Abstract
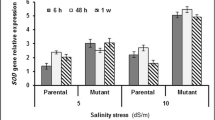
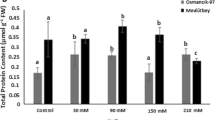
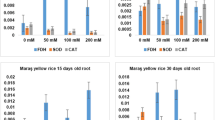
The rice cultivar (Oryza sativa L.) BRS AG, developed by Embrapa Clima Temperado, is the first cultivar designed for purposes other than human consumption. It may be used in ethanol production and animal feed. Different abiotic stresses negatively affect plant growth. Soil salinity is responsible for a serious reduction in productivity. Therefore, the objective of this study was to evaluate the gene expression and the activity of antioxidant enzymes (SOD, CAT, APX and GR) and identify their functions in controlling ROS levels in rice plants, cultivar BRS AG, after a saline stress period. The plants were grown in vitro with two NaCl concentrations (0 and 136 mM), collected at 10, 15 and 20 days of cultivation. The results indicated that the activity of the enzymes evaluated promotes protection against oxidative stress. Although, there was an increase of reactive oxygen species, there was no increase in MDA levels. Regarding genes encoding isoforms of antioxidant enzymes, it was observed that OsSOD3-CU/Zn, OsSOD2-Cu/Zn, OsSOD-Cu/Zn, OsSOD4-Cu/Zn, OsSODCc1-Cu/Zn, OsSOD-Fe, OsAPX1, OsCATB and OsGR2 were the most responsive. The increase in the transcription of all genes among evaluated isoforms, except for OsAPX6, which remained stable, contributed to the increase or the maintenance of enzyme activity. Thus, it is possible to infer that the cv. BRS AG has defense mechanisms against salt stress.


Similar content being viewed by others
References
Ahmad P (2014) Oxidative damage to plants: antioxidant networks and signaling. Elsevier Science, Academic Press, Amsterdam, p 672. ISBN 9780128004609
Ahmad P, Bhardwaj R, Tuteja N (2012) Plant signaling under abiotic stress environment. In: Ahmad P, Prasad MNV (eds) Environmental adaptations and stress tolerance of plants in the era of climate change. Springer, New York, pp 297–323
Alscher RG, Erturk N, Heath LS (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341
Azevedo RA, Alas RM, Smith RJ, Lea PJ (1998) Responses of antioxidant enzymes to transfer from elevated carbon dioxide to air and ozone fumigation, in the leaves androots of wild type and catalase-deficient mutant of barley. Physiol Plant 104:280–292
Bashir K, Nagasaka S, Itai RN, Kobaysahi T, Takahashi M, Nakanishi H (2007) Expression and enzyme activity of glutathione reductase is upregulated by Fe-deficiency in graminaceo us plants. Plant Mol Biol 64:277–284
Bowler C, Montagu MV, Inze D (1992) Superoxide dismutase and stress tolerance. Ann Rev 43:83–116
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cakmak I, Strbac D, Marschner H (1993) Activities of hydrogen peroxide-scavenging enzymes in germinating wheat seeds. J Exp Bot 44:127–132
Chawla S, Jain S, Jain V (2013) Salinity induced oxidative stress and antioxidant system in salt-tolerant and salt-sensitive cultivars of rice (Oryza sativa L.). J Plant Biochem Biotechnol 22:27–34
Chunthaburee S, Dongsansuk S, Sanitchon J, Pattanagul W, Theerakulpisut P (2015) Physiological and biochemical parameters for evaluation and clustering of rice cultivars differing in salt tolerance at seedling stage. Saudi J Biol Sci 23:467–477
Dale JW, Schantz MV (2002) From genes to genomes. Concptes and applications of DNA technology. Wiley, Hoboken
Das P, Seal P, Biswas AK (2016) Regulation of growth, antioxidants and sugar metabolism in rice (Oryza sativa L.) seedlings by NaCl and its reversal by silicon. Am J Plant Sci 7:623–638
Del Rio D, Stewart AJ, Pellegrini N (2005) A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr Metab Cardiovasc 15:316–328
Ferreira ICFR, Abreu RMV (2007) Stress oxidativo, antioxidantes e fitoquímicos. Bioanálise 2:32–39
Giannopolitis CN, Reis SK (1977) Superoxide dismutases: II. Purification and quantitative relationship with water soluble protein in seedlings. Plant Physiol 59:315–318
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidante machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Guan G, Xia D, Liu S (2010) OsAPX4 gene response to several environmental stresses in rice (Oryza sativa L.). Afr J Biotechnol 9:5908–5913
Gupta B, Huang B (2014) Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization. Int J Genomics 701596:18. doi:10.1155/2014/701596
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hernández JA, Ferrer MA, Jiménez A, Barceló AR, Sevilla F (2001) Antioxidant system and O2/H2O2 production in the apoplast of pea leaves: its relation with salt-induced necrotic lesion in minor veins. Plant Physiol 127:817–831
Hu L, Li H, Pang H, Fu J (2012) Responses of antioxidant gene, protein and enzymes to salinity stress in two genotypes of perennial ryegrass (Lolium perenne) differing in salt tolerance. J Plant Physiol 169:146–156
Kaminaka H, Morita S, Nakajima M, Masumura T, Tanaka K (1998) Gene Cloning and Expression of Cytosolic Glutathione Reductase in Rice (Oryza Sativa L.). Plant Cell Physiol 39(12):1269–1280
Khan MH, Panda SK (2008) Alterations in root lipid peroxidation and antioxidative responses in two rice cultivars under NaCl-salinity stress. Acta Physiol Plant 30:81–89
Kim D, Shibato J, Agrawal JK, Fujihara S, Iwahashi H, Kim DH, Shim LS, Rakwal R (2007) Gene transcription in the leaves of rice undergoing salt-induced morphological changes (Oryza sativa L.). Mol Cell 24:45–59
Li C, Bai T, Maa F, Hana M (2010) Hypoxia tolerance and adaptation of anaerobic respiration to hypoxia stress in two Malus species. Sci Hortic 124:274–279
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 22DDCT method. Methods 25:402–408
Magalhães Júnior AM, Fagundes P, Franco DF, Andres A, Nunes CD, Petrini JÁ, Martins JF, Severo A, Moraes OP, MouraF, Streck EA, Aguiar G, Facchinello PH, Huber MP, Krüger TK (2012) BRS AG: cultivar de arroz irrigado desenvolvida para produção de álcool de cereais e/ou alimentação animal. Comunicado Técnico—Embrapa Clima Temperado. https://www.embrapa.br/web/mobile/publicacoes/-/publicacao/1021670/brs-ag-cultivar-de-arroz-irrigado-desenvolvida-como-materia-prima-para-producao-dealcool-de-cereais-eou-alimentacao-animal. Accessed 02 Mar 2016
Mandhania S, Madan S, Sawhney V (2006) Antioxidant defense mechanism under salt stress in wheat seedlings. Biol Plant 50:227–231
Menezes-Benavente L, Teixeira FK, Kamei CLA, Margis-Pinheiro M (2004) Salt stress induces altered expression of genes encoding antioxidant enzymes in seedlings of a Brazilian indica rice (Oryza sativa L.). Plant Sci 166:323–331
Mhamdi A, Queval G, Chaouch S, Vanderauwera S, Van Breusegem F, Noctor G (2010) Catalase function in plants: a focus on Arabidopsis mutants as stress-mimic models. J Exp Bot 61:4197–4220
Mishra P, Bhoomika K, Dubey RS (2013) Differential responses of antioxidative defense system to prolonged salinity stress in salt-tolerant and salt-sensitive Indica rice (Oryza sativa L.) seedlings. Protoplasma 250:3–19
Moraes GP, Benitez LC, Do Amaral MN, Vighi IL, Auler PA, Maia Da LC, Bianchi VJ, Braga EJB (2015) Evaluation of reference genes for RT-qPCR studies in the leaves of rice seedlings under salt stress. Genet Mol Res 14:2384–2398
Morita S, Nakatani S, Koshib T, Masumura T, Ogihara Y, Tanaka K (2011) Differential expression of two cytosolic ascorbate peroxidases and two superoxide dismutase genes in response to abiotic stress in rice. Rice Sci 18:157–166
Murashige T, Skoog FA (1962) Revised medium for rapid growth and bio essays with tobacco tissue culture. Physiol Plant 15:473–497
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascobate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Pitman MG, Lauchli A (2002) Global impact of salinity and agricultural ecosystems. In: Lauchli A, Luttge V (eds) Salinity: environment-plants molecules, 1st edn. Springer, Dordrecht, pp 3–20
Prisco JT, Gomes Filho E (2010) Fisiologia e bioquímica do estresse salino em plantas. In: Ghey HR, da Dias NS, de Lacerda CF (eds) Manejo da salinidade na agricultura: Estudos básicos e aplicados 1st edn. Expressão Gráfica e Editora, Fortaleza, pp 143–159
Sharma P, Bhushan A, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–26
Shigeoka S, Ishikawa T, Tamoi M, Miyagawa Y, Takeda T, Yabuta Y (2002) Regulation and function of ascorbate peroxidase isoenzymes. J Exp Bot 53:1305–1319
Silveira JAG, Silva SLF, Silva EN, Viégas RA (2010) Mecanismos biomoleculares envolvidos com a resistência ao estresse salino em plantas. In: Ghey HR, da Dias NS, de Lacerda CF (eds) Manejo da salinidade na agricultura: Estudos básicos e aplicados 1st edn. Expressão Gráfica e Editora, Fortaleza, pp 161–180
Sofo A, Scopa A, Nuzzaci M, Vitti A (2015) Ascorbate peroxidase and catalase activities and their genetic regulation in plants subjected to drought and salinity stresses. Int J Mol Sci 16:13561–13578
Teixeira FK, Menezes-Benavente L, Margis R, Margis-Pinheiro M (2004) Analysis of the molecular evolutionary history of the ascorbate peroxidase gene family: inferences from the rice genome. J Mol Evol 59:761–770
Teixeira FK, Menezes-Benavente L, Galvão VC, Margis R, Margis-pinheiro M (2006) Rice ascorbate peroxidase gene family encodes functionally diverse isoforms localized in different subcellular compartments. Planta 224:300–314
Turan S, Tripathy BC (2013) Salt and genotype impact on antioxidative enzymes and lipid peroxidation in two rice cultivars during de-etiolation. Protoplasma 250:209–222
Umnajkitikorn K, Faiyue B, Saengnil K (2013) Enhancing antioxidant properties of germinated Thai rice (Oryza sativa L.) cv. Kum Doi Saket with salinity. Rice Res Open Access 1:1–8
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Plant Sci 151:59–66
Vighi IL, Benitez LC, Amaral MN, Moraes GP, Auler PA, Rodrigues GS, Deuner S, Maia LC, Braga EJB (2017) Functional characterization of the antioxidant enzymes in rice plants exposed to salinity stress. Biol Plant 61:1–11
Wang Y, Wisniewski M, Meilan R, Cui M, Webb R, Fuchigami L (2005) Overexpression of cytosolic ascorbate peroxidase in tomato confers tolerance to chilling and salt stress. J Am Soc Hortic Sci 130:167–173
Yamane K, Mitsuya S, Taniguchi M, Miyake H (2010) Transcription profiles of genes encoding catalase and ascorbate peroxidase in the rice leaf tissues under salinity. Plant Prod Sci 13:164–168
Acknowledgements
This work was supported by the Brazilian funding agency Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Rio Grande do Sul (FAPERGS) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments LSP, LCB and AMMJ. Wrote, edited and analyzed the data: TR, LCB, MNA and ILV. Revised the paper: LSP, AMMJ and EJBB. Conducted the experiments: TR, LCB, MNA and ILV. All authors readed the paper and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rossatto, T., do Amaral, M.N., Benitez, L.C. et al. Gene expression and activity of antioxidant enzymes in rice plants, cv. BRS AG, under saline stress. Physiol Mol Biol Plants 23, 865–875 (2017). https://doi.org/10.1007/s12298-017-0467-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-017-0467-2