Abstract
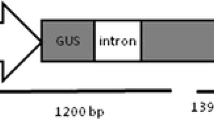
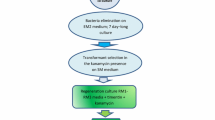
Proliferation of axillary shoot buds and multiple shoot formation in Catharanthus roseus was obtained in 96 % explants on MS medium (3 % sucrose) containing NAA + BA. 2,4-D induced callusing in both, the nodal as well as in leaf segments. Leaf-derived callus was used for transformation with Agrobacterium tumefaciens LBA4404/pBI-S1. Bacterial cell concentration, duration of co-cultivation and acetosyringone concentration influenced transformation efficiency. Under optimal co-cultivation conditions, 98 % of the explants showed GUS expression. PCR based amplification of the transformed and subsequently selected callus tissue indicated the presence of uidA, Gly I and nptII genes.
Similar content being viewed by others
Abbreviations
- AA:
-
1-naphthaleneacetic acid
- BA:
-
N6-benzyladenine
- CH:
-
Casein hydrolysate
- IAA:
-
Indole-3-acetic acid
- Kn:
-
Kinetin
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- RAPD:
-
Random amplification of polymorphic DNA
- PCR:
-
Polymerase chain reaction
References
Batra J, Dutta A, Singh D, Kumar S and Sen J (2004). Growth and terpenoid indole alkaloid production in Catharanthus roseus hairy root clones in relation to left- and right-termini-linked Ri T-DNA gene integration. Pl. Cell Rep. 23: 148–154.
Bhadra R, Vani S and Shanks JV (1993). Production of indole alkaloids by selected hairy root lines of Catharanthus roseus. Biotech. Bioengin. 41: 581–592.
Canel C, Lopes-cardoso MI, Whitmer S, Van der fits L, Pasquali G, Van der heijden R, Hoge JHC and Verpoorte R (1998). Effects of over-expression of strictosidine synthase and tryptophan decarboxylase on alkaloid production by cell cultures of Catharanthus roseus. Planta 205: 414–419.
Cervera M, Pina JA, Juárez J, Navarro L and Peòa L (1998). Agrobacterium-mediated transformation of citrange: factors affecting transformation and regeneration. Pl. Cell Rep. 18: 271–278.
Choi PS, Kim YD, Choi KM, Chung HJ, Choi DW and Liu JR (2004). Plant regeneration from hairy root cultures transformed by infection with Agrobacterium rhizogenes in Catharanthus roseus. Pl. Cell Rep. 22: 828–831.
Doyle JJ and Doyle JL (1990). Isolation of plant DNA from fresh tissue. Focus 12: 13–15.
Edwards K, Johnstone C and Thompson C (1991). A simple and rapid method for the preparation of plant genomic DNA for PCR analysis. Nucleic Acids Res. 19: 1349.
Eilert U, De luca V, Kurz WGW and Constabel F (1987). Alkaloid formation by habituated and tumorous cell suspension cultures of Catharanthus roseus. Pl. Cell Rep. 6: 271–274.
Jefferson RA, Kavanagh TA and Bevan MW (1987). GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO Jour. 13: 3901–3907.
Jung KH, Kwak SS, Kim SW, Lee H, Choi CY and Liu JR (1992). Improvement of catharanthine productivity in hairy root cultures of Catharanthus roseus by using monosaccharides as carbon source. Biotech. Lett. 14: 695–700.
Kim SW, Jung KH, Kwak SS and Liu JR (1994). Relationship between cell morphology and indole alkaloid production in suspension cultures of Catharanthus roseus. Pl. Cell Rep. 14: 23–26.
Li XQ, Liu CN, Ritchie SW, Peng J, Gelvin SB and Hodges TK (1992). Factors influencing Agrobacterium-mediated transient expression of gus A in rice. Pl. Mol. Bio. 20: 1037–1048.
Murashige T and Skoog F (1962). A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plantarum 15: 473–497.
Nammi S, Boini MK, Lodagala SD and Behara RBS (2003). The juice of fresh leaves of Catharanthus roseus Linn. reduces blood glucose in normal and alloxan diabetic rabbits. BMC Complementary and Alternative Medicine, vol. 3, p. 4.
Ross IA (1999). Catharanthus roseus G. Don. In: Medicinal plants of the world. Totowa, New Jersey, Hum. Press 9: 109–118.
Rout GR, Das P, Goel S and Raina SN (1998). Determination of genetic stability of micropropagated plants of ginger using random amplified polymorphic DNA (RAPD) markers. Botanical Bull. Aca. Sin. 39: 23–27.
Salvi ND, George L and Eapen S (2002). Micropropagation and field evaluation of micropropagated plants of turmeric. Pl. Cell Tis. Org. Cult. 68: 143–151.
Singh D, Mehta SS, Neoliya NK, Shukla YN and Mishra M (2003). New possible insect growth regulators from Catharanthus roseus. Curr. Sci. 84: 1184–1186.
Veena, Reddy VS and Sopory SK (1999). Glyoxalase I from Brassica juncea: molecular cloning, regulation and its over-expression confer tolerance in transgenic tobacco under stress. Pl. Jour. 17: 385–395.
Zarate R, Memelink J, Van der heijden R and Verpoorte R (1999). Genetic transformation via particle bombardment of Catharanthus roseus plants through adventitious organogenesis of buds. Biotech. Lett. 21: 997–1002.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srivastava, T., Das, S., Sopory, S.K. et al. A reliable protocol for transformation of Catharanthus roseus through Agrobacterium tumefaciens . Physiol Mol Biol Plants 15, 93–98 (2009). https://doi.org/10.1007/s12298-009-0010-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-009-0010-1