Abstract
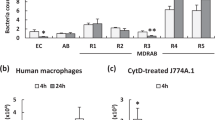
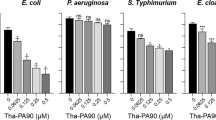
Available antibiotics to treat Acinetobacter baumannii infection is limited due to increasing resistance and the emergence of multiple drug-resistant strains. Hence, discovering effective agents against A. baumannii to reduce the number of infection-related deaths is imperative. In search of novel and alternative antibiotics, the antibacterial function of lipocalin2 (Lcn2) was investigated to treat systemic infections of A. baumannii using a mouse neutropenia model. We observed a significant increase in serum Lcn2 levels upon bacterial injection into the mouse, and the administration of recombinant Lcn2 (rmLcn2) extended their survival. Such protective effects were also observed in rmLcn2-pretreated macrophages, where rmLcn2 reduced the survival of the pathogen inside the macrophages. The underlying molecular mechanism of Lcn2 protection was also investigated. We observed that pretreatment of the Raw-264.7 macrophages with rmLcn2 markedly altered the expression of tonB3, which encodes a component of the transporter for ferrisiderophores in A. baumannii. However, the expression of katG, the gene encoding catalase, remained unaffected. These indicate that Lcn2-mediated defense against the pathogen is related to nutritional immunity rather than reactive oxygen species (ROS) production. Furthermore, the addition of rmLcn2 in infected mice diminished bacterial burden in multiple organs and enhanced the expression of tonB3 in the liver, spleen, and lungs of the infected mice. Increased survival rate due to rmLcn2 treatment declined when the infection model was established using lcn2-defective (lcn2-/-) mice, which indicated the necessity of endogenous Lcn2. Therefore, the antibacterial function of Lcn2 can be exploited to develop an alternative therapeutic agent against A. baumannii.
Similar content being viewed by others
References
Asahara, T., Takahashi, A., Yuki, N., Kaji, R., Takahashi, T., and Nomoto, K. 2016. Protective effect of a synbiotic against multidrug-resistant Acinetobacter baumannii in a murine infection model. Antimicrob. Agents Chemother. 60, 3041–3050.
Asimakopoulou, A., Weiskirchen, S., and Weiskirchen, R. 2016. Lipocalin 2 (LCN2) expression in hepatic malfunction and therapy. Front. Physiol. 7, 430.
Asokan, G.V., Ramadhan, T., Ahmed, E., and Sanad, H. 2019. WHO global priority pathogens list: a bibliometric analysis of Medline-PubMed for knowledge mobilization to infection prevention and control practices in Bahrain. Oman Med. J. 34, 184–193.
Cai, Y., Jogasuria, A., Yin, H., Xu, M.J., Hu, X., Wang, J., Kim, C., Wu, J., Lee, K., Gao, B., et al. 2016. The detrimental role played by lipocalin-2 in alcoholic fatty liver in mice. Am. J. Pathol. 186, 2417–2428.
Cronin, S.J.F., Woolf, C.J., Weiss, G., and Penninger, J.M. 2019. The role of iron regulation in immunometabolism and immunerelated disease. Front. Mol. Biosci. 6, 116.
Dixon, S.J. and Stockwell, B.R. 2014. The role of iron and reactive oxygen species in cell death. Nat. Chem. Biol. 10, 9–17.
Fish, R.E. 1990. Continuing dialogue on use of live dogs in student surgery. J. Am. Vet. Med. Assoc. 196, 1905–1907.
Flo, T.H., Smith, K.D., Sato, S., Rodriguez, D.J., Holmes, M.A., Strong, R.K., Akira, S., and Aderem, A. 2004. Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature 432, 917–921.
Harris, G., Kuo Lee, R., Lam, C.K., Kanzaki, G., Patel, G.B., Xu, H.H., and Chen, W. 2013. A mouse model of Acinetobacter baumanniiassociated pneumonia using a clinically isolated hypervirulent strain. Antimicrob. Agents Chemother. 57, 3601–3613.
Jennings, T., Rusin, M.M., Tessler, H.H., and Cunha-Vaz, J.G. 1988. Posterior sub-Tenon's injections of corticosteroids in uveitis patients with cystoid macular edema. Jpn. J. Ophthalmol. 32, 385–391.
Jeon, J., Ryu, C.M., Lee, J.Y., Park, J.H., Yong, D., and Lee, K. 2016. In vivo application of bacteriophage as a potential therapeutic agent to control OXA-66-Like carbapenemase-producing Acinetobacter baumannii strains belonging to sequence type 357. Appl. Environ. Microbiol. 82, 4200–4208.
Kumar, A., Doan, V.M., Kunkli, B., and Csosz, É. 2021. Construction of unified human antimicrobial and immunomodulatory peptide database and examination of antimicrobial and immunomodulatory peptides in Alzheimer's disease using network analysis of proteomics datasets. Front. Genet. 12, 633050. Kusradze, I., Karumidze, N., Rigvava, S., Dvalidze, T., Katsitadze, M., Amiranashvili, I., and Goderdzishvili, M. 2016. Characterization and testing the efficiency of Acinetobacter baumannii phage vB-GEC_Ab-M-G7 as an antibacterial agent. Front. Microbiol. 7, 1590.
Lim, D., Kim, H.K., Jeong, J.H., Jung, Y.S., Lee, S.E., Jang, H.C., Jung, S.I., Choi, H.S., Rhee, J.H., Lee, S.G., et al. 2020. Lipocalin2 induced by bacterial flagellin protects mice against cyclophosphamide mediated neutropenic sepsis. Microorganisms 8, 646.
Lim, D., Kim, K.S., Jeong, J.H., Kim, H.J., Song, M., Lee, T.H., Kim, J.I., Choi, H.S., Min, J.J., Bumann, D., et al. 2018. The hepcidinferroportin axis controls the iron content of Salmonella-containing vacuoles in macrophages. Nat. Commun. 9, 2091.
Lin, M.F. and Lan, C.Y. 2014. Antimicrobial resistance in Acinetobacter baumannii: from bench to bedside. World J. Clin. Cases 2, 787–814.
Mulani, M.S., Kamble, E.E., Kumkar, S.N., Tawre, M.S., and Pardesi, K.R. 2019. Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: a review. Front. Microbiol. 10, 539.
Munoz-Price, L.S. and Weinstein, R.A. 2008. Acinetobacter infection. N. Engl. J. Med. 358, 1271–1281.
Ondee, T., Gillen, J., Visitchanakun, P., Somparn, P., Issara-Amphorn, J., Dang Phi, C., Chancharoenthana, W., Gurusamy, D., Nita-Lazar, A., and Leelahavanichkul, A. 2019. Lipocalin-2 (Lcn-2) attenuates polymicrobial sepsis with LPS preconditioning (LPS tolerance) in FcGRIIb deficient lupus mice. Cells 8, 1064.
Runci, F., Gentile, V., Frangipani, E., Rampioni, G., Leoni, L., Lucidi, M., Visaggio, D., Harris, G., Chen, W., Stahl, J., et al. 2019. Contribution of active iron uptake to Acinetobacter baumannii pathogenicity. Infect. Immun. 87, e00755–18.
Spapen, H., Jacobs, R., Van Gorp, V., Troubleyn, J., and Honoré, P.M. 2011. Renal and neurological side effects of colistin in critically ill patients. Ann. Intensive Care 1, 14.
Stokes, J.M., Yang, K., Swanson, K., Jin, W., Cubillos-Ruiz, A., Donghia, N.M., MacNair, C.R., French, S., Carfrae, L.A., Bloom-Ackermann, Z., et al. 2020. A deep learning approach to antibiotic discovery. Cell 180, 688–702.
Thompson, M.G., Black, C.C., Pavlicek, R.L., Honnold, C.L., Wise, M.C., Alamneh, Y.A., Moon, J.K., Kessler, J.L., Si, Y., Williams, R., et al. 2014. Validation of a novel murine wound model of Acinetobacter baumannii infection. Antimicrob. Agents Chemother. 58, 1332–1342.
Thummeepak, R., Kitti, T., Kunthalert, D., and Sitthisak, S. 2016. Enhanced antibacterial activity of Acinetobacter baumannii bacteriophage ØABP-01 endolysin (LysABP-01) in combination with colistin. Front. Microbiol. 7, 1402.
Trouplin, V., Boucherit, N., Gorvel, L., Conti, F., Mottola, G., and Ghigo, E. 2013. Bone marrow-derived macrophage production. J. Vis. Exp. 81, e50966.
Uribe-Querol, E. and Rosales, C. 2017. Control of phagocytosis by microbial pathogens. Front. Immunol. 8, 1368.
Windom, R.E. 1988. AIDS update. Ky. Nurse 36, 6–7.
Xu, M.J., Feng, D., Wu, H., Wang, H., Chan, Y., Kolls, J., Borregaard, N., Porse, B., Berger, T., Mak, T.W., et al. 2015. Liver is the major source of elevated serum lipocalin-2 levels after bacterial infection or partial hepatectomy: a critical role for IL-6/STAT3. Hepatology 61, 692–702.
Zhang, W., Yang, S., Cui, L., and Zhang, J. 2016. Neutrophil gelatinase-associated lipocalin worsens ischemia/reperfusion damage of kidney cells by autophagy. Ren. Fail. 38, 1136–1140.
Acknowledgements
This work was supported by the National Research Foundation of Korea grant funded by the Korean government (2021R1F1A1060072 and 2019M3E5D5066666), and by the Hankuk University of Foreign Studies Research Fund (of 2020). M.S. was supported by a National Research Foundation of Korea grant funded by the Korean government (MSIP) (2016R1D1A1B01008960). D.L. was supported by a National Research Foundation of Korea grant funded by the Korean government (MSIP) (NRF-2019R1C1C1003999).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All mouse experiments were carried out following guidelines of the Institutional Mouse Use and Care Committee of Chonnam National University (CNU IACUC-H-2021-15).
Additional information
Conflict of Interest
The authors have no conflict of interest to report.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Lim, D., Park, SJ., Kim, H.Y. et al. Lipocalin2 as a potential antibacterial drug against Acinetobacter baumannii infection. J Microbiol. 60, 444–449 (2022). https://doi.org/10.1007/s12275-022-2007-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-022-2007-1