Abstract
Introduction
Statins are at the forefront of strategies to manage dyslipidemia, although they are not always well tolerated. At 6–7 months after the drug was supplied, discontinuation rates averaged 30%. Alternate agents to statins have been studied. Some nutraceuticals demonstrated an efficacy in reducing cholesterol concentrations. However, there are no data regarding the use of nutraceuticals in elderly dyslipidemic patients. The purpose of this study was to examine the efficacy, safety, and tolerability of a nutraceutical-based protocol in elderly hypercholesterolemic patients previously intolerant to statins.
Methods
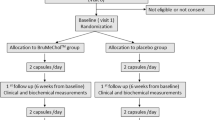
This study was performed as a randomized, prospective, parallel group, single-blind study. Patients were included in the study if they had high total cholesterolemia, high low-density lipoprotein cholesterol (LDL-C), >75 years of age, statin-intolerant, and were refusing other pharmaceutical treatments for hypercholesterolemia. At the baseline visit, eligible patients were randomized to either nutraceutical-combined pill (containing berberine 500 mg, policosanol 10 mg, red yeast rice 200 mg, folic acid 0.2 mg, coenzyme Q10 2.0 mg, and astaxanthin 0.5 mg) or placebo, and the first dose was dispensed. The efficacy, safety, and tolerability of the proposed treatment were fully assessed after 3, 6, and 12 months of treatment.
Results
Out of 106 consecutive patients screened, 80 eligible patients were randomized to receive either nutraceutical-combined pill (40 patients) or placebo (40 patients). No patients were lost and no deaths occurred during the follow-up. There was a statistically significant reduction in total cholesterolemia (−20%), LDL-C (−31%), and insulin resistance (−10%) with nutraceutical treatment. No significant changes were detected for plasma high-density lipoprotein cholesterol (HDL-C). Furthermore, no statistical differences were found between baseline and end-study safety parameters. Medication compliance and tolerability were high.
Conclusion
In this study the authors have demonstrated that combined nutraceuticals significantly reduce cholesterolemia and achieved acceptable plasma LDL-C levels in elderly hypercholesterolemic patients who were previously statin-intolerant. Combined nutraceuticals is also safe and well tolerated in these patients.
Similar content being viewed by others
References
American Heart Association 2011. Heart and Stroke Statistical Update. Available at: http://www.heart.org/HEARTORG/General/Heart-and-Stroke-Association-Statistics_UCM_319064_SubHomePage.jsp. Last accessed November 7, 2011.
Wald NJ, Law MR. A strategy to reduce cardiovascular disease by more than 80%. BMJ. 2003;326: 1419. Erratum in: BMJ. 2003;327:586. BMJ. 2006;60:823.
Yusuf S, Hawken S, Ounpuu S, et al. INTERHEART Study Investigators. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364:937–952.
Lewington S, Whitlock G, Clarke R, et al. Blood cholesterol and vascular mortality by age, sex, and blood pressure: a meta-analysis of individual data from 61 prospective studies with 55,000 vascular deaths Prospective Studies Collaboration Lancet. 2007;70:1829–1839.
Kronmal RA, Cain KC, Omenn GS. Total serum cholesterol levels and mortality risk as a function of age. A report based on the Framingham data. Arch Intern Med. 1993;153:1065–1073.
Vaughan CJ, Gotto AM Jr. Update on statins: 2003. Circulation. 2004;110:886–892.
Afilalo J, Duque G, Steele R, et al. Statins for secondary prevention in elderly patients. J Am Coll Cardiol. 2008;51:37–45.
Thompson PD, Clarkson P, Karas RH. Statin-associated myopathy. JAMA. 2003;289:1682–1690.
Thompson PD, Clarkson PM, Rosenson RS. National Lipid Association Statin Safety Task Force Muscle Safety Expert Panel. An assessment of statin safety by muscle experts Am J Cardiol. 2006;97:69C–76C.
Jackevicius CA, Mamdani M, Tu JV. Adherence with statin therapy in elderly patients with and without acute coronary syndromes. JAMA. 2002;288:462–467.
Heeschen C, Hamm CW, Laufs U, et al. Platelet Receptor Inhibition in Ischemic Syndrome Management (PRISM) Investigators. Withdrawal of statins increases event rates in patients with acute coronary syndromes. For the Platelet Receptor Inhibition in Ischemic Syndrome Management (PRISM) Investigators. Circulation. 2002;105:1446–1452.
Thomas MK, Narang D, Lakshmy R, et al. Correlation between inflammation and oxidative stress in normocholesterolemic coronary artery disease patients ‘on’ and ‘off’ atorvastatin for short time intervals. Cardiovasc Drugs Ther. 2006;20:37–44.
Kalra EK. Nutraceutical — definition and introduction. AAPS PharmSci. 2003;5:E25.
Cicero AFG, Rovati L, Setnikar I. Eulipidemic effects of berberine administered alone or in combination with other natural cholesterol-lowering agents in humans. Arzneimittelforschung. 2007;57:26–30.
Kong W, Wei J, Abidi P, et al. Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nature Med. 2004;10;1344–1351.
Heber D, Yip I, Ashley JM, Elashoff DA, Elashoff RM, Go VL. Cholesterol-lowering effects of a proprietary Chinese red-yeast-rice dietary supplement. Am J Clin Nutr. 1999;69:231–236.
Becker DJ, Gordon RY, Halbert SC, French B, Morris PB, Rader DJ. Red yeast rice for dyslipidemia in statin-intolerant patients: a randomized trial. Ann Intern Med. 2009;150:830–839, W147–9.
Pons P, Rodriguez M, Robaina C, et al. Effects of successive dose increases of policosanol on the lipid profile of patients with type II hypercholesterolaemia and tolerability to treatment. Int J Clin Pharmacol Res. 1994;14:27–33.
Greyling A, De Witt C, Oosthuizen W, et al. Effects of a policosanol supplement on serum lipid concentrations in hypercholesterolaemic and heterozygous familial hypercholesterolaemic subjects. Br J Nutr. 2006;95:968–975.
Janikula M. Policosanol: a new treatment for cardiovascular disease? Altern Med Rev. 2002;7:203–217.
Singh DK, Li L, Porter TD. Policosanol inhibits cholesterol synthesis in hepatoma cells by activation of AMP-kinase. J Pharmacol Exp Ther. 2006;318:1020–1026.
Affuso F, Ruvolo A, Micillo F, et al. Effects of a nutraceutical combination (berberine, red yeast rice and policosanols) on lipid levels and endothelial function randomized, double-blind, placebo-controlled study. Nutr Metab Cardiovasc Dis. 2010;20:656–661.
Ni YX. Therapeutic effect of berberine on 60 patients with type II diabetes mellitus and experimental research. Zhong Xi Yi Jie He Za Zhi. 1988;8707:711–713.
Brusq JM, Ancellin N, Grondin P, et al. Inhibition of lipid synthesis through activation of AMPkinase: an additional mechanism for the hypolipidemic effects of Berberine. J Lipid Res. 2006;47:1281–1288.
Zhang Y, Li X, Zou D, et al. Treatment of type 2 diabetes and dyslipidemia with the natural plant alkaloid berberine. J Clin Endocr Metab. 2008;93:2559–2565.
Pan GY, Huang ZJ, Wang GJ. The antihyperglycaemic activity of berberine arises from a decrease of glucose absorption. Planta Med. 2003;69:632–636.
Kong WJ, Zhang H, Song DQ, et al. Berberine reduces insulin resistance through protein kinase C-dependent up-regulation of insulin receptor expression. Metabolism. 2009;58:109–119.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marazzi, G., Cacciotti, L., Pelliccia, F. et al. Long-term effects of nutraceuticals (berberine, red yeast rice, policosanol) in elderly hypercholesterolemic patients. Adv Therapy 28, 1105–1113 (2011). https://doi.org/10.1007/s12325-011-0082-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-011-0082-5