neurorehabilitation. Eur J Phys Rehabil Med. 2014;50(2):231–42.
Rittweger J. Vibration as an exercise modality: how it may work, and what its potential might be. Eur J Appl Physiol. 2010;108:877–904.
Cardinale M, Bosco C. The use of vibration as an exercise intervention. Exerc Sport Sci Rev. 2003;31:3–7.
Wakeling JM, Nigg BM, Rozitis AI. Muscle activity damps the soft tissue resonance that occurs in response to pulsed and continuous vibrations. J Appl Physiol. 2002;93:1093–103.
Pujari AN, Neilson RD, Cardinale M. Effects of different vibration frequencies, amplitudes and contraction levels on lower limb muscles during graded isometric contractions superimposed on whole body vibration stimulation. J Rehabil Assist Technol Eng. 2019;6:2055668319827466.
Zhou W, Benharash P. Effects and mechanisms of acupuncture based on the principle of meridians. J Acupunct Meridian Stud. 2014;7(4):190–3.
Marzbani H, Marateb HR, Mansourian M. Neurofeedback: a comprehensive review on system design, methodology and clinical applications. Basic Clin Neurosci. 2016;7(2):143–58.
Caro XJ, Winter EF. EEG biofeedback treatment improves certain attention and somatic symptoms in fibromyalgia: a pilot study. Appl Psychophysiol Biofeedback. 2011;36:193–200.
de Charms RC. Applications of real-time fMRI. Nat Rev Neurosci. 2008;9:720–9.
Surmeli T, Eralp E, Mustafazade I, Kos IH, Özer GE, Surmeli OH. Quantitative EEG neurometric analysis-guided neurofeedback treatment in postconcussion syndrome (PCS): forty cases. How is neurometric analysis important for the treatment of PCS and as a biomarker? Clin EEG Neurosci. 2017;48(3):217–30.
Kinreich S, Podlipsky I, Jamshy S, Intrator N, Hendler T. Neural dynamics necessary and sufficient for transition into pre-sleep induced by EEG neurofeedback. Neuroimage. 2014;97:19–28.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Andreas Liampas, Martina Rekatsina, Athina Vadalouca and Antonella Paladini have nothing to disclose. Giustino Varrassi and Panagiotis Zis are members of the journal’s Editorial Board.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12738569.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Liampas, A., Rekatsina, M., Vadalouca, A. et al. Non-Pharmacological Management of Painful Peripheral Neuropathies: A Systematic Review. Adv Ther 37, 4096–4106 (2020). https://doi.org/10.1007/s12325-020-01462-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-020-01462-3