Abstract
Introduction
The objective of the study was to assess the efficacy of extended-release methylphenidate (Mph-ER) (Medikinet®; MEDICE Arzneimittel Pütter GmbH & Co KG, Iserlohn, Germany) in the pediatric population with attention deficit/hyperactivity disorder (ADHD); a parallel analysis of the validity of various tools for monitoring short-term clinical response to treatment was made.
Methods
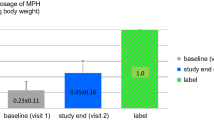
This was a retrospective analysis of 94 children with ADHD who received treatment with Mph-ER. The ADHD Rating Scale-IV (ADHD-RS) was used to assess clinical efficacy. The following neuropsychological tests were used to assess cognitive-attentional efficacy: the faces test, the D2 test, the Magallanes visual attention scale (EMAV; Escalas Magallanes de Attencion Visual), and the Conners’ Continuous Performance Test II (CPT-II). The ADHD-RS scale was completed by the parents at the time of diagnosis and after 3 months of treatment. The tests were taken by patients both without treatment and under the effects of treatment. The results of these variables were transformed into Z values for subsequent analysis.
Results
In all, 84% of the patients lowered their ADHD-RS score with Mph-ER. Regarding the neuropsychological tests, a significant change was seen when the results of patients without treatment were compared with their later results with treatment. When the order of test conditions was reversed (with and then without treatment), the CPT-II was the only test for which there was still a significant difference.
Conclusion
Mph-ER improved attention and self-control from a clinical and cognitive point of view. Of those studied, the CPT-II was the most effective neuropsychological test for monitoring efficacy of Mph in the short-term.
Similar content being viewed by others
References
Connor DF. Stimulants. In: Barkley RA, ed. Attention-Deficit Hyperactivity Disorder: A Handbook for Diagnosis and Treatment. New York: Guilford Press; 2006:608–647.
Barkley RA. A review of stimulant drug research with hyperactive children. J Child Psychol Psychiatry. 1977;18:137–165.
Connor DF, Steingard RJ. New formulations of stimulants for attention-deficit hyperactivity disorder: therapeutic potential. CNS Drugs. 2004;18:1011–1030.
Spencer TJ, Biederman J, Wilens TE, Harding M, O’Donnell D, Griffin S. Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry. 1996;35:409–432.
Wilens TE, Spencer TJ. The stimulants revisited. Child Adolesc Psychiatr Clin N Am. 2000;9:573–603.
The MTA Cooperative Group. A 14-month randomised clinical trial of treatment strategies for attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 1999;56:1073–1086.
The MTA Cooperative Group. Moderators and mediators of treatment response for children with attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 1999;56:1088–1096.
The MTA Cooperative Group. The NIMH MTA follow-up: 24-month outcomes of treatment strategies for attention-deficit/hyperactivity disorder (ADHD). Pediatrics. 2004;113:754–761.
The MTA Cooperative Group. The NIMH MTA follow-up: changes in effectiveness and growth after the end of treatment. Pediatrics. 2004;113:762–769.
Jensen PS, Arnold LE, Swanson JM, et al. 3-year follow-up of the NIMH MTA study. J Am Acad Child Adolesc Psychiatry. 2007;46:989–1002.
Schachter HM, Pham B, King J, Langford S. How efficacious and safe is short-acting methylphenidate for the treatment of attention-deficit disorder in children and adolescents? A meta-analysis. CMAJ. 2001;165:1475.
Fischer R, Schütz H, Grossmann M, et al. Bioequivalence of a methylphenidate hydrochloride extended-release preparation: comparison of an intact capsule and an opened capsule sprinkled on applesauce. Int J Clin Pharmacol Ther. 2006;44:135–141.
Farré-Ribá A, Narbona J. Escalas Para la Evaluación del Déficit de Atención con Hiperactividad (EDAH). Madrid: TEA Ediciones; 1998.
Thurstone LL, Yela M. Caras, Test de Percepción de Diferencias. Adaptación Española. 9° Ed. Madrid: TEA Ediciones; 1991.
Brickenkamp R. D2, Test de Atención. Adaptación Española. Madrid: TEA Ediciones; 1997.
García M, Magaz A. EMAV, Escala Magallanes de Atención Visual. Bilbao: Editorial Grupo Albor Cohs; 2000.
Conners CK. Conners’ Continuous Performance Test (CPT II). New York: MHS; 2002.
Fernández-Jaén A, Martín Fernández-Mayoralas D, Calleja-Pérez B, et al. Efectos del metilfenidato en los procesos cognitivo-atencionales. Uso de los test de ejecución continuada. Rev Neurol. 2008;46(suppl. 1):S47–S49.
Chacko A, Pelham WE, Gnagy EM, et al. Stimulant medication effects in a summer treatment program among young children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2005;44:249–257.
Sinzig JK, Döpfner M, Plück J, et al. Does a morning dose of methylphenidate retard reduce hyperkinetic symptoms in the afternoon? [in German]. Z Kinder Jugendpsychiatr Psychother. 2004;32:225–233.
Döpfner M, Gerber WD, Banaschewski T, et al. Comparative efficacy of once-a-day extendedrelease methylphenidate, two-times-daily immediate-release methylphenidate, and placebo in a laboratory school setting. Eur Child Adolesc Psychiatry. 2004;13(suppl. 1):93–101.
Steele M, Jensen PS, Quinn DM. Remission versus response as the goal of therapy in ADHD: a new standard for the field? Clin Ther. 2006;28:1892–1908.
American Academy of Child and Adolescent Psychiatry. Summary of the practice parameters for the assessment and treatment of children, adolescents and adults with ADHD. J Am Acad Child Adolesc Psychiatry. 1997;36:1311–1317.
Pelham WE, Gnagy EM, Burrows-Maclean L, et al. Once-a-day Concerta methylphenidate versus three-times-daily methylphenidate in laboratory and natural settings. Pediatrics. 2001;107:1–15.
Banaschewski T, Coghill D, Santosh P, et al. Longacting medications for the hyperkinetic disorders. A systematic review and European treatment guideline. Eur Child Adolesc Psychiatry. 2006;15:476–495.
Newcorn JH, Halperin JM, Jensen PS, et al. Symptom profiles in children with ADHD: effects of comorbidity and gender. J Am Acad Child Adolesc Psychiatry. 2001;40:137–146.
Coghill DR, Rhodes SM, Matthews K. The neuropsychological effects of chronic methylphenidate on drug-naive boys with attention-deficit/hyperactivity disorder. Biol Psychiatry. 2007;62:954–962.
Madaan V, Daughton J, Lubberstedt B, Mattai A, Vaughan BS, Kratovchvil CJ. Assessing the efficacy of treatments for ADHD: overview of methodological issues. CNS Drugs. 2008;22:275–290.
Wassenberg R, Hendriksen JG, Hurks PP, et al. Development of inattention, impulsivity, and processing speed as measured by the d2 test: results of a large cross-sectional study in children aged 7–13. Child Neuropsychol. 2008;14:195–210.
Rubio-Morell B, Martín-González R, Herreros-Rodríguez O, et al. Evaluación neuropsicológica de la eficacia del metilfenidato-OROS en el trastorno por déficit de atención/hiperactividad. Rev Neurol. 2008;46:602–608.
Gordon M. The Gordon Diagnostic System. DeWitt, NY: Gordon Systems; 1983.
Gordon M, Barkley RA, Lovett BJ. Tests and Observational Measures. In: Barkley RA (ed). Attention-Deficit Hyperactivity Disorder: A Handbook for Diagnosis and Treatment. New York: Guilford Press; 2006:369–388.
Inoue Y, Inagaki M, Gunji A, et al. Response switching process in children with attentiondeficit-hyperactivity disorder on the novel continuous performance test. Dev Med Child Neurol. 2008;50:462–466.
Losier BJ, McGrath PJ, Klein RM. Error patterns on the continuous performance test in non-medicated and medicated samples of children with and without ADHD: a meta-analytic review. J Child Psychol Psychiatry. 1996;37:971–987.
Coons HW, Klorman R, Borgstedt AD. Effects of methylphenidate on adolescents with a childhood history of ADD: II. Information processing. J Am Acad Child Adolesc Psychiatry. 1987;26:368–374.
Fischer M, Newby RF, Gordon M. Who are the false negatives on continuous performance tests? J Clin Child Psychol. 1995;24:427–433.
Garfinkel BD, Brown WA, Klee SH, et al. Neuroendocrine and cognitive responses to amphetamine in adolescents with a history of attention deficit disorder. J Am Acad Child Adolesc Psychiatry. 1986;25:503–508.
Aman MG. Drugs, learning, and the psychotherapies. In: Werry JS (ed). Pediatric Psychopharmacology: The Use of Behavior Modifying Drugs in Children. New York: Brunner/Mazel; 1978:79–108.
Riccio CA, Waldrop JJ, Reynolds CR, Lowe P. Effects of stimulants on the Continuous Performance Test (CPT): implications for CPT use and interpretation. J Neuropsychiatry Clin Neurosci. 2001;13:326–335.
Musten LM, Firestone P, Pisterman S, Bennett S, Mercer J. Effects of methylphenidate on preschool children with ADHD: cognitive and behavioural functions. J Am Acad Child Adolesc Psychiatry. 1997;36:1407–1415.
Byrne JM, Bawden HN, DeWolfe NA, Beattie TL. Clinical assessment of psychopharmacological treatment of preschoolers with ADHD. J Clin Exp Neuropsychol. 1998;20:613–627.
Pearson DA, Santos CW, Roache JD, et al. Treatment effects of methylphenidate on behavioral adjustment in children with mental retardation and ADHD. J Am Acad Child Adolesc Psychiatry. 2003;42:209–216.
Martin CA, Guenthner G, Bingcang C, et al. Measurement of subjective effects of methylphenidate in 11- to 15-year old children with attentiondeficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2007;17:63–73.
Teicher MH, Lowen SB, Polcari A, et al. Novel strategy for the analysis of CPT data provides new insight into the effects of methylphenidate on attentional states in children with ADHD. J Child Adolesc Psychopharmacol. 2004;14:219–32.
O’Toole K, Abramowitz A, Morris R, Dulcan M. Effects of methylphenidate on attention and nonverbal learning in children with attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1997;36:531–538.
Kupietz SS, Balka EB. Alterations in vigilance performance of children receiving amitriptyline and methylphenidate pharmacotherapy. Psychopharmacology. 1976;50:29–33.
Weis R, Totten SJ. Ecological validity of the Conners’ Continuous Performance Test II in a schoolbased sample. J Psychoeduc Assess. 2004;22:47–61.
Sadeh A, Gruber R, Raviv A. The effects of sleep restriction and extension on school-age children: what a difference an hour makes. Child Dev. 2003;74:444–455.
Trommer BL, Hoeppner JB, Lorber R, Armstrong K. Pitfalls in the use of a continuous performance test as a diagnostic tool in attention deficit disorder. J Dev Behav Pediatr. 1988;9:339–345.
Sostek AJ, Buchsbaum MS, Rapoport JL. Effects of amphetamine on vigilance performance in normal and hyperactive children. J Abnorm Child Psychol. 1980;8:491–500.
Riccio CA, Reynolds CR, Lowe PA. Monitoring medication effects with continuous performance tests. In: Riccio CA (ed). Clinical Applications of Continuous Performance Tests. Somerset, NJ: John Wiley & Sons, Inc; 2001:258–290.
Bannert M, Arbinger RS. Gender-related differences in exposure to and use of computers: results of a survey of secondary school students. Eur J Psychol Educ. 1996;11:269–282.
Concannon PE, Tang YP. Management of attention deficit hyperactivity disorder: a parental perspective. J Paediatr Child Health. 2005;41:625–630.
Sandler AD, Bodfish JW. Open-label use of placebos in the treatment of ADHD: a pilot study. Child Care Health Dev. 2008;34:104–110.
Keulers EH, Hendriksen JG, Feron FJ, et al. Methylphenidate improves reading performance in children with attention deficit hyperactivity disorder and comorbid dyslexia: an unblinded clinical trial. Eur J Paediatr Neurol. 2007;11:21–28.
Kobel M, Bechtel N, Weber P, et al. Effects of methylphenidate on working memory functioning in children with attention deficit/hyperactivity disorder. Eur J Paediatr Neurol. 2008;13:516–523.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fernández-Jaén, A., Fernández-Mayoralas, D., Pardos, A. et al. Clinical and cognitive response to extended-release methylphenidate (Medikinet®) in attention deficit/hyperactivity disorder: Efficacy evaluation. Adv Therapy 26, 1097–1110 (2009). https://doi.org/10.1007/s12325-009-0083-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-009-0083-9